Пример 17. Вычислить окислительно-восстановительный потенциал, если к 15,0 мл 0,20 М раствора KMnO4 добавили 50,0 мл 0,10 М раствора Na2SO3 при рН = 1.
Решение. При смешении растворов протекает реакция (см. пример 16). Рассчитаем количество вещества эквивалента в обоих растворах до сливания:
 n (
n ( KMnO4) = 15,0 ∙ 10–3 ∙ 1,00 = 15,0 ∙ 10–3 моль экв
KMnO4) = 15,0 ∙ 10–3 ∙ 1,00 = 15,0 ∙ 10–3 моль экв
 n (
n (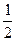 Na2SO3) = 50,0 ∙ 10–3 ∙ 0,20 = 10,0 ∙ 10–3 моль экв.
Na2SO3) = 50,0 ∙ 10–3 ∙ 0,20 = 10,0 ∙ 10–3 моль экв.
Следовательно, KMnO4 взят в избытке. После протекания реакции в растворе будут содержаться продукты реакции и остаток KMnO4. Значит, величина потенциала будет определяться присутствием окислительно-восстановительной пары MnO4–/Mn2+. Рассчитаем количества этих ионов:
n (
 ) = 15,0 ∙ 10–3 – 10,0 ∙ 10–3 = 5,0 ∙ 10–3моль экв Þ
) = 15,0 ∙ 10–3 – 10,0 ∙ 10–3 = 5,0 ∙ 10–3моль экв Þ
n ( ) = 1,0 ∙ 10–3моль
) = 1,0 ∙ 10–3моль
n ( Mn2+) = 10,0 ∙ 10–3 моль экв Þ n (Mn2+) = 2,0 ∙ 10–3моль.
Mn2+) = 10,0 ∙ 10–3 моль экв Þ n (Mn2+) = 2,0 ∙ 10–3моль.
Объем раствора после смешения составит
15,0 + 50,0 = 65,0 мл = 65 ∙ 10–3 л.
Рассчитаем молярные концентрации ионов:
С ( ) =1,0 ∙ 10–3/ 65 ∙ 10–3 = 0,0154 моль/л
) =1,0 ∙ 10–3/ 65 ∙ 10–3 = 0,0154 моль/л
С (Mn2+) =2,0 ∙ 10–3/ 65 ∙ 10–3 = 0,0308 моль/л
C (H+) = 10– pH = 10–1 = 0,1 моль/л.
Рассчитаем окислительно-восстановительный потенциал раствора по уравнению Нернста:
E =  + 0,059 / 5 ∙ lg (
+ 0,059 / 5 ∙ lg ( ) =
) =
= 1,51 + 0,059 / 5 ∙ lg(0,0154 ∙ (0,1)8/ 0,0308) = 1,49 В.
Расчет значения Е при протекании конкурирующих реакций осаждения
Пример 18. Вычислить окислительно-восстановительный потенциал пары  /Hg в присутствии ионов Cl–, приняв концентрацию ионов Cl– равной 0,1 моль/л.
/Hg в присутствии ионов Cl–, приняв концентрацию ионов Cl– равной 0,1 моль/л.
Решение. В присутствии ионов Cl– в растворе образуется малорастворимое соединение Hg2Cl2. Полуреакцию
Hg2Cl2 + 2 e = 2Hg + 2Cl–
можно представить как сочетание реакций окисления-восстановления и осаждения:
 + 2 e = 2Hg (4.2)
+ 2 e = 2Hg (4.2)
 + 2Cl– = Hg2Cl2
+ 2Cl– = Hg2Cl2
Найдем по справочнику [1; 2, с. 24, 49] необходимые константы:
 = 0,792 В
= 0,792 В
 = 1,3∙10–18
= 1,3∙10–18
Ионы ртути (I) появляются в растворе только за счет растворимости Hg2Cl2. Найдем их равновесную концентрацию из величины произведения растворимости и заданной концентрации хлоридов:
[  ] =
] =  / [Cl–]2 = 1,3 ∙ 10–18/(0,1)2 =1,3 ∙ 10–16 моль/л
/ [Cl–]2 = 1,3 ∙ 10–18/(0,1)2 =1,3 ∙ 10–16 моль/л
Запишем уравнение Нернста для полуреакции (4.2) и подставим имеющиеся данные:

Расчет значения Е при протекании конкурирующих реакций комплексообразования
Пример 19. Вычислить окислительно-восстановительный потенциал пары Cd2+/Cd в растворе, полученном смешением 40 мл 0,2 моль/л CdCl2 и 60 мл 4 моль/л KCN.
Решение. Окислительно-восстановительный потенциал пары Cd2+/Cd рассчитывается по уравнению Нернста:
 =
=  + 0,059 / 2 ∙ lg [Cd2+].
+ 0,059 / 2 ∙ lg [Cd2+].
Рассчитаем концентрации веществ после смешения:


После смешения протекает реакция комплексообразования
Cd2+ + 4CN– ↔ 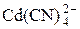
Равновесная концентрация ионов Cd2+ определяется процессом комплексообразования, протекающим в присутствии избытка CN–. Равновесие образования комплекса 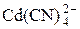 характеризуется константой устойчивости:
характеризуется константой устойчивости:
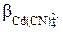 =
=  = 1,29 ∙ 1017
= 1,29 ∙ 1017
При значительном избытке лиганда и достаточно большом значении константы устойчивости можно принять, что
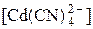 =
= 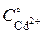 = 0,08 моль/л
= 0,08 моль/л
[CN–] = 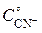 – 4 ∙
– 4 ∙ 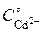 = 2,4 – 4 ∙ 0,08 = 2,08 моль/л
= 2,4 – 4 ∙ 0,08 = 2,08 моль/л
Подставим эти данные в выражение для константы устойчивости, выразим равновесную концентрацию ионов Cd2+ и рассчитаем ее:

Найдем по справочнику  = – 0,402 В.
= – 0,402 В.
Отсюда
 = – 0,402 + 0,059/2 ∙ lg 3,31 ∙ 10–20 = – 0,977 В.
= – 0,402 + 0,059/2 ∙ lg 3,31 ∙ 10–20 = – 0,977 В.
КОМПЛЕКСООБРАЗОВАНИЕ
Основные понятия
Реакции комплексообразования – это реакции с переносом электронных пар и образованием связей по донорно-акцепторному механизму. В них участвуют металл-комплексообразователь (центральный атом, ЦА) и лиганды (L). В результате протекания реакции образуются комплексные соединения (комплексы).
Комплекс – это сложная частица, состоящая из составных частей, способных к автономному существованию.
Состав комплекса:

В состав лиганда входят донорные атомы – атомы, которые связывают лиганд с центральным атомом, например, атомы O, N, S и др.
Комплексообразователь характеризуют координационным числом (КЧ), а лиганд – дентатностью.
Координационное число центрального атома – это число мест во внутренней сфере комплекса, которые могут быть заняты лигандами.
Дентатность лиганда – это число донорных атомов лиганда, которые образуют связи с ЦА.






