ѕример 1. —колько граммов 30 % (по массе) раствора NaCl надо добавить к 300 г воды, чтобы получить 10 % раствор соли? (ответ: 150 г).
| ƒано | –ешение |
| w1(NaCl) = 30 % | ћассова€ дол€ (w) Ц отношение количества граммов растворенного вещества к массе раствора.
|
| w2(NaCl) = 10 % | |
| m¢(H2O) = 300 г | |
| m1(NaCl) =? |
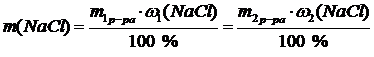 ††††††††††††
††††††††††††
m2(р-ра) = m1(р-ра) + m¢(H2O) = m + 300
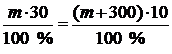
30m = 10m + 3000
20m = 3000
m = 150.
о твет: чтобы получить 10 % раствор соли, необходимо вз€ть 150 г 30 % раствора NaCl.
ѕример 3. ¬ычислите мол€рную, нормальную и мол€льную концентрации 16 %-го раствора хлорида алюмини€ плотностью 1,149 г/см3. †ќбъем раствора 4 л.
| ƒано | –ешение |
| w(Al—l3) = 16 % | ћассова€ дол€ (w) Al—l3 Ц отношение количества граммов растворенного вещества к массе раствора. ѕоэтому расчет навески Al—l3 можно проводить по следующей формуле: |
| r(р-ра) = 1,149 г/см3 | |
| Cм (Al—l3) =? | |
| Cн (Al—l3) =? | |
| Cm(Al—l3) =? |
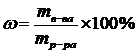 , m(р-ра)= V(р-ра) ´ r(р-ра) Þ
, m(р-ра)= V(р-ра) ´ r(р-ра) Þ 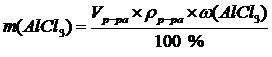 †††††††††††††††
†††††††††††††††
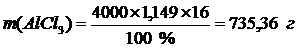 .
.
1. ћол€рна€ концентраци€ Ц число молей растворенного вещества в 1 л раствора:
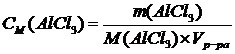
 .
.
††† 2. Ќормальна€ концентраци€ Ц число эквивалентов растворенного вещества в 1 л раствора††††††† ††††††††††††††††††††††††††††††††††
††††††††††††††††††††† ††††††††††††††††††††††††††††m (AlCl3 )†
†††††††††††††††††††††††† ††††— N = ††††___________________ ††††††††††††††
†††††††††††††††††††††††††††††††††††† ††††1/3 †ћ(AlCl3 ) х V (р-ра)
††††††††††††††††††††††††††
††††††††††††††††††† ††††††††††††††††††††††††† † 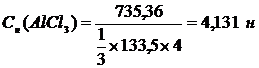
†3. ћол€льна€ концентраци€ Ц показывает, сколько молей растворенного вещества содержитс€ в 1 килограмме растворител€.
ћассу Al—l3 в 1000 г воды (растворител€) находим из соотношени€:
| (100 Ц 16) г Ќ2ќ | Ц | 16 г Al—l3 |
| 1000 г Ќ2ќ | Ц | m г Al—l3 |
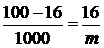 Þ
Þ 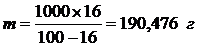 .
.
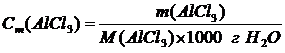 ,† [моль/1 кг Ќ2ќ]
,† [моль/1 кг Ќ2ќ]
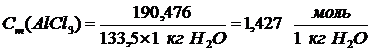 .
.
ѕример 4. –аствор содержащий 0,512 г неэлектролита в 100 г бензола, кри≠сталлизуетс€ при 5,296 ∞—. “емпература кристаллизации бензола 5,5 ∞—. риоскопическа€ константа 5,1 ∞. ¬ычислите мольную массу рас≠творенного вещества.
| ƒано | –ешение |
| m(X) = 0,512 г | —огласно 2 закону –аул€ понижение температуры кристаллизации раствора (Dt) по сравнению с температу≠рами кристаллизации выражаютс€ уравнением
|
| m(C6H6) = 100 г | |
| t(р-р) = 5,296 ∞— | |
| t(C6H6) = 5,5 ∞— | |
| K = 5,1 ∞ | |
| M(X) =? |
где K Ц криоскопическа€ посто€нна€.
m и ћ Ц соответственно масса растворенного вещества и его мольна€ масса; m1Цмасса растворител€.
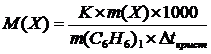
Dt = t(C6H6) Ц t(р-р)
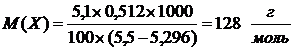
ќтвет: мольна€ масса рас≠творенного вещества 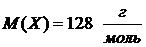 .
.
ѕример 5. –ассчитайте степень гидролиза и значение рЌ в 0,1 ћ растворе ацетата натри€, 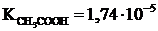 .
.
| ƒано | –ешение |
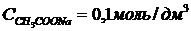
| CH3COONa + H2O Û CH3COOH + NaOH CH3COOЦ + H2O Û CH3COOH + OHЦ 1. онстанта гидролиза соли:
|
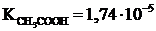
| |
| h =? | |
| pH =? |
2. —тепень гидролиза соли:
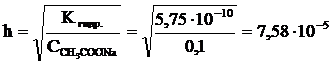 †или 0,00758 %.
†или 0,00758 %.
3. ћол€рна€ концентраци€ гидроксид-ионов:
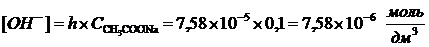
4. ¬одородный показатель:
pH = 14 + lg[ќЌЦ] = 14 + lg7,58´10Ц6 = 8,88
|
|
|
ќтвет: степень гидролиза h = 0,00758 %, pH = 8,88.
ѕример 6. ќбразуетс€ ли осадок хлорида свинца, если смешать равные объемы растворов сол€ной кислоты с концентрацией 0,02 моль/дм3 и нитрата свинца с мол€рной концентрацией 0,01 моль/дм3?
–ешение
1. Pb(NO3)2 + 2HCl Ѓ PbCl2¯ + HNO3;
2. PbCl2 (тв.) Û Pb2+ + 2ClЦ;
3. ѕ–(PbCl2) =[Pb2+] . [ClЦ]2 = 1,6 . 10Ц5;
4. —оль и кислота Ц сильные электролиты:
Pb(NO3)2 Û Pb2+ + 2NO3Ц;
HCl Û Ќ+ + ClЦ;
поэтому: —0(Pb2+) = 0,01 моль/дм3; —0(ClЦ) = 0,02 моль/дм3.
ѕри сливании общий объем раствора увеличилс€ вдвое. —ледовательно, после сливани€ растворов концентрации ионов уменьшились вдвое и стали равны:
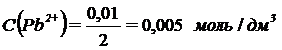 ;
;
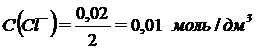 ;
;
5. ѕс = —(Pb2+) . —2(ClЦ) = 0,005 . (0,01)2 = 5 . 10Ц7;
6. 5 . 10Ц7 < 1,6 . 10Ц5; ѕс < ѕ–
ќтвет: полученный раствор €вл€етс€ ненасыщенным, осадок не образуетс€.
«адание 4.





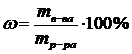
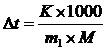 ,†††††††††††††††††
,†††††††††††††††††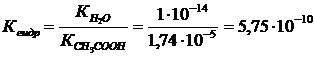 .
.
