’имическа€ св€зь Ц форма выигрыша энергии при переходе вещества из атомарного в молекул€рное состо€ние.
“ри основных типа химической св€зи: ковалентна€, ионна€ и металлическа€.
“еоретические методы изучени€ химической св€зи: метод валентных схем (ћ¬—), метод молекул€рных орбиталей (ћћќ), теори€ кристаллического пол€ (“ ѕ).
ѕример 1. ќпределить тип иона
у катионов: —а2+, Ge2+, Fe2+, Zn2+.
–ешение.
1. Ќеобходимо записать электронные формулы элементов.
2. — внешней электронной оболочки атома необходимо убрать такое число электронов, которое бы соответствовало зар€ду катиона.
3. ѕодсчитать число электронов, оставшеес€ на внешней оболочке катиона, и определить тип иона.
у анионов: F−, Cl−, Br−, J−. |
|
»з таблицы видно, что повтор€етс€ вс€ последовательность решени€ предыдущего примера. ќднако в данном случае необходимо прибавить такое число электронов, которое бы соответствовало зар€ду аниона.
¬ыводы.
1. “ип иона у всех простых анионов 8  .
.
2. ¬ общем случае тип иона соответствует числу электронов на внешней оболочке анионов и катионов. »сключение составл€ют ионы типа (18 + п).
ѕример 2. ќбъ€снить изменение окраски в р€ду сле≠дующих соединений: a) AgCl (белый), AgJ (желтый); б) –bCl2 (белый), PbS (черный).
–ешение.
ѕервоначально рассмотрим изменение окраски в р€ду галогенидов серебра. —оединени€ содержат в своем составе один и тот же катион - Ag+, а потому изменение окраски св€зано только с деформируемостью анионов хлора и йода. ƒл€ доказательства детально обсудим свойства анионов (зар€д, радиус, тип иона).
| —ое≠- | «ар€д | –адиус | “ип | ƒеформи≠ | —тепень | ќкраска |
| дине-≠ | аниона | иона | иона | руемость | ионности | |
| ние | иона | св€зи | ||||
| –b—12 PbS | -1 -2 | увеличи≠ваетс€ | 8  8
8 
| увеличи≠ваетс€ | умень≠шаетс€ | более интен≠сивна€ |
ƒва свойства анионов хлора и йода (зар€д и тип иона) одинаковы, наблюдаетс€ лишь увеличение радиуса при переходе от хлора к йоду. ”величение же радиуса аниона приводит к увеличению его деформируемости и соответственно − пол€ризации ионов, что приводит к уменьшению степени ионности св€зи в молекуле AgJ, отчего соединение приобретает окраску.
»так, йодиды в соединени€х с одинаковыми катионами должны быть чаще окрашены, чем соответствующие хлориды, сходна€ закономерность характерна дл€ перехода по р€ду оксиды - сульфиды: сульфиды окрашены, как правило, интенсивнее, чем соответствующие оксиды. |
¬ывод. ”величение прежде всего отрицательного зар€да аниона в р€ду CI−, S2− способствует увеличению деформируемости S2−, уменьшению степени ионности св€зи в р€ду –№—12 Ч PbS и по€влению черной окраски у PbS. јналогичным образом можно объ€снить, почему гидроксиды металлов (производные Cd2+, Fe2+, Mn2+, Pb2+, Sn2+, Jn3+, Bi3+) имеют менее глубокое окрашивание, чем соответствующие оксиды (это св€зано с меньшей деформируемостью ќЌ− по сравнению с O2−).
ѕример 3. ќпределить закономерности изменени€ силы кислот и оснований по периоду и по группе.
–ешение.
»зменение кислотно-основных свойств гидроксидов по периоду рассмотрим на примере элементов III периода системы ƒ.». ћенделеева: с этой целью разделим элементы периода на металлы и неметаллы; отметим основные свойства ионов элементов (зар€д, равный номеру группы, сравнительный радиус, тип иона); уменьшение или увеличение пол€ризующего действи€ Ёn+ на ќ2−, соответственно увеличение или уменьшение степени ионности св€зи, согласу€ все это с характеристикой силы оснований и кислот по периоду:
а) ионы Na+ Mg2+ Al3+
 Z иона возрастает Z иона возрастает
 R иона уменьшаетс€
“ип иона одинаков Ц 8 ē R иона уменьшаетс€
“ип иона одинаков Ц 8 ē
 рэп+ увеличиваетс€
—тепень ионности рэп+ увеличиваетс€
—тепень ионности
 св€зи Ёn+ − ќ2− уменьшаетс€ св€зи Ёn+ − ќ2− уменьшаетс€
|
| основные свойства гидроксидов | NaOH сильное основание | Mg(OH)2 основание средней силы | ј1(ќЌ)3 амфотерный гидроксид (слабое основание) | |
 ”меньшаетс€ ”меньшаетс€
| ||||
б) ионы Si4+ P5+ S6+ Cl7+
 Z иона возрастает Z иона возрастает
 R иона уменьшаетс€
“ип иона одинаков - 8 R иона уменьшаетс€
“ип иона одинаков - 8 
 рэп+ увеличиваетс€
—тепень ионности рэп+ увеличиваетс€
—тепень ионности
 св€зи Ёn+ − ќ2− уменьшаетс€ св€зи Ёn+ − ќ2− уменьшаетс€
| ||||
| кислотные свойства гидроксидов | H2SiO3 слаба€ кислота | Ќ3–ќ4 кислота средней силы | H2SO4 сильна€ кислота | HClO4 сама€ сильна€ кислота |
 ”величиваетс€ ”величиваетс€
|
¬ывод. —тепень ионности химической св€зи Ёn+ − ќ2− и кислотно-основные свойства гидроксидов элементов одного и того же периода завис€т от двух свойств ионов: зар€да и радиуса. »зменение кислотно-основных свойств гидроксидов по подгруппе на примере элементов II A -группы (основные свойства) и некоторых элементов VI A -группы (кислотные свойства) показано в таблицах:
| »он | Z | R иона | “ип | P Ёn+1 | —тепень | ќснова≠ | —ила |
| иона | иона | ионности | ние | осно≠ | |||
| св€зи Ёn+−ќ2− | вани€ | ||||||
| Mg2+ | +2 | увели≠чиваетс€ | 8ē | умень≠шаетс€ | Mg(OH)2 | воз≠растает | |
| —а2+ | +2 | 8ē | увели≠- | —а(ќЌ)2 | |||
| Sr2+ | +2 | 8ē | чиваетс€ | Sr(OH)2 | |||
| Ba2+ | +2 | 8ē | ¬а(ќЌ)2 |
|
|
|
|
¬ывод. —тепень ионности химической св€зи Ёn+ − ќ2−и кислотно-основные свойства гидроксидов элементов в под- групппе периодической системы завис€т от радиусов ионов.
“еори€ валентных св€зей (¬—) предполагает участие в образовании ковалентных св€зей не только "чистых" атомных орбиталей (јќ), но и "смешанных", так называемых гибридных јќ. ѕри гибридизации первоначальна€ форма и энерги€ орбиталей (электронных облаков) взаимно измен€ютс€, и образуютс€ орбитали (облака) новой одинаковой формы и одинаковой энергии. „исло гибридных орбиталей g равно числу исходных.
| “ип | »сходные | “ип | „исло | ѕространствен≠ |
| моле≠ | орбитали | гибри≠ | гибридных | на€ конфигу≠ |
| кул | атома ј | дизации | орбиталей | раци€ молекул |
| атома ј | ||||
| ј¬2 | s + p | sp | линейна€ | |
| ј¬3 | s + 2 р | sp 2 | треугольна€ | |
| ј¬4 | s + 3 p | sp 3 | тетраэдрическа€ |
ѕример 4. ќпределить тип св€зей в Ќ2, —1, —аќ, ј1—13, CS2.
–ешение.
ƒл€ определени€ типа св€зи необходимо:
1. ¬ыписать (из таблицы в приложении) и сопоставить значени€ относительной электроотрицательности (ќЁќ) дл€ атомов, вход€щих в состав соединений, на основе сопоставлени€ вычислить ј≈ дл€ них.
2. —делать на основании вычисленного Δ≈ и состава соединени€ вывод о типе св€зи в данном соединении:
а) дл€ совокупности атомов ЌЧЌ, —ЧS Δ≈ = 0, следова≠тельно, св€зь между данными атомами ковалентно-непол€рна€
б) дл€ совокупности атомов Ч—1, ј1Ч—1, —аЧќ Δ≈ > ќ, но к ионному соединению следует отнести лишь —1, так как известно, что лишь галогениды щелочных и щелочно-земельных металлов образуют ионные соединени€. ќбразование же таких многозар€дных катионов, как ј13+, и анионов, как ќ2−, энергетически невыгодно, а потому ј1—13 и —аќ - ковалентно-пол€рные соединени€.
¬следствие волновых свойств электронов чистых типов св€зей нет. ѕоэтому по разности в электроотрицательности атомов (соедин€ющих молекулу) чаще всего суд€т о степени ионности химической св€зи. —огласно ѕолингу, чем больше разница в электроотрицательности атомов (вход€щих в состав молекулы), тем больше степень ионности химической св€зи.
ѕример 5. Ќайти геометрию молекул H2Se, ¬е—12.
–ешение.
ƒл€ определени€ геометрии ковалентных молекул необходимо:
1. ¬ыписать концовки электронных формул атомов, вход€≠щих в состав молекул.
2. –аспределить электроны многовалентного атома по атом≠ным орбитал€м согласно правилу √унда и про€вл€емой им валентности в данном соединении и определить форму взаимодействующих электронных облаков атома.
3. Ќайти ориентацию взаимодействующих электронных облаков многовалентного атома и распределение химических св€зей в пространстве, т.е. геометрию молекулы.
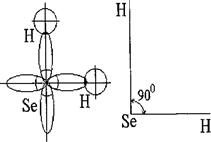
¬ состав молекулы H2Se вход€т атомы водорода и селена, концовки их электронных формул будут: Ќ - 1 s 1, Se - 4s24p4.
–аспределим электроны Se по атомным орбитал€м:

“ак как Se в H2Se двухвалентен, то, следовательно, валентными будут лишь два р -электрона на 4 р -подуровне.
—огласно квантовой механике два p -облака одного и того же атома (в данном случае селена) будут ориентированы в пространстве под углом 90∞. —ледовательно, s -облака двух атомов водорода будут перекрыватьс€ двум€ р -облаками по схеме:

|
|

| 
|
”гол между двум€ химическими св€з€ми будет равен 90∞, и поэтому молекула имеет угловую геометрическую конфигурацию.
Ќетрудно сделать вывод, что если многовалентный атом молекулы трехвалентен и в образовании св€зи участвуют 3 р -облака, то они также будут ориентированы под углом 90∞ относительно друг друга, и молекула будет иметь в этом случае пирамидальную конфигурацию, как у –Ќ3:
|
|
|

ѕримечание. ¬озможно отклонение ориентации нескольких р -облаков от угла 90∞. “ак, угол между двум€ св€з€ми ќ-Ќ в молекуле воды равен 104∞28/ ќтклонение от угла 90∞ объ€сн€етс€ пол€рностью св€зи ќ-Ќ, в результате чего у атома водорода по€вл€етс€ положительный зар€д; отталкивание этих зар€дов увеличивает угол между св€з€ми.
¬ывод. ≈сли в образовании химической св€зи участвуют несколько одинаковых электронных облаков одного и того же атома, то геометрическа€ конфигураци€ молекул асимметрична (угловые, пирамидальные молекулы).
| атомов водорода будут перекрыватьс€ двум€ р-облаками по схеме: |

|

|
ћолекула ¬е—12: в состав этой молекулы вход€т атомы хлора и берилли€, и концовки их электронных формул выгл€д€т: CI - 3s23p5; Be - 2s2.
–аспределим электроны берилли€ по јќ 2-го энергетического уровн€ с учетом его валентности в ¬е—12:

¬алентными электронами у берилли€ будут s - и р -электроны. Ќо известно, что во всех случа€х, когда в образовании химических св€зей у данного атома участвуют электроны разных подуровней, наблюдаетс€ гибридизаци€, или смешение электронных облаков. √ибридизаци€ одного s - и одного р -электронных облаков приводит к образованию двух гибридных облаков - g2. Ёто sp -гибридизаци€.
—хема гибридизации электронных облаков Be:

|

|

|
ќтносительно друг друга гибридные облака всегда ориентируютс€ симметрично. ¬ данном случае симметричность достигаетс€, если между двум€ g -облаками будет угол 180∞. ƒве химические св€зи, образованные путем перекрыти€ двух р -облаков двух атомов хлора, двух g -облаков атома берилли€, расположены по пр€мой и противоположно направлены. ѕоэтому молекулы с этим видом гибридизации линейны:

Ќетрудно представить, что если у атома в результате sp2 или sp 3-гибридизации участвуют соответственно g3- и g 4-облака, то их симметричное распределение в пространстве достигаетс€, если в первом cлучае угол между ними будет 120∞, а во втором - 109∞28/ (треугольна€ и тетраэдрическа€ молекулы).
¬ывод. ”частие гибридных облаков в химической св€зи приводит к возникновению молекул с симметричной геометри≠ческой конфигурацией (линейна€, треугольна€, тетраэдрическа€ и т.д.).
–азличают два типа ковалентных молекул: непол€рные (дипольный момент молекулы μ м равен 0), пол€рные молекулы (дипольный момент μ м > 0). Ќо дипольный момент молекулу μ только дл€ двухатомной молекулы совпадает по значению и направлению с μ св. ¬ общем случае дипольный момент- молекулы (μ м) есть геометрическа€ сумма дипольных моментов μ св всех св€зей. —ледовательно, дл€ того чтобы определить дипольный момент молекулы, необходимо знать: а) пол€рность химических св€зей (т.е. Δ≈ св€зей равен нулю или больше нул€); б) геометрическую конфигурацию молекул. «десь можно встретитьс€ со следующим: если дипольный момент μ св св€зей равен нулю, то молекула в целом непол€рна€ (μ м равен нулю); если | μ св больше нул€, то тип молекулы будет определ€тьс€ всецело геометрической конфигурацией молекулы. ≈сли геометри€ молекулы асимметрична, то дипольные моменты μ св св€зей складываютс€ по правилу параллелограмма и дипольный момент молекулы μ м больше нул€, т.е. молекула пол€рна.
≈сли геометрическа€ конфигураци€ симметрична, “о дипольный момент μ м молекулы равен 0, т.е. молекула непол€рна€.
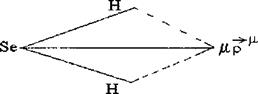
ќпределим тип молекулы H2Se. ƒл€ этого:
1.ќпредел€ем пол€рность св€зи H-Se-H на основе электроотрицательности Ќ и Se. “ак как Δ≈ = (ESe-EH) > 0, то св€зь пол€рна.
|
|
|
2. ќпредел€ем геометрическую конфигурацию молеклы H2Se. ƒелаем вывод: H2Se - молекула углова€, т.е. асимметрична.
3. —кладываем дипольные моменты св€зей μ св по правилу параллелограмма и находим дипольный момент молекулы μ м:
|
ƒипольный момент молекулы H2Se в данном случае больше 0, следовательно, молекула пол€рна.
ќпределим тип молекулы ¬е—12. ƒл€ этого:
1. ќпредел€ем пол€рность св€зи —1-¬е-—1 на основе электроотрицательности. “ак как Δ≈ = (≈а - ≈¬е) > 0, то св€зь пол€рна.
2. ќпредел€ем геометрическую конфигурацию ¬е—12. ƒелаем вывод: молекула линейна (симметрична).
3. —кладываем дипольные моменты св€зей μ св:
  CI Be —l
CI Be —l
|
но геометрическа€ сумма двух равных векторов, имеющих различное направление, равна 0, т.е. дипольный момент молекулы равен 0, а молекула непол€рна.
¬ методе молекул€рных орбиталей состо€ние молекулы описываетс€ как совокупность электронных ћќ. ѕри этом число ћќ равно сумме јќ; ћќ, возникающей от сложени€ јќ, соответствует более низка€ энерги€, чем исходным орбитал€м. “ака€ ћќ имеет повышенную электронную плотность в пространстве между €драми, способствует образованию химической св€зи и называетс€ св€зывающей. ћќ, образовав≠шейс€ от вычитани€ атомных орбиталей, соответствует более высока€ энерги€, чем јќ. Ёлектронна€ плотность в этом случае сконцентрирована за €драми атомов, а между ними равна 0. ѕодобные ћќ энергетически менее выгодны, чем исходные јќ, они привод€т к ослаблению химической св€зи и называютс€ разрыхл€ющими. Ёлектроны, занимающие св€зывающие и разрыхл€ющие орбитали, называют соответственно св€зывающими и разрыхл€ющими электронами. «аполнение молекул€рных орбиталей происходит при соблюдении принципа ѕаули и правила √унда по мере увеличени€ их энергии в такой последовательности:
σсв1s <σ*1s < σсв2s < σ*2 s < σсв2 р х < π св2 р у = π св2 p z < π *2 р у = π *2 р z < π *2 р х.
ѕри образовании молекул€рных орбиталей из атомных дл€ двухатомных гомо€дерных (одного и того же элемента) молекул элемента II периода число св€зывающих и разрыхл€ющих электронов зависит от их числа в атомах исходных элементов (рис. 1):
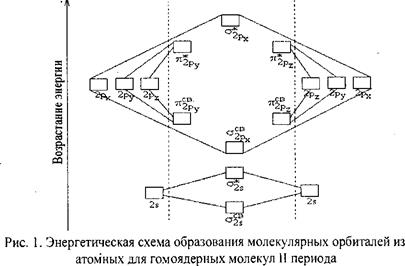
—ледует отметить, что при образовании молекул ¬2, —2 и N2 энерги€ св€зывающей сигма 2 р х -орбитали больше энергии св€зывающих π 2 р у - и π 2 p z -орбиталей, тогда как в молекулах кислорода и фтора, наоборот, энерги€ св€зывающих π 2 р у - и π 2 p z-орбиталей больше энергии св€зывающей σ2 р х -орбитали. Ёто нужно учитывать при изображении энергетических схем соответствующих молекул.
ѕор€док св€зи в молекуле определ€етс€ разностью чисел св€зывающих и разрыхл€ющих орбиталей, деленной на два. ѕор€док св€зи может быть равен 0 (молекула не существует), целому или дробному положительному числу.
ѕодобно электронным формулам, показывающим распределение электронов в атоме по атомным орбитал€м, в методе ћќ составл€ютс€ формулы молекул, отражающие их электронную конфигурацию. ѕо аналогии с атомными s-, p-, d-, f- орбитал€ми ћќ обозначаютс€ греческими буквами σ, π, δ, φ. “ак, электронна€ конфигураци€ молекул кислорода описываетс€ следующим образом:
O2 [KK (σs св2s)2 (σs разр2s)2 (πy св2 р у)2(πy св2 р у)2(σxсв2 р х)2 πy разр2 р1 у  πz разр 2 р z1].
πz разр 2 р z1].
Ѕуквами показано, что четыре 1s -электрона (два св€зывающих и два разрыхл€ющих) практически не оказывают вли€ни€ на химическую св€зь.
ќЌ“–ќЋ№Ќџ≈ ¬ќѕ–ќ—џ » «јƒјЌ»я
46. акую химическую св€зь называют ковалентной? „ем можно объ€снить направленность ковалентной св€зи? ак метод валентных св€зей (¬—) объ€сн€ет строение молекулы воды?
47. акую ковалентную св€зь называют пол€рной? „то служит количественной мерой пол€рности ковалентной св€зи? »сход€ из значений электроотрицательности атомов соответствующих элементов, определите, кака€ из св€зей: Ќ—1, JC1, BrF - наиболее пол€рна€.
48. акой способ образовани€ ковалентной св€зи называют донорно-акцепторным? акие химические св€зи имеютс€ в ионах: NH5+ и BF5+? ”кажите донор и акцептор.
49. ак метод валентных св€зей объ€сн€ет линейное строение молекулы ¬е—12 и тетраэдрическое —Ќ4?
50. акую ковалентную св€зь называют σ-св€зью и какую π-св€зью? –азберите на примере строени€ молекулы азота.
51. —колько неспаренных электронов имеет атом хлора в нормальном и возбужденном состо€ни€х? –аспределите эти электроны по квантовым €чейкам. „ему равна валентность хлора, обусловленна€ неспаренными электронами?
52. –аспределите электроны атома серы по квантовым €чейкам. —колько неспаренных электронов имеют ее атомы в нормальном и возбужденном состо€ни€х? „ему равна валентность серы, обусловленна€ неспаренными электронами?
|
|
|
53. акую химическую св€зь называют ионной? аков механизм ее образовани€? акие свойства ионной св€зи отличают ее от ковалентной? ѕриведите два примера типичных ионных соединений. Ќапишите уравнени€ превращени€ соответствующих ионов в нейтральные атомы.
54. акие силы молекул€рного взаимодействи€ называют ориентационными, индукционными и дисперсионными? огда возникают эти силы и какова их природа?
55. »зобразите схемы перекрывани€ электронных обаков при образовании св€зей в молекулах
| «адание | молекулы | «адание | молекулы |
| 55 а | HI, Na2 | 55 в | HBr, KH |
| 55 б | KH, Cl2 | 55 г | Br2, Li2 |
56. »зобразите валентные схемы молекул.
| «адание | молекулы | «адание | молекулы |
| 56 а | HNO3, H3PO4, CaSO4 | 56 г | Cu(HCO3)2, O3, ZnOHNO3 |
| 56 б | C6H6, AlOHSO4, H2CO3 | 56 д | O2, NaNO3, Ca(HCO3)2 |
| 56 в | Fe2(SO4)3, NO, Na2CO3 | 56 е | NO2, HNO2, H2SO3 |
57. Ќазовите тип гибридизации атомных орбиталей в атоме ј, имеющем указанную электронную конфигурацию валентного сло€, вход€щем в молекулу названного состава. »зобразите схему гибридизации. ѕриведите примеры соединений.
| «адание | —остав молекулы | электронна€ конфигураци€ атома ј |
| 57 а | ј¬2 | Еns 2 |
| 57 б | ј¬3 | Еns 2 np 1 |
| 57 в | ј¬4 | Еns 2 np 2 |
58. »зобразите перекрывание электронных обаков при образовании св€зей; укажите типы гибридизации атомных орбиталей в атомах ј.
| «адание | молекулы | атомы ј |
| 58 а | ¬е—12, ¬—13 | ¬е и ¬ |
| 58б | C2H2, SiH4 | C и Si |
| 58 в | C2H4, CaH2 | C и Ca |
| 58 г | C2H6, ј1—13 | C и ј1 |
59. –асположите молекулы в пор€дке возрастани€ пол€рности св€зей между атомами.
| «адание | молекулы | «адание | молекулы |
| 59 а | NH3, CH4, BH3, H2O | 59 в | CH4, SnH4, SiH4, GeH4 |
| 59 б | SiH4, H2S, PH3, HCl | 59 г | HI, HBr, HF, HCl |
60. »спользу€ метод молекул€рных орбиталей, запишите электронные формулы молекул. –ассчитайте кратность св€зей в молекулах.
| «адание | молекулы | «адание | молекулы |
| 60 а | O2, BeH2 | 60 г | CO, N2 |
| 60 б | O3, HF | 60 д | CO2, H2O |
| 60 в | B2, NH3 | 60 е | C2, H2S |
61. »сход€ из пол€ризационных представлений, расположите приведенные соединени€ в пор€дке уменьшени€: а) ионности св€зи, б) температуры плавлени€, в) термической устойчивости, г) гидролитической устойчивости, д) электролитической диссоциации.
| «адание | —оединени€ | «адание | —оединени€ |
| 61 а | LiCl, NaCl, KCl | 61в | Ca3N2, Ca3P2, Ca3As2, |
| 61 б | Ca—12, Sr—12, Ba—12 | 61 г | Ca3N2, Cr3N2, Ba3N2 |
62. акие валентности и почему может про€вл€ть элемент? ќтвет подтвердите записью распределени€ электронов валентных энергетических уровней и подуровней.
| «адание | элементы | «адание | элементы |
| 62 а | Li, Sc, I | 62 е | Cs, La, Br |
| 62 б | Ca, Si, S | 62 ж | Mo, Sn, Ba |
| 62 в | Cd, Ge, Cl | 62 з | Cr, Se, In |
| 62 г | Ga, W, F | 62 и | Sb, Zn, Be |
| 62 д | Pb, S, Al | 62 к | Hf, Re, Mg |
63. ак измен€етс€ характер св€зи в р€ду соединений?
| «адание | ¬ещества |
| 63 а | NaCl ― Mg—12―ј1—13― Si—14 |
| 63 б | ¬—13―ј1—13― Ga—13 ―In—13 |
| 63 в | HF ― HCl ― HBr― HI |
| 63 г | CH4― NH3 ―H2O ―HF |
| 63 д | H2O― H2S ―H2Se ―H2Te |
| 63 е | GeH4― AsH3 ― H2Se ― HBr |
| 63 ж | ¬е—12― Mg—12― Ca—12 ―Sr—12 |
64. Ќарисуйте энергетическую схему образовани€ молекулы кислорода по методу молекул€рных орбиталей (ћќ). ак метод ћќ объ€сн€ет парамагнитные свойства молекулы кислорода?
65. Ќарисуйте энергетическую схему образовани€ молекулы фтора по методу ћќ. —колько электронов находитс€ на св€зывающих и разрыхл€ющих орбитал€х? „ему равен пор€док св€зи в этой молекуле?
66. Ќарисуйте энергетическую схему образовани€ молекулы азота по методу ћќ. —колько электронов находитс€ на св€зывающих и разрыхл€ющих орбитал€х? „ему равен пор€док св€зи в этой молекуле?
67. Ќарисуйте энергетическую схему образовани€ молекулы —ќ по методу ћќ. —колько электронов находитс€ на св€зывающих и разрыхл€ющих орбитал€х? „ему равен пор€док св€зи в этой молекуле?
68. ќпределите тип св€зи в молекулах: NaF, —а—12, AIF3, —12, —аќ.
69. ќпределите тип молекул –—13 и PCI5.
70. ѕокажите, как вли€ет тип св€зи на окраску соединений, на примере следующих соединений: Ni(OH)2, NiO, NiS.






