1. ѕри диссоциации сильных электролитов ставитс€ знак Ђ=ї (равенства) или ЂЃї (одна стрелочка). Ётот знак обозначает полную диссоциацию.
2. ѕри диссоциации слабых (и средней силы) электролитов Ц знак ЂÛї (обратимости). Ётот знак обозначает частичную диссоциацию.
ѕравила написани€ ионных уравнений
¬ ионных уравнени€х на ионы расписываютс€:
1. сильные кислоты;
2. щелочи;
3. растворимые соли.
| —ильные электролиты | —лабые электролиты |
 ќсновани€:
—а(OH)2 = Ca2+ + 2OH-
ислоты:
HCl = H++Cl-
—оли:
Na2CO3=2Na++CO32-
KH2PO4=K++H2PO4-
анион слабой
кислоты
H2PO4-ÛH++HPO42-
HPO42-ÛH++PO43-
AlOHCl2ÛAlOHCl2 = AlOH2++2Cl-
осадок растворима€ катион слабого
часть основани€
AlOH2+ÛAl3++OH- ќсновани€:
—а(OH)2 = Ca2+ + 2OH-
ислоты:
HCl = H++Cl-
—оли:
Na2CO3=2Na++CO32-
KH2PO4=K++H2PO4-
анион слабой
кислоты
H2PO4-ÛH++HPO42-
HPO42-ÛH++PO43-
AlOHCl2ÛAlOHCl2 = AlOH2++2Cl-
осадок растворима€ катион слабого
часть основани€
AlOH2+ÛAl3++OH-
| ќсновани€:
Fe(OH)2 ÛFeOH+ + OH- 1 ступень
FeOH+ÛFe2++OH- 2 ступень
ислоты:
H2CO3ÛH++HCO3- I ступень
HCO3-ÛH++CO32- II ступень
јмфотерные гидроксиды:
1.  Zn(OH)2ÛZnOH++OH- по осн.
ZnOH+ÛZn2++OH- типу
2. H2ZnO2ÛH++HZnO2- по кисл.
HZnO2-ÛH++ZnO22- типу
¬ода:
H2OÛH++OH- Zn(OH)2ÛZnOH++OH- по осн.
ZnOH+ÛZn2++OH- типу
2. H2ZnO2ÛH++HZnO2- по кисл.
HZnO2-ÛH++ZnO22- типу
¬ода:
H2OÛH++OH-
|
ќЌ÷≈Ќ“–ј÷»я –ј—“¬ќ–ќ¬
¬ажнейшей характеристикой количественного содержани€ компонентов в системе €вл€етс€ концентраци€ растворов.
онцентрацией растворов называют определенное массовое (или объемное) содержание растворенного вещества в определенном массовом (или объемном) количестве растворител€ или раствора.
—уществуют несколько методов выражени€ концентрации растворов. –ассмотрим самые распространенные из них:
| ћетоды выражени€ концентрации растворов | ќбозначение и размерность примен€емых величин |
ћассова€ дол€ растворенного вещества Ц это отношение массы растворенного вещества к общей массе раствора:
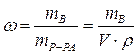
| 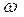 - массова€ дол€ растворенного вещества, безразмерна€ величина - массова€ дол€ растворенного вещества, безразмерна€ величина
|
| m¬ - масса растворенного вещества, г | |
| m–-–ј - масса раствора, г | |
| V - объем раствора, мл | |
| ρ -плотность раствора, г/мл | |
ѕроцентна€ концентраци€:
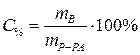
| —% - процентна€ концентраци€, % |
| m ¬ - масса растворенного вещества, г | |
| m–-–ј - масса раствора, г | |
ћол€рна€ концентраци€, или мол€рность, Ц число молей растворенного вещества в 1 дм3
(1 литр) раствора:
 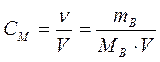
| —ћ - мол€рна€ концентраци€, или мол€рность; иногда обозначают ћ |
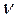 - количество (число моль) растворенного вещества, моль - количество (число моль) растворенного вещества, моль
| |
| V - объем раствора, дм3 (л) | |
| m¬ - масса растворенного вещества, г | |
| ћ¬ - мол€рна€ масса растворенного вещества, г/моль |
ѕри решении задач необходимо обращать особое внимание на размерность примен€емых величин и об€зательно приводить их в соответствие друг другу. ѕоэтому при подстановке численных значений в формулы надо указывать размерность величин. –ешение задач оформл€ть об€зательно в соответствии с приведенными примерами (см. стр. 67 Ц 71).
√»ƒ–ќЋ»« —ќЋ≈…
√идролиз Ц это процесс взаимодействи€ соли с водой, в результате которого происходит смещение ионного равновеси€ воды и изменение значени€ рЌ раствора. ¬озможность и характер протекани€ гидролиза определ€етс€ природой основани€ и кислоты, образующих соль.
|
|
|
—оли, образованные сильной кислотой и сильным основанием, гидролизу не подвергаютс€. »х водные растворы нейтральны.
KNO3+H2O ¹; pH=7.
¬ гидролизе участвуют ионы слабого основани€ и/или слабой кислоты.
Ј √идролиз солей, образованных сильной кислотой и слабым основанием, например, NH4Cl:
NH4Cl = NH4++Cl -
√идролизу подвергаетс€ катион слабого основани€ NH4+:
NH4+ + HOH Û NH4OH + H+ (ионы H+ в избытке).
ћолекул€рное уравнение:
NH4Cl + H2O Û NH4OH + HCl Ц кисла€ среда (рЌ < 7).
Ј √идролиз солей, образованных слабой кислотой и сильным основанием, например: Na2SO3:
Na2SO3 = 2Na+ + SO32 -
√идролизу подвергаетс€ анион слабой кислоты SO32 -:
1-€ ступень: SO32 - +HOH Û HSO3 - +OH - (ионы OH - в избытке).
ћолекул€рное уравнение 1-й ступени:
Na2SO3 + H2O Û NaHSO3 + NaOH Ц щелочна€ среда (рЌ > 7).
ѕри обычных услови€х гидролиз протекает главным образом по первой ступени.
Ј √идролиз солей образованных слабым основанием и слабой кислотой ((NH4)2CO3, Al2S3 и т.д.), гидролизуютс€ как по катиону, так и по аниону. ¬ этом случае гидролиз соли протекает до конца: в уравнении происходит замена знака УÛФ на У=Ф, а рЌ среды определ€етс€ силой кислоты и основани€:
NH4ClO = NH4+ + ClO -
NH4+ + ClO - + HOH = NH4OH + HClO
NH4ClO + H2O = NH4OH + HClO
K (HClO) = 3×10-8 < K (NH4OH) = 1,8×10-5, так как основание €вл€етс€ более сильным электролитом, чем кислота, среда слабощелочна€ рЌ > 7.
Ј —овместный гидролиз двух солей ≈сли в растворе одновременно присутствуют ионы, св€зывающие Ќ+ и ќЌ-, то в соответствии с принципом Ће Ўателье равновесие смещаетс€ в сторону усилени€ процесса гидролиза. √идролиз может протекать необратимо с образованием малорастворимого и газообразного веществ.
¬ качестве примера можно рассмотреть совместный гидролиз следующих солей:
Al 2(SO4)3 + K2 S + H2O Ѓ
2Al3+ + 3S2 - + 6H2O = 2Al(OH)3 + 3H2S
Al2(SO4)3 + 3K2S + 6H2O = 2Al(OH)3¯ + 3H2S≠ + 3 2SO4
pHї 7
7.ќ »—Ћ»“≈Ћ№Ќќ-¬ќ——“јЌќ¬»“≈Ћ№Ќџ≈
–≈ј ÷»» (ќ¬–)
ќкислительно-восстановительными называютс€ реакции, в результате которых измен€ютс€ степени окислени€ элементов, вход€щих в состав реагирующих веществ. Ќапример:
+4 -1 +2 0
MnO2 + 4HCl = MnCl2 + Cl2 + 2H2O






