= Ц0,2145 B.
ѕри составлении гальванического элемента более отрицательным электродом будет система Zn | Zn2+, более положи-тельным Ц Pb | Pb2+. —хема гальванического элемента примет следующий вид
(Ц) Zn | ZnSO4 (0,01 M) || Pb(NO3)2 (0,001 M) | Pb (+).
ѕри наличии внешней цепи на электродах протекают следующие реакции
Zn0 = Zn+2 + 2 
| окисление | восста- новитель | јнод | |
Pb2+ + 2  = Pb0 = Pb0
| восста- новление | окисли- тель | атод |

 –еакци€ в элементе в целом Zn0 + Pb2+ = Zn2+ + Pb0.
–еакци€ в элементе в целом Zn0 + Pb2+ = Zn2+ + Pb0.
 |
2 
Ёлектродвижуща€ сила этого элемента может быть рассчитана как по ранее определенным электродным потенциалам, так и непосредственно.
1 вариант E 298 = φкатода Ц φанода = φ  Ц φ
Ц φ  =
=
= Ц 0,2145 Ц (Ц 0,822) = 0,6075 ¬.
2 вариант
E 298 = φ  Ц φ
Ц φ  = φ
= φ  +
+  lg
lg  Ц φ
Ц φ  Ц
Ц
≠Ц  lg
lg  = (φ
= (φ  Цφ
Цφ  ) +
) +  lg
lg  = (Ц 0,126 Ц
= (Ц 0,126 Ц
Ц (Ц0,763)) +  lg
lg  = 0,6075 B.
= 0,6075 B.
≈сли бы в условии были заданы металлы в разных степен€х окислени€, то следовало бы использовать величины количеств электронов z 1, z 2.
ѕример 6.4. ѕри электролизе раствора данной соли металла током I, A, масса катода возросла на m грамм. ”читыва€, что выход по току металла B i, %, рассчитайте, какое количество электричества и в течение какого времени пропущено. —оставьте схему электролиза.
ƒано: CoSO4; I = 1,25 A; m = 1,0883 г; B i = 72 %.
– е ш е н и е
—оставл€ем схему электролиза с нерастворимым анодом. Ёлектрохимическа€ система имеет следующий вид
(Ц) Fe | CoSO4, H2O | Ti (+).
¬ качестве покрываемого металла выбрано железо; нерастворимого анода Ц титан.
¬ растворе присутствуют следующие ионы и молекулы
CoSO4  Co2+ + SO
Co2+ + SO 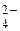 (электролитическа€ диссоциаци€);
(электролитическа€ диссоциаци€);
CoSO4 + 2H2O = Co(OH)2 + H2SO4 (гидролиз);
—о2+ + 2Ќ2ќ = —о(ќЌ)2  + 2Ќ+, рЌ < 7, среда кисла€.
+ 2Ќ+, рЌ < 7, среда кисла€.
—ледовательно, при составлении схемы электролиза надо учитывать ионы —о2+, SO 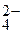 , Ќ+, молекулы —оSO4, H2O, Co(OH)2.
, Ќ+, молекулы —оSO4, H2O, Co(OH)2.
–еакции на электродах
ј: 2Ќ2ќ = ќ2 + 4Ќ+ + 4  ;
;
: —о2+ + 2  = —о0; 2Ќ+ + 2
= —о0; 2Ќ+ + 2  = Ќ2.
= Ќ2.
»з-за выделени€ водорода на катоде совместно с восстановлением ионов —о(II) выход по току металла меньше 100 %.
—хема электролиза водного раствора CoSO4 с нерастворимым анодом:
CoSO4
атод јнод


 |
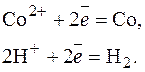
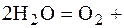 + 4
+ 4  .+ 4
.+ 4 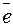 .
.
ƒалее составл€ем схему электролиза с растворимым анодом. Ёлектрохимическа€ система:
(Ц) Fe | CoSO4, H2O | Co (+).
–еакции на электродах:
ј:2—о0 = 2—о2+ + 4  .
.
:—о2+ + 2  = —о0,
= —о0,
2Ќ+ + 2  = Ќ2.
= Ќ2.
—хема электролиза водного раствора —оSO4 c растворимым анодом:
CoSO4
атод јнод


 —о2+ + 2
—о2+ + 2  = —о0,
= —о0,
2Ќ+ + 2  = Ќ2. 2—о0 = 2
= Ќ2. 2—о0 = 2 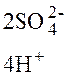 + 4
+ 4  .
.
оличество электричества по закону ‘араде€ составит

ќно пропущено в течение времени t:
 ч ≈ 1,1 ч.
ч ≈ 1,1 ч.
ѕример 6.5. ”становите, в какой последовательности веро€тно восстановление на катоде при электролизе данных ионов, пользу€сь значени€ми стандартных электродных потенциалов и перенапр€жений. ќбъ€сните, когда возможно совместное восстановление металла и водорода на катоде. „ем отличаетс€ последовательность электрохимических реакций на аноде от аналогичной последовательности на катоде?
|
|
|
¬ растворе присутствуют следующие ионы: Ag+, Cr3+, Zn2+, Sn2+, H+.
– е ш е н и е
Ќаходим значени€ стандартных электродных потенциалов соответствующих электрохимических систем и перенапр€жений.
| »он | Ag+ | Cr3+ | Zn2+ | Sn2+ | H+ |
| Ёлектрохимическа€ система | Ag+/Ag0 | Cr3+/Cr0 | Zn2+/Zn0 | Sn2+/Sn0 | 2H+/H 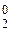
|
| φ0, ¬ | 0,799 | -0,744 | -0,763 | -0,136 | |
| ηк, ¬ | 0,18 | 0,41 | 0,03 | 0,01 | 0,18 (на Ag) |
–ассчитываем значени€ катодных потенциалов в реальных услови€х электролиза, под током
 .
.
| Ёлектрохимическа€ система | Ag+/Ag0 | Cr3+/Cr0 | Zn2+/Zn0 | Sn2+/Sn0 | 2Ќ+/Ќ02 |
| φ iк, ¬ | 0,619 | -1,154 | -0,766 | -0,146 | -0,18 (на Ag) |
ѕоследовательность разр€да ионов на катоде устанавливаем исход€ из того, что на катоде восстанавливаютс€ в первую очередь ионы с наиболее положительным электродным потенциалом соответствующей электрохимической системы.
| φ iк, ¬ | 0,619 | -0,146 | -0,18 | -0,766 | -1,154 |
| Ёлектрохимическа€ система | Ag+/Ag0 | Sn2+/Sn0 | 2H+/H 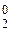
| Zn2+/Zn0 | Cr3+/Cr0 |
| »он | Ag+ | Sn2+ | H+ | Zn2+ | Cr3+ |
ќтсюда видно, что на катоде будут восстанавливатьс€ в первую очередь ионы Ag+. ѕри повышенных плотност€х тока, в случае, если достигаетс€ предельный ток по ионам Ag+, может начатьс€ восстановление ионов Sn2+, что используют, в частности, при электролитическом рафинировании металлов Ц можно отдельно выделить серебро, затем олово. ¬осстановлени€ водорода в обычных услови€х совместно с металлом Ц серебром не происходит, оно возможно лишь при плотност€х тока, намного превышающих предельную по ионам Ag+.
јнодное поведение рассматриваемых ионов пр€мо противоположно катодному: на аноде в первую очередь будут идти процессы окислени€ в электрохимических системах с наиболее отрицательным электродным потенциалом.
«адачи
6.1. ƒл€ данного окислительно-восстановительного процесса:
а) составьте реакции окислени€ и восстановлени€; б) укажите окислитель и восстановитель; в) составьте сокращенное ионное и полное молекул€рное уравнени€ ионно-электронным методом; г) покажите переход электронов; д) рассчитайте 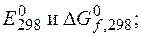
е) укажите возможное направление протекани€ процесса;
ж) составьте гальванический элемент на основе данной реакции.
“ а б л и ц а 6.1
| ¬ариант | –еакци€ |
H2O2 + KMnO4 + H2SO4  MnSO4 + K2SO4 + O2 + H2O MnSO4 + K2SO4 + O2 + H2O
| |
NaNO2 + NaI + H2SO4  NO + I2 + Na2SO4 + H2O NO + I2 + Na2SO4 + H2O
| |
NaI + H2SO4 + MnO2  NaHSO4 + H2O + I2 + MnSO4 NaHSO4 + H2O + I2 + MnSO4
| |
MnSO4 + S + K2SO4 + H2O  KMnO4 + H2S + H2SO4 KMnO4 + H2S + H2SO4
| |
K2SO4+ Cr2(SO4)3+ Fe2(SO4)3+ H2O  K2Cr2O7+H2SO4+FeSO4 K2Cr2O7+H2SO4+FeSO4
| |
KCl + Fe2(SO4)3 + H2O  KClO3 + FeSO4 + H2SO4 KClO3 + FeSO4 + H2SO4
| |
FeCl3 + MnCl2 + KCl + H2O  FeCl2 + KMnO4 + HCl FeCl2 + KMnO4 + HCl
| |
K2SO4 + Cr2(SO4)3 + H2O  K2Cr2O7 + H2SO4 + SO2 K2Cr2O7 + H2SO4 + SO2
| |
K2Cr2O7 + H2S + H2SO4  Cr2(SO4)3 + S + K2SO4 + H2O Cr2(SO4)3 + S + K2SO4 + H2O
| |
K2SO4 + I2 + NO2 + H2O  KI + HNO3 + H2SO4 KI + HNO3 + H2SO4
| |
Fe2(SO4)3 + NO + H2O  FeSO4 + HNO3 + H2SO4 FeSO4 + HNO3 + H2SO4
| |
MnSO4+Br2+Na2SO4+K2SO4 + H2O  KMnO4 + NaBr + H2SO4 KMnO4 + NaBr + H2SO4
| |
| ќкончание табл. 6.1 | |
HMnO4 + Pb(NO3)2 + H2O  PbO2 + Mn(NO3)2 + HNO3 PbO2 + Mn(NO3)2 + HNO3
| |
KCl + NaCl + H2O + CO2  KClO3 + Na2C2O4 + HCl KClO3 + Na2C2O4 + HCl
| |
Na2C2O4+KMnO4+H2SO4  MnSO4+K2SO4+Na2SO3+H2O+CO2 MnSO4+K2SO4+Na2SO3+H2O+CO2
| |
| 1Т | K2MnO4 + O2 + H2O  KMnO4 + H2O2 + KOH KMnO4 + H2O2 + KOH
|
| 2Т | MnCl2 + Cl2 + KCl + H2O  KMnO4 + HCl KMnO4 + HCl
|
| 3Т | HNO2 + KMnO4 + H2SO4  HNO3 + MnSO4 + K2SO4 + H2O HNO3 + MnSO4 + K2SO4 + H2O
|
| 4Т | PbSO4 + ZnSO4 + H2O  PbO2 + H2SO4 + Zn PbO2 + H2SO4 + Zn
|
| 5Т | Al2(SO4)3+Cr2(SO4)3+K2SO4+H2O  Al+K2Cr2O7+H2SO4 Al+K2Cr2O7+H2SO4
|
| 6Т | K2CrO4 + KBr +H2O + KCl  CrCl3 + Br2 + KOH CrCl3 + Br2 + KOH
|
| 7Т | Na2SO4 + I2 + K2SO4 + H2O  Na2SO3 + KIO3 + H2SO4 Na2SO3 + KIO3 + H2SO4
|
| 8Т | Na2SO4 + Ag + NaNO3 + H2O  Na2SO3 + AgNO3 + NaOH Na2SO3 + AgNO3 + NaOH
|
| 9Т | KI + KNO2 + H2SO4  I2 + NO + K2SO4 + H2O I2 + NO + K2SO4 + H2O
|
| 10Т | CO2 + MnSO4 + H2O  H2C2O4 + MnO2 + H2SO4 H2C2O4 + MnO2 + H2SO4
|
| 11Т | H2O2 + PbO2 + CH3COOH  O2 + Pb(CH3COO)2 + H2O O2 + Pb(CH3COO)2 + H2O
|
| 12Т | Ti(SO4)2+MnSO4+ K2SO4 + H2O  Ti2(SO4)3 + KMnO4 + H2SO4 Ti2(SO4)3 + KMnO4 + H2SO4
|
| 13Т | KBr + MnO2 + KCl + H2O  KBrO + MnCl2 + KOH KBrO + MnCl2 + KOH
|
| 14Т | MnSO4 + NaHSO4 + H2O + Cl2  MnO2 + NaCl + H2SO4 MnO2 + NaCl + H2SO4
|
| 15Т | K2Cr2O7 + KI + H2SO4  Cr2(SO4)3 + I2 + K2SO4 + H2O Cr2(SO4)3 + I2 + K2SO4 + H2O
|
|
|
|
6.2. –асчет электродвижущей силы гальванического элемента в стандартных услови€х по известной реакции в элементе.
ƒана реакци€, протекающа€ в гальваническом элементе. –ассчитайте  по данным
по данным 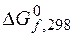 , составьте схему элемента, реакции на электродах.
, составьте схему элемента, реакции на электродах.
“ а б л и ц а 6.2
| ¬ариант | –еакции |
| PbO2 (к) + 2H2SO4 (ж) + Zn (к) = PbSO4 (к) + ZnSO4 (к) + 2H2O (ж) | |
| PbO2 (к) + 2H2SO4 (ж) + Cd (к) = PbSO4 (к) + CdSO4 (к) + 2H2O (ж) | |
| Cu2Cl2 (к) + Mg (к) = 2Cu (к) + MgCl2 (к) | |
| 2Ag (к) + MgCl2 (к) = 2AgCl (к) + Mg (к) | |
| PbO2 (к) + 2H2SO4 (ж) + Pb (к) = 2PbSO4 (к) + 2H2O (ж) | |
| 2Pb (к) + O2 (г) + 2H2O (ж) = 2Pb(OH)2 (к) | |
| O2 (г) + 2Sn (к) + 2H2O (ж) = 2Sn(OH)2 (к) | |
| O2 (г) + 2Mn (к) + 2H2O (ж) = 2Mn(OH)2 (к) | |
| Ag2O (к) + Cd (к) = 2Ag (к) + CdO (т) | |
| O2 (г) + 2Fe (т) + 2H2O (ж) = 2Fe(OH)2 (к) | |
| HgO (красн.к) + Zn (к) = ZnO (к) + Hg (к) | |
| 2CuCl (к) + Mg (к) = MgCl2 (к) + 2Cu (к) | |
| Th (к) + O2 (г) + 2H2O (ж) = Th(OH)4 (к) | |
| ќкончание табл. 6.2 | |
| 2Ga (к) + 3/2 O2 (г) + 3H2O (ж) = 2Ga(OH)3 (к) | |
| 1Т | O2 (г) + 2Mg (к) + 2H2O (ж) = 2Mg(OH)2 (т) |
| 2Т | O2 (г) + 2Ni (к) + 2H2O (ж) = 2Ni(OH)2 (т) |
| 3Т | 3/2 O2 (г) + 2Fe (к) + 3H2O (ж) = 2Fe(OH)3 (к) |
| 4Т | Ag2O (к) + Zn (к) = 2Ag (к) + ZnO (к) |
| 5Т | CuCl2 (к) = CuCl (к) + ½ Cl2 (г) |
| 6Т | Cl2 (г) + Zn (к) = ZnCl2 (к) |
| 7Т | 2H2O (ж) + 2Li (к) = 2LiOH (к) + H2 (г) |
| 8Т | O2 (г) + 4Li (т) + 2CO2 (г) = 2Li2CO3 (т) |
| 9Т | Mg (к) + 2H2O (ж) = Mg(OH)2 (к) + H2 (г) |
| 10Т | O2 (г) + 2Zn (к) = 2ZnO (к) |
| 11Т | 3/2 O2 (г) + 2Au (к) + 3H2O (ж) = 2Au(OH)3 (к) |
| 12Т | 2Cd (к) + O2 (г) + 2H2O (ж) = 2Cd(OH)2 (к) |
| 13Т | 5Zn(к)+2KIO3(к)+6H2SO4(ж)=5ZnSO4(к)+I2(к)+K2SO4(к)+6H2O (ж) |
| 14Т | PbSO4 (к) + H2O (ж) = Pb (к) + ½ O2 (г) + H2SO4 (ж) |
6.3. —оставьте схему гальванического элемента, образованного двум€ данными металлами, погруженными в растворы солей с известной активностью ионов; рассчитайте Ёƒ— этого элемента и 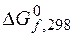 .
.
“ а б л и ц а 6.3
| ¬ариант | ћеталлы | —оли | јктивности |
| Cr, Al | CrCl3, AlI3 | 
| |
| Mn, Co | MnCl2, CoCl2 | 
| |
| Fe, Cd | FeCl2, CdBr2 | 
| |
| Cu, Ag | CuSO4, AgNO3 | 
| |
| Sn, Al | SnBr2, AlCl3 | 
| |
| Au, Fe | AuCl3, FeCl2 | 
| |
| Cd, In | CdBr2, In(NO3)3 | 
| |
| Sb, Co | SbF3, CoCl2 | 
| |
| Cd, Ni | CdCl2, NiSO4 | 
| |
| Re, Co | ReCl3, Co(NO3)2 | 
| |
| Pd, Cd | PdSO4, CdI2 | 
| |
| Sb, Co | SbCl3, CoSO4 | 
| |
| ќкончание табл. 6.3 | |||
| Cr, Sn | CrBr3, SnCl4 | 
| |
| Nd, Mn | NdCl3, MnSO4 | 
| |
| Sn, Cd | SnBr2, CdSO4 | 
| |
| 1Т | Ti, Cr | TiCl3, CrCl2 | 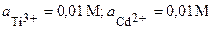
|
| 2Т | Mn, In | MnSO4, InBr3 | 
|
| 3Т | Pd, Cr | PdCl2, CrCl3 | 
|
| 4Т | Ni, Nd | Ni(NO3)2, NdCl3 | 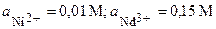
|
| 5Т | Re, Cr | ReCl3, Cr(NO3)3 | 
|
| 6Т | Cu, Cd | CuCl2, CdBr2 | 
|
| 7Т | In, Cr | In(NO3)3, Cr2(SO4)2 |  
|
| 8Т | Mn, Cu | MnCl2, Cu(NO3)2 | 
|
| 9Т | Ni, Cu | NiSO4, CuBr2 | 
|
| 10Т | Ti, Fe | TiCl3, FeBr2 | 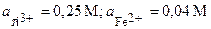
|
| 11Т | Cr, Fe | CrCl3, FeCl3 | 
|
| 12Т | Mn, In | Mn(NO3)2, InCl3 | 
|
| 13Т | Pd, Fe | PdCl2, Fe(NO3)3 | 
|
| 14Т | Ni, In | NiCl2, InBr3 | 
|
| 15Т | Re, Fe | ReCl3, Fe2(SO4)3 | 
|
|
|
|






