7.36. ѕри получении золота используетс€ реакци€ его вытесне-ни€ из цианистых электролитов с использованием цинка:
Zn + 2K[Au(CN)2] = 2Au + K2[Zn(CN)4].
–ассчитайте, кака€ масса золота может быть получена, если на протекание реакции израсходовано 120 г дицианоаурата кали€.
7.37. ѕирометаллургический метод получени€ цинка из цинко-вых руд включает удаление серы по реакции
2ZnS + 3O2 = 2ZnO + 2SO2↑
с последующим восстановлением цинка:
ZnO + C = Zn + CO↑.
–ассчитайте, кака€ масса цинка может быть получена, если на удаление серы затрачено 45 л кислорода в расчете на нормальные услови€.
7.38. арбид хрома Cr3C2 образуетс€ или при непосредственном взаимодействии хрома с углеродом, или оксида Cr2O3 в атмосфере —ќ. Ќапишите уравнени€ химических реакций и рассчитайте, кака€ масса карбида может быть получена из 10 кг хрома в том и другом случа€х.
7.39. –туть Ц малоактивный элемент и ее получают пр€мым окислением киновари HgS кислородом при 600Ц700 о—.
Ќапишите уравнение химической реакции и рассчитайте массу ртути, котора€ может быть получена из 150 кг киновари, если потери составл€ют 12 %.
7.40. јлюминий используетс€ дл€ получени€ кремни€ путем восстановлени€ кварцевого песка SiO2.
Ќапишите уравнение химической реакции получени€ кремни€ методом металлотермии и рассчитайте, кака€ масса алюмини€ потребуетс€ дл€ получени€ 50 кг кремни€.
7.41. —урьма активно взаимодействует с хлором. –ассчитайте, кака€ масса пентахлорида сурьмы может быть получена из 30 кг металла и какой объем хлора расходуетс€ на проведение реакций? (–асчет вести дл€ н.у.).
7.42. —оль рутени€ Ц рутенат кали€ получена по реакции
5Ru + 4KOH + 6KNO3 = 5K2RuO4 + 3N2 + 2H2O.
–ассчитайте, сколько литров раствора ќЌ плотностью 1,18 г/см3 и массовым содержанием ќЌ ω = 20,5 % потребуетс€ дл€ получени€ 180 г рутената кали€?
7.43. ѕри восстановлении хлорида цези€ карбидом кальци€ получаетс€ цезий, загр€зненный углем.
Ќапишите уравнение химической реакции получени€ цези€ и рассчитайте степень загр€знени€ цези€ углем (ω, % масс.), если в реакцию вступило 40 г —а—2.
7.44. ћагний получают из оксида путем восстановлени€ кремнием по реакции в вакууме

–ассчитайте массу оксидов кальци€ и магни€, а также кремни€, котора€ необходима дл€ получени€ 1 кг магни€.
7.45. ƒл€ получени€ металлического олова из силикатов смесь силиката олова, оксид кальци€ и уголь прокаливают при 1200 о—
SnSiO3 + CaO + C = Sn + CaSiO3 + CO.
–ассчитайте, кака€ масса шлака в виде силиката кальци€ всплывает на поверхность расплавленного металла при получении 2 т олова?
’»ћ»я Ќ≈ћ≈“јЋЋќ¬
–ешение типовых задач
ѕример 8.1. ƒл€ йода (53I) определите валентные электроны в нормальном и возбужденном состо€нии. ”кажите возможные степени окислени€. ќпишите физические и химические свойства.
– е ш е н и е
Ќапишем распределение электронов дл€ 53I.
1s22s22p63s23p64s23d104p65s24d105p5.
ќпределим валентные электроны йода
Ќормальное состо€ние
|
|
|







степени окислени€: Ц1; 0; +1;
¬озбужденное состо€ние

степени окислени€: Ц1; 0; +1; +2; +3; +4; +5; +6; +7.
…од при обычных услови€х представл€ет собой чернофио-летовые кристаллы с металлическим блеском. “ пл. = 113,6 о—. “ кип. = = 184,3 о—. ѕри обычных услови€х растворимость йода составл€ет 0,02 г на 100 г воды. ¬ органических растворител€х (бензин, спирт и др.) йод раствор€етс€ значительно лучше.
ѕолучают йод окислением йодидов хлором, извлекают из селитры и морских водорослей. ћировое производство йода составл€ет 10 тыс. т в год.
…од находит применение в производстве чистых металлов и полупроводниковых материалов; как антисептик; дл€ йодировани€ поваренной соли.
…од токсичен, его ѕƒ составл€ет 1 мг/м3.
…од достаточно сильный окислитель, о чем свидетельствует потенциал реакции I2 + 2 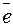 = 2 I
= 2 I  , равный 0,54 ¬.
, равный 0,54 ¬.
ѕример 8.2. Ќапишите уравнени€ реакции взаимодействи€ азота с простыми веществами (Mg и Cl2). акие химические свойства про€вл€ет азот в этих реакци€х? „ем можно объ€снить его способность взаимодействовать с окислител€ми и восстановител€ми? Ќазовите продукты реакций.
– е ш е н и е
ѕри высоких температурах азот окисл€ет многие металлы, образу€ нитриды, например
3Mg + N2 = Mg3N2.
Mg3N2 Ц нитрид магни€ Ц кристаллическое вещество, химически довольно активное, легко разлагаетс€ водой, образу€ щелочь и аммиак.
ѕри взаимодействии с галогенами азот образует нитриды галогенов. Ќитриды галогенов Ц эндотермические соединени€, например
N2 + 3Cl2 = 2Cl3N, ΔЌ  = 229 кƒж/моль.
= 229 кƒж/моль.
Ќитрид хлора Ц кислотное соединение, легко разлагающеес€ со взрывом по реакции
3H2O + Cl3N = NH3 + 3HClO.
ѕри образовании нитридов элементов азот ведет себ€ как окислитель. јзот €вл€етс€ восстановителем лишь при взаимодействии со фтором и кислородом, образу€ трифторид азота и различные оксиды азота соответственно.
ѕр€мым взаимодействием простых веществ образуетс€ лишь NO Ц оксид азота (II)
½ N2 (г) + ½ O2 (г) = NO (г).
NO Ц бесцветный газ, в природе образуетс€ при грозовых разр€дах по цепной реакции
ќ2 + h υ → OХ + ХOХ
+ Х
N2 → NO + ХNХ
+ O2 → NO + ХOХ и т.д.
NF3 Ц трифторид азота Ц бесцветный газ, получаетс€ при окислении аммиака фтором. NF3 химически устойчив, вступает в реакции при температуре выше 100 о—, кислотное соединение
NF3 + 3H2O (пар) = N2O3 + 6HF.
ѕример 8.3. ќпишите способы получени€ хлора. Ќапишите уравнени€ реакций и услови€ их протекани€, принципиальную схему получени€.
– е ш е н и е
¬ лабораторных услови€х хлор получают из сол€ной кислоты, действу€ сильными окислител€ми. „аще всего примен€ют оксид марганца (IV) MnO2 и перманганат кали€ KMnO4. ¬ этих соединени€х окислителем €вл€етс€ марганец со степен€ми окислени€ +4 и +7. ¬ыделение хлора сразу можно обнаружить по характерному удушливому запаху
4HCl (конц.) + MnO2 → MnCl2 + Cl2↑ + 2H2O.
¬ промышленности хлор получают электролизом расплав-ленного хлорида натри€
|
Ќа катоде: Ќа аноде:
Na+ + 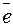 → Na0 2Cl
→ Na0 2Cl  Ц 2
Ц 2 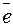 → Cl2↑
→ Cl2↑
ѕример 8.4. ќпишите окислительно-восстановительные свойст-ва неметаллов. ”равн€йте окислительно-восстановительные реакции ионно-электронным методом.
ƒана реакци€
|
|
|
I2 + Cl2 + H2O = HCl + HIO3.
– е ш е н и е
1. ѕроставим степени окислени€ всех элементов, чтобы определить окислитель и восстановитель

2. ” хлора степень окислени€ понижаетс€, следовательно он выполн€ет роль окислител€ (принимает электроны), а у йода степень окислени€ повышаетс€, он играет роль восстановител€ (отдает электроны). ¬ода играет роль среды.
3. «апишем полуреакции окислени€ и восстановлени€, найдем наименьшее общее кратное и проставим соответствующие коэффициенты


 Ц 10
Ц 10 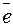 + 6H2O → 2(IO3)Ц + 12H+ 1
+ 6H2O → 2(IO3)Ц + 12H+ 1
 + 2
+ 2 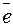 → 2ClЦ 5
→ 2ClЦ 5
4. ислород уравниваем в форме Ќ2ќ, водород Ц в форме Ќ+.
5. —уммируем левые и правые части полуреакций, умножив на соответствующие коэффициенты
I2 + 6H2O + 5Cl2 → 2(IO3)Ц + 12H+ + 10ClЦ.
6. ƒобавив к ионам св€занные с ними частицы, составл€ем полное уравнение со всеми стехиометрическими коэффициентами
I2 + 5Cl2 + 6H2O → 10HCl + 2HIO3.
ѕример 8.5. Ќапишите формулы оксидов селена и соответствующих им кислот, назовите эти кислоты и образуемые ими соли.
– е ш е н и е
| Ёлемент | ќксид | ‘ормула кислоты | Ќазвание кислоты | Ќазвание соли |
| Se | SeO2 | H2SeO3 | —елениста€ | —еленит |
| Se | SeO3 | H2SeO4 | —еленова€ | —еленат |
«адачи
8.1. ƒл€ данного неметалла определите валентные электроны в нормальном и возбужденном состо€нии. ”кажите возможные степени окислени€. ќпишите физические свойства неметаллов.
“ а б л и ц а 8.1
| ¬ариант | Ќеметалл | ¬ариант | Ќеметалл |
| — | 1Т | Kr | |
| Si | 2Т | Xe | |
| N | 3Т | Rn | |
| P | 4Т | At | |
| As | 5Т | Se | |
| O | 6Т | B | |
| S | 7Т | Si | |
| Se | 8Т | P | |
| F | 9Т | Cl | |
| Cl | 10Т | N | |
| Br | 11Т | Br | |
| Ar | 12Т | S | |
| At | 13Т | Ar | |
| Ne | 14Т | I | |
| I | 15Т | O |
8.2. Ќапишите уравнени€ реакций взаимодействи€ данного неметалла с простыми веществами. акие химические свойства про€вл€ет неметалл в этих реакци€х? ак можно объ€снить его способность взаимодействовать с восстановител€ми и окислител€ми? Ќазовите продукты реакций.
“ а б л и ц а 8.2
| ¬ариант | Ќеметалл | ¬ещество | ¬ариант | Ќеметалл | ¬ещество |
| Cl2 | Na | 1Т | P | Ca | |
| Cl2 | Cu | 2Т | P | Na | |
| Cl2 | Fe | 3Т | P | Al | |
| Cl2 | H2 | 4Т | C | O2 (недост.) | |
| S | F2 | 5Т | C | O2 (изб) | |
| S | O2 | 6Т | C | F2 | |
| S | H2 | 7Т | C | S | |
| S | Na | 8Т | C графит | H2 | |
| S | Mg | 9Т | C графит | Ca | |
| S | Al | 10Т | C графит | Al | |
| P | F2 | 11Т | C графит | Si | |
| P | Cl2 | 12Т | C графит | O2 | |
| P | O2 | 13Т | Si | O2 | |
| P | S | 14Т | Si | Cl2 | |
| P | Mg | 15Т | Si | Mg |
8.3. ќпишите способы получени€ неметалла. Ќапишите уравнени€ реакций и услови€ их протекани€, принципиальную схему получени€.
“ а б л и ц а 8.3
| ¬ариант | Ќеметалл | ¬ариант | Ќеметалл |
| O2 | Te | ||
| S | At2 | ||
| F2 | B | ||
| Br2 | P | ||
| I2 | As | ||
| Se | Ar | ||
| C | Ne | ||
| N2 |
8.4. ќпишите окислительно-восстановительные свойства неме-таллов. ”равн€йте окислительно-восстановительные реакции ионно-электронным методом.
“ а б л и ц а 8.4
| ¬ариант | –еакци€ |
| Br2 + Cl2 + H2O = HBrO3 + HCl | |
| At2 + HClO + H2O = HAtO3 + HCl | |
| Cl2 + KIO3 + KOH = K2IO6 + KCl + 3H2O | |
| F2 + NaBrO3 + NaOH = NaBrO4 + NaF + H2O | |
| I2 + H2O2 = HIO3 + H2O | |
| Br2 + H2S + H2O = H2SO4 + HBr | |
| S + NaOH = Na2S + Na2SO3 + H2O | |
| Cl2 + H2O + Na2S2O3 = Na2SO4 + S + HCl | |
| Se + KOH = K2SeO3 + K2Se + H2O | |
| I2 + KOH + NH2OH = N2 + KI + H2O | |
| As + HNO3 + H2O = H3AsO4 + NO | |
| Si + HNO3 + HF = H2SiF6 + NO + H2O | |
| Si + H2O + KOH = K2SiO3 + 2H2 | |
| I2 + HNO3 = HIO3 + NO + H2O | |
| Cl2 + NaOH = NaCl + NaClO + H2O | |
| 1Т | Cl2 + KOH  KCl + KClO3 + H2O KCl + KClO3 + H2O
|
| 2Т | I2 + NO + H2SO4 + H2O = NaNO2 + NaI + H2SO4 |
| 3Т | I2 + NaHSO4 + H2O + MnSO4 = NaI + H2SO4 + MnO2 |
| 4Т | S + MnSO4 + K2SO4 + H2O = KMnO4 + H2S + H2SO4 |
| 5Т | S + K2SO4 + Cr2(SO4)3 + H2O = K2Cr2O7 + H2S +H2SO4 |
| 6Т | I2 + K2SO4 + NO2 + H2O = KI + HNO3 + H2SO4 |
| 7Т | Br2 + MnSO4 + Na2SO4 + K2SO4 + H2O = KMnO4 + NaBr + H2SO4 |
| 8Т | Cl2 + MnCl2 + KCl + H2O = KMnO4 + HCl |
| 9Т | Br2 + KOH + CrCl3 = K2CrO4 + KBr + H2O + KCl |
| 10Т | I2 + Na2SO4 + K2SO4 + H2O = Na2SO3 + KIO3 + H2SO4 |
| 11Т | I2 + NO + K2SO4 + H2O = KI + KNO2 + H2SO4 |
| 12Т | Cl2 + MnSO4 + NaHSO4 + H2O = MnO2 + NaCl + H2SO4 |
| 13Т | I2 + Cr2(SO4)3 + K2SO4 + H2O = K2Cr2O7 + KI + H2SO4 |
| 14Т | P + H2SO4 = H3PO4 + SO2 + H2O |
| 15Т | I2 + HNO3 = HIO3 + NO + H2O |
|
|
|
8.5. Ќапишите формулы оксидов селена и соответствующих им кислот, назовите эти кислоты и образуемые ими соли.
“ а б л и ц а 8.5
| ¬ариант | Ёлемент | ¬ариант | Ёлемент | ¬ариант | Ёлемент |
| N | C | Cl | |||
| P | Cl | Si | |||
| As | I | S | |||
| B | P | N | |||
| Br | S | P |
8.6. ќпределите массовую долю хлора в соединени€х: KClO, KClO2, KClO3, KClO4. Ќазовите эти соединени€.
8.7. ќпределите, как измен€етс€ устойчивость соединений хлора в р€ду: HClO Ц HClO3 Ц HClO4. акое из соединений €вл€етс€ самым сильным окислителем и почему?
8.8. ќпределите объем образовавшегос€ газа (при н.у.) при разложении 2,45 г бертолетовой соли. Ќапишите уравнение реакции.
8.9. ¬ 5 л воды растворили 11,2 л газообразного триоксида серы. –ассчитайте мол€рную концентрацию полученного раствора.
8.10. —оставьте уравнени€ реакций гидролиза трихлорида и трифторида бора. ”кажите все возможные продукты реакций.
8.11. –ассмотрите про€вление диагональной периодичности свойств в направлении бор → кремний. ”кажите свойства, сходные дл€ бора и кремни€ и одновременно различные дл€ бора и алюмини€, на примерах оксидов, гидроксидов, галогенидов и гидридов.
8.12. Ќапишите уравнени€ реакций взаимодействи€ фторидов натри€, алюмини€, кремни€, фосфора с водой. акие свойства (основные, кислотные, амфотерные) по отношению к воде про€вл€ют данные соединени€?
8.13. Ќапишите уравнени€ реакций взаимодействи€ хлоридов магни€, алюмини€, фосфора и серы с водой. акие свойства (основные, кислотные, амфотерные) про€вл€ют данные хлориды?
8.14. Ќапишите уравнени€ реакций взаимодействи€ нитридов натри€, алюмини€, фосфора и серы с водой. акие свойства по отношению к воде про€вл€ют данные соединени€ (основные, кислотные, амфотерные)?
8.15. Ќапишите уравнени€ реакций гидролиза следующих солей: Na2SO3, NaHSO3. ќпределите рЌ раствора. ¬ычислите рЌ 0,001 ћ раствора Na2SO3.
8.16. ак измен€ютс€ кислотные и окислительно-восстанови-тельные свойства в р€ду серна€ Ц селенова€ Ц теллурова€ кислоты? ƒайте обоснованный ответ. Ќапишите уравнени€ соответствующих реакций.
8.17. —равните устойчивость, окислительно-восстановительные и кислотные свойства следующих кислот: серниста€, селениста€, теллуриста€. —оставьте уравнени€ соответствующих реакций.
8.18. –ассчитайте объем сернистого газа, необходимого дл€ восстановлени€ 16,0 г хлорноватой кислоты до хлороводорода (н.у.). «апишите уравнение реакции.
8.19. ѕриведите примеры соединений азота, в которых осуществл€етс€ донорно-акцепторный механизм образовани€ ковалентной св€зи.
8.20. »звестны несколько аллотропных модификаций фосфора. ƒайте краткую характеристику каждой модификации.
8.21. ”кажите все аллотропные модификации углерода и различи€ в их свойствах.
8.22. ”кажите все аллотропные модификации мышь€ка и различи€ в их свойствах.
|
|
|
8.23. — позиций метода ¬— опишите строение молекул —Ќ4, —2Ќ6, —2Ќ4, —2Ќ2. акие типы гибридизации јќ характерны дл€ углерода в этих соединени€х?
8.24. арбид кальци€ получают восстановлением углерода по реакции
—аќ + — = —а—2 + —ќ.
¬ычислите массу —аќ, необходимую дл€ получени€ 4,5 т —а—2. акой объем —ќ при этом образуетс€ (н.у.)?
8.25. Ќапишите уравнени€ реакций, доказывающих кислотный характер оксидов SeO2, SO3, P2O5.
8.26. —оставьте формулы нормальных и кислых солей натри€, образованных мышь€ковистой кислотой.
8.27. —оставьте формулы нормальных и кислых солей натри€, образованных фосфорной кислотой.
8.28. —оставьте формулы нормальных и кислых солей натри€, образованных мышь€ковой кислотой.
8.29. —оставьте формулы нормальных и кислых солей натри€, образованных селенистой кислотой.
8.30. —оставьте формулы нормальных и кислых солей натри€, образованных селеновой кислотой.
8.31. —оставьте формулы соединений из указанных неметаллов:
Si, P, Cl, H.
8.32. акой объем водорода (н.у.) потребуетс€ дл€ восстанов-лени€ оксида меди (II), полученного при термическом разложении гидрооксида меди (II) массой 19,6 г? ѕриведите уравнени€ химических реакций.
8.33. —мешали 1 л хлора и 2 л водорода (н.у.). —колько граммов хлороводорода можно получить из такой смеси? „ему будет равен объем смеси после реакции?
8.34. ќпределите объем хлора, который может быть получен при взаимодействии 2 моль хлороводорода и 3 моль оксида марганца (IV). ѕриведите уравнение реакции.
8.35. –ассчитайте объем хлороводорода, полученный при взаи-модействии 20 л хлора с водородом.
8.36. ѕримен€ема€ в медицине йодна€ настойка €вл€етс€ 5 % раствором кристаллического йода в спирте. акой объем спирта, плотность которого равна 0,8 г/мл, требуетс€ дл€ приготовлени€ 250 г такого раствора?
8.37. Ќайдите, кака€ масса кислорода вступила в реакцию, если получено 71 г оксида фосфора (V).
8.38. Ќайдите, кака€ масса кислорода вступила в реакцию, если получено 4,4 г оксида углерода (IV).
8.39. Ќайдите, кака€ масса кислорода вступила в реакцию, если получено 4 г оксида магни€.
8.40. Ќайдите, кака€ масса кислорода вступила в реакцию, если получено 4,5 г оксида азота (II).
8.41. ќпределите массу 1 л смеси, содержащей 10 % озона и 90 % кислорода.
8.42. –ассчитайте количество кислорода, выделившеес€ при разложении 1 мол€ пероксида водорода Ќ2ќ2.
8.43. –ассчитайте количество кислорода, выделившеес€ при раз-ложении 1 мол€ хлората кали€ KClO3.
8.44. ¬ычислите массу сульфида цинка, котора€ образуетс€ при взаимодействии 2,24 л сероводорода (н.у.) с раствором нитрата цинка. ѕриведите уравнение реакции.
8.45. акое количество сернистого газа можно получить из 1 т руды, содержащей 48 % пирита?
8.46. ¬ычислите массовую долю сероводородной кислоты в растворе, полученном при растворении 44,8 л (н.у.) сероводорода в 20 л воды.
8.47. —колько граммов сернистого газа выделитс€ при взаимо-действии 6,4 г меди с концентрированной серной кислотой?
8.48. –ассчитайте массу сульфита натри€, необходимого дл€ получени€ 5,6 л SO2 (н.у.). ѕриведите уравнение реакции.
8.49. акое количество серы выпадает в осадок при пропускании 11,2 л сероводорода (н.у.) через раствор, содержащий 41 г серной кислоты?
8.50. —колько литров и молей аммиака требуетс€ дл€ получени€ 6,3 т азотной кислоты.
8.51. акую массу фосфорита, содержащую 70 % —а3(–ќ4)2, нужно вз€ть, чтобы получить из него 500 кг фосфора?
8.52. ѕри нагревании 20 г гидрокарбоната натри€ выделилось 2,24 л оксида углерода (IV) при н.у. ака€ массова€ дол€ (в %) гидрокарбоната разложилась?
8.53. —колько молекул SO2 получаетс€ при сгорании 2,0Ј10-6 кг серы?
8.54. ¬ какой массе четыреххлористого углерода CCl4 содержитс€ столько же молекул, сколько их в 5,0Ј10-3 м3 воды?
8.55. акой объем хлора необходимо пропустить через 0,35 л 0,2 н раствора тиосульфата натри€ до окислени€ его в серную кислоту?
8.56. акой объем азота может быть получен при термическом разложении 1 кг нитрита аммони€, если потери составл€ют 12 %?
8.57. ¬ычислите массу фосфора, которую надо сжечь дл€ получени€ оксида фосфора (V) массой 3,55 г.







