6.4. ѕри электролизе раствора данной соли металла током I, ј, масса катода возросла на m г. ”читыва€, что выход металла по току Bi, %, рассчитайте, какое количество электричества и в течение какого времени пропущено. —оставьте схему электролиза.
“ а б л и ц а 6.4
| ¬ариант | ‘ормула соли | I, A | m, г | Bi (Me), % |
| AuCl3 | 0,3 | 0,92 | ||
| FeCl2 | 0,9 | 0,77 | ||
| SnBr2 | 2,1 | 0,84 | ||
| CuSO4 | 0,79 | 0,62 | ||
| AgNO3 | 1,94 | 0,31 | ||
| ќкончание табл. 6.4 | ||||
| CdBr2 | 3,79 | 0,88 | ||
| MnCl2 | 1,12 | 0,94 | ||
| CoCl2 | 1,5 | 1,12 | ||
| CrCl3 | 2,11 | 1,18 | ||
| In(NO3)3 | 3,17 | 0,64 | ||
| SbF3 | 0,99 | 0,77 | ||
| CdCl2 | 1,77 | 0,84 | ||
| NiSO4 | 1,1 | 0,12 | ||
| Co(NO3)2 | 0,9 | 0,2 | ||
| ReCl3 | 1,94 | 0,91 | ||
| 1Т | PdSO4 | 2,03 | 1,17 | |
| 2Т | CdI2 | 3,11 | 1,42 | |
| 3Т | SbCl3 | 4,0 | 1,50 | |
| 4Т | CoSO4 | 4,4 | 1,39 | |
| 5Т | CrBr3 | 3,5 | 1,44 | |
| 6Т | InCl3 | 4,4 | 1,61 | |
| 7Т | MnSO4 | 2,1 | 0,92 | |
| 8Т | SnCl4 | 1,1 | 0,89 | |
| 9Т | CdSO4 | 1,8 | 0,97 | |
| 10Т | CrCl2 | 1,9 | 0,95 | |
| 11Т | InBr3 | 3,1 | 0,85 | |
| 12Т | PdCl2 | 4,2 | 1,32 | |
| 13Т | Ni(NO3)2 | 1,7 | 1,43 | |
| 14Т | Cr(NO3)3 | 1,5 | 1,11 | |
| 15Т | CuCl2 | 1,9 | 1,18 |
6.5. ”становите, в какой последовательности веро€тно восстановление на катоде при электролизе данных ионов, пользу€сь значени€ми стандартных электродных потенциалов и перенапр€жений. ќбъ€сните, когда возможно совместное восстановление металла и водорода на катоде. „ем отличаетс€ последовательность электрохимических реакций на аноде от аналогичной последовательности на катоде?
“ а б л и ц а 6.5
| ¬ариант | »оны | ¬ариант | »оны |
| Zn2+,Ag+,Pb2+,Cr3+,H+ | 1Т | Nd3+,Sn4+,Cr2+,Ni2+,H+ | |
| Mn2+,Co2+,Fe2+,Cd2+,H+ | 2Т | In3+,Fe3+,Zn2+,Pb2+,H+ | |
| H+,Cu2+,Sn2+,Au3+,Sb3+ | 3Т | Cr3+,Mo2+,Fe2+,Cd2+,Sn2+ | |
| Re3+,Pd2+,Sn4+,H+,Cu2+ | 4Т | Au3+,Re3+,Sn4+,H+,Cd2+ | |
| ќкончание табл. 6.5 | |||
| H+,Cr2+,Ni2+,In3+,Fe3+ | 5Т | Zn2+,Ag+,Cr3+,Fe3+,H+ | |
| H+,Zn2+,Pb2+,Mn2+,Fe2+ | 6Т | Au3+,Sb3+,Sn4+,H+,Fe3+ | |
| Cu2+,H+,Au3+,Re3+,Pb2+ | 7Т | Fe3+,In3+,Ni2+,Cr2+,H+ | |
| Sn4+,Ni2+,Fe3+,H+,Cr2+ | 8Т | Sn4+,Re3+,Sb3+,H+,Ni2+ | |
| Zn2+,Cr3+,Fe2+,Sn2+,H+ | 9Т | Au3+,Cu2+,Cd2+,Fe2+,H+ | |
| Re3+,Sn4+,In3+,Fe3+,H+ | 10Т | Mn2+,Cr3+,Pb2+,H+,Zn2+ | |
| Zn2+,Mn2+,Cu2+,Re3+,H+ | 11Т | Ag+,Cd2+,Pd2+,Fe3+,H+ | |
| Cr2+,Fe3+,H+,Zn2+,Ag+ | 12Т | Zn2+,Co2+,Au3+,Sn4+,H+ | |
| Pb2+,Cr3+,Mn2+,Co2+,H+ | 13Т | Ag+,Co2+,Sn2+,Pd2+,H+ | |
| Fe2+,Cd2+,Cu2+,Sn2+,H+ | 14Т | Pb2+,Fe2+,Au3+,Nd3+,H+ | |
| Au3+,Sb3+,Re3+,Pd2+,H+ | 15Т | H+,Zn2+,Mn2+,Cu2+,Re3+ |
6.6. ѕроведите классификацию данных окислительно-вос-становительных реакций, составьте уравнени€, рассчитайте  и укажите веро€тные направлени€ их протекани€.
и укажите веро€тные направлени€ их протекани€.
1) Au + H2SO4 + HCl  Au2Cl6 + H2SO3 + H2O;
Au2Cl6 + H2SO3 + H2O;
|
|
|
2) K—lO3 (к)  KCl (к) + O2 (г);
KCl (к) + O2 (г);
3) —l2 + KOH  KCl + KClO3 + H2O.
KCl + KClO3 + H2O.
6.7. ƒл€ электродов, представленных в таблице, укажите типы Ц первого, второго рода или окислительно-восстановительные, рассчитайте электродные потенциалы при заданных услови€х.
| Cхема электрода | —u+ | Cu0 | AgCl | Ag,Cl- | Pt | CuCl2, CuCl | |
”слови€
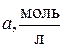
| 
| 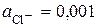
| 

| |
| Hg2Cl2 | Hg, Cl- | Hg2O | Hg, OH- | |||

| 
| |||
6.8. –ассчитайте активность ионов Ni2+ в электролите гальванического элемента
Ni | NiSO4, H2O || ZnSO4 | Zn,
если  моль/л; ≈ = 0,5425 ¬. —оставьте уравнение протекающей окислительно-восстановительной реакции в сокращен-ном ионном виде.
моль/л; ≈ = 0,5425 ¬. —оставьте уравнение протекающей окислительно-восстановительной реакции в сокращен-ном ионном виде.
6.9. –ассчитайте Ёƒ— гальванического элемента, образованного оловом и свинцом в растворах перхлоратов
Sn | Sn(ClO4)2, H2O || Pb(ClO4)2, H2O | Pb,
если известно отношение активностей:  . —оставьте уравнение протекающей окислительно-восстановительной реакции в сокращенном ионном виде.
. —оставьте уравнение протекающей окислительно-восстановительной реакции в сокращенном ионном виде.
6.10. »сход€ из схемы свинцового (кислотного) аккумул€тора Pb | H2SO4 | PbO2, составьте уравнени€ реакций на электродах и уравнение окислительно-восстановительной реакции при разр€де аккумул€тора, выведите уравнение дл€ расчета Ёƒ—. –ассчитайте Ёƒ— при  моль/л,
моль/л,  1 моль/л.
1 моль/л.
6.11. ¬ цинк-медноокисном элементе Zn | NaOH | CuO протекают различные реакции:
1) Zn + 2Cu(OH)2 = ZnO + Cu2O + 2H2O;
2) Zn + Cu2O = ZnO + 2Cu;
3) Zn + CuO = ZnO + Cu;
4) Zn + Cu(OH)2 = ZnO + Cu + H2O;
5) Zn + 2CuO = ZnO + Cu2O.
–ассчитайте  дл€ каждой из них и, сравнив с экспериментальным значением Ёƒ— элемента 0,89 ¬, сделайте вывод, кака€ из реакций €вл€етс€ основной.
дл€ каждой из них и, сравнив с экспериментальным значением Ёƒ— элемента 0,89 ¬, сделайте вывод, кака€ из реакций €вл€етс€ основной.
6.12. ¬ цинк-йодатном элементе положительна€ активна€ масса содержит 57,1 % KIO3 и протекает реакци€:
5Zn + 2KIO3 + 6H2SO4 = 5ZnSO4 + I2 + K2SO4 + 6H2O.
Cоставьте схему элемента. –ассчитайте величину положительной активной массы, необходимой дл€ работы элемента в течение 4 ча-сов при силе тока 2 ј, выходе по току 100 %.
6.13. ¬ герметичном колодце установлен цинк-воздушный элемент Zn | KOH | O2 (—). “ок разр€да 1 ј, выход по току 1, содержание кислорода 21 %, объем колодца 0,21 м3. –ассчитайте, через какое врем€ будет израсходовано 15 % кислорода в атмосфере колодца (услови€ нормальные).
6.14. ѕо уравнени€м реакций при разр€де аккумул€торов составьте схемы электрохимических систем, рассчитайте 
1) 2NiOOH + 2H2O + Cd = 2Ni(OH)2 + Cd(OH)2;
2) 2NiOOH + 2H2O + Fe = 2Ni(OH)2 + Fe(OH)2;
3) 2NiOOH + Zn + H2O = 2Ni(OH)2 + ZnO.
6.15. ѕо схемам реакций при разр€де гальванических элементов составьте полные молекул€рные уравнени€, рассчитайте 
1) SO2Cl2 + Li  LiCl + SO2 + S;
LiCl + SO2 + S;
2) MnO2 + Mg + H2O  MnOOH + Mg(OH)2;
MnOOH + Mg(OH)2;
3) Mg + C6H4(NO2)2 + H2O  Mg(OH)2 + C6H4(NHOH)2.
Mg(OH)2 + C6H4(NHOH)2.
6.16. »сход€ из схем первых вариантов химических источников тока, составьте уравнени€ токообразующих реакций, рассчитайте  :
:
1) элемент ƒаниэл€-якоби Zn | ZnSO4 || CuSO4 | Cu;
2) элемент ƒаниэл€-якоби (вариант якоби) Zn | NH4Cl || CuSO4 | Cu;
3) элемент ¬ольта Zn | H2O | (Ag, Cu);
4) элемент √рене Zn | K2Cr2O7, H2SO4 | (—);
5) элемент Ѕунзена Zn | H2SO4 | HNO3 | (—).
6.17. »сход€ из схем топливных элементов, составьте уравнени€ токообразующих реакций и рассчитайте 
1) CH3OH (Pt) | H2SO4 | O2 (Pt);
2) N2H4 (Ni) | KOH | O2 (C, Ag);
3) H2 (Pt) | H2SO4 | O2 (Pt);
4) CH3OH (Pt) | KOH | O2 (C, Ag);
5) H2 (Ni) | KOH | O2 (C, Ag).
6.18. “ермоэлектрохимические циклы позвол€ют преобразовать тепловую энергию в электрическую без помощи тепловых машин, например, по реакции
|
|
|
CuCl2  CuCl + ½ Cl2.
CuCl + ½ Cl2.
ѕроцесс (1) реализуетс€ при “ > 500 о—, процесс (2) Ц в гальва-ническом (топливном) элементе при стандартных услови€х. ƒл€ процесса (2) рассчитайте  . ќбъ€сните, почему с повы-шением температуры начинаетс€ реакци€ (1)?
. ќбъ€сните, почему с повы-шением температуры начинаетс€ реакци€ (1)?
6.19. ѕри работе гальванического элемента протекает реакци€
Zn + Cu2O = ZnO + 2Cu.
«а 1 час работы элемента выделилось 0,45 г меди. –ассчитайте ток в цепи и массу растворившегос€ цинка. ¬ыходы по току данных метал-лов равны 65 %.
6.20. ≈сли соединить оксиды Ag2O (т) и ZnO (т) внешней цепью и погрузить в водный раствор ќЌ, то в системе начинает протекать электрический ток. —оставьте схему гальванического элемента, уравнение протекающей окислительно-восстановительной реакции, рассчитайте  , укажите направление ее протекани€.
, укажите направление ее протекани€.
6.21. ѕри получении алюмини€ электролизом расплавов происходит электрохимическое сгорание углеродистых анодов
3— + 2Al2O3 = 3CO2 + 4Al.
¬ыход по току —ќ2 равен 60 %. ќбъем выделившегос€ оксида углерода (IV) 70 л (после пересчета к нормальным услови€м). –ассчитайте количество пропущенного электричества и массу сгоревших анодов.
6.22. ѕри электролитическом получении железного порошка ток был пропущен как через ванну с раствором FeSO4, так и через последовательно соединенную с ней ванну с раствором CuSO4. оличество осажденной меди составило 10,5 г. –ассчитайте количество пропущенного электричества и массу полученного железного порошка, если выхода по току: железа 70 %, меди Ц 95 %.
6.23. ѕри электролитическом рафинировании меди регенерацию раствора CuSO4 провод€т электролизом с нерастворимыми анодами. –ассчитайте продолжительность процесса и массу осажденного металла, если при электролизе выделилось 500 мл ќ2 (объем пере-считан к нормальным услови€м), ток 2 ј, выхода по току: кислорода 100 %, меди Ц 90 %.
6.24. ѕри электролитическом хромировании протекают реакции
а) CrO3  Cr + 1,5 O2;
Cr + 1,5 O2;
б) Cr6+ + 3 
 Cr3+;
Cr3+;
в) Cr3+  Cr6+ + 3
Cr6+ + 3  ;
;
г) Ќ2ќ  Ќ2 + ½ O2.
Ќ2 + ½ O2.
”кажите анодные и катодные продукты этих реакций, рассчитайте массу хромового покрыти€, полученного при токе 5 ј, продол-жительности процесса 2 часа, выходе хрома по току 13 %.
6.25. «а 40 минут цинковани€ при токе 34 ј из раствора ZnSO4 с растворимыми цинковыми анодами выделилось 1,2 л водорода (объем пересчитан к нормальным услови€м). –ассчитайте выход цинка по току.
6.26. ѕри вытравливании рисунка печатных плат протекают реакции
а) анодного травлени€: Cu + 3ClЦ  CuCl
CuCl 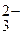 +
+ 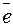 ;
;
б) химического травлени€: Cu + CuCl2 + 4KCl  2K2CuCl3.
2K2CuCl3.
ѕри токе 5 ј, продолжительности процесса 2 часа, анодном выходе меди по току 90 % удалено 27,24 г металла. –ассчитайте долю анод-ной составл€ющей в травлении.
6.27. ѕри оксидировании меди протекают следующие анодные реакции
Cu + 2OHЦ= CuO + H2O + 2  ;
;
4OHЦ= O2 + 2H2O + 4  .
.
ќбъем выделившегос€ кислорода составил 0,9 л (после пересчета к нормальным услови€м), выход кислорода по току 6 %. –ассчитайте количество пропущенного электричества и массу меди, израсходованной на образование оксидной пленки.
6.28. ƒл€ получени€ медных покрытий ток при электролизе водного раствора CuSO4 мен€ют по закону

где Q = 1 AЈч Ц количество электричества, AЈч; (t+ 3) Ц врем€ в часах, (t, ч).
–ассчитайте массу покрыти€, полученного при продолжительности электролиза 2 часа, выходе меди по току 90 %.
6.29. ѕри электролизе раствора NaCl с графитовыми электродами протекает реакци€ окислени€ графита на аноде
— + 4ќЌЦ  —ќ2 + 2Ќ2ќ + 4
—ќ2 + 2Ќ2ќ + 4  .
.
–ассчитайте расход анодно окислившегос€ графита, если при электролизе выделилось 36 л —ќ2 (объем пересчитан к нормальным услови€м), выход по току —ќ2 1,5 %. акое количество электри-чества было пропущено?
6.30. ѕри электролизе водного раствора MgCl2 с графитовыми электродами на получение 120 л хлора (объем пересчитан к нор-мальным услови€м) затрачено 12,5Ј105 л. –ассчитайте выход хлора по току.
6.31. ƒл€ получени€ хлората натри€ провод€т электролиз, при котором протекает реакци€
|
|
|
NaCl + 3H2O = NaClO3 + 3H2.
–ассчитайте, какое количество NaClO3 можно получить при токе 15 ј, если при электролизе выдел€етс€ 10 л водорода (объем пересчитан к нормальным услови€м). акова будет продолжи-тельность электролиза? ¬ыход по току NaClO3 равен 70 %.
6.32. ѕри электрохимическом получении перманганата кали€ протекает следующа€ реакци€
K2MnO4 + H2O  KMnO4 + KOH + ½ H2.
KMnO4 + KOH + ½ H2.
ѕри электролизе продолжительностью 3 часа выделилось 5 л водо-рода (объем пересчитан к нормальным услови€м). –ассчитайте массу полученного KMnO4 и ток. ¬ыход по току KMnO4 равен 70 %.
6.33. ѕри электролизе раствора кислоты выделилось 15 л водо-рода. –ассчитайте ток, если продолжительность процесса составила 2 часа, температура 18 о—, давление 0,9891Ј105 ѕа, выход по току 100 %.
6.34. ѕри электрохимическом получении пербората натри€ протекает реакци€
Na2B4O7 + 2NaOH + 19H2O = 4(NaBO3Ј4H2O) + 4H2.
–ассчитайте массу полученного пербората натри€ и продолжительность электролиза, если ток равен 20 ј, объем выде-лившегос€ водорода составил 8,1 л, выход по току водорода 15 %.
6.35. —оставьте уравнение химической реакции при электролизе раствора NaCl и рассчитайте, какое количество щелочи можно получить при токе 17,5 ј, продолжительности процесса 1,5 часа, выходе по току 100 %.
6.36. Ќа положительном электроде свинцового аккумул€тора при разр€де протекает реакци€
PbO2 + SO 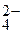 + 4H+ + 2
+ 4H+ + 2 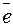 = PbSO4 + 2H2O.
= PbSO4 + 2H2O.
Ќа сколько изменилась масса положительного электрода, если аккумул€тор разр€жали током 50 ј в течение 20 c?
6.37. ѕри анодном растворении 50 г цинковой руды металл был полностью переведен в раствор за 1 ч при силе тока 10 ј, выходе по току 60 %. –ассчитайте массу выделившегос€ металла и его содержание в руде.
6.38. »з раствора AgNO3 полностью выдел€етс€ серебро при электролизе за 1 ч, при I = 1,5 A, Bi (Ag) = 0,8.
ѕри каком токе надо вести электролиз, чтобы исходна€ концентраци€ раствора по ионам Ag+ уменьшилась вдвое в течение 15 мин? ¬ыход по току прин€ть тот же.
6.39. ќпределить степень окислени€ золота в растворе его хлорида, если известно, что при электролизе раствора током в 1 ј за 4 мин выделилось 0,42 г золота при выходе металла по току 85 %.
6.40. ѕри электролизе раствора NaOH графитовые аноды постепенно разрушаютс€ по реакции
— + 2ќЌ  → —ќ2 + 2Ќ2ќ + 4
→ —ќ2 + 2Ќ2ќ + 4 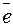 .
.
–ассчитайте массу окислившегос€ графита и объем выделившегос€ —ќ2 (услови€ нормальные) при I = 10 ј, t = 2 ч, выходе по току графита 1,5 %.
’»ћ»я ћ≈“јЋЋќ¬
–ешение типовых задач
ѕример 7.1. ƒл€ тантала (“а) выпишите валентные электроны в нормальном и возбужденном состо€ни€х, перечислите все теоретически возможные степени окислени€, приведите формулы оксидов и гидроксидов дл€ наиболее устойчивых степеней окислени€, указав их окислительно-восстановительный и кислотно-основной характер.
– е ш е н и е
“антал є 73 (“а).
¬алентные электроны тантала:


 нормальное состо€ние
нормальное состо€ние
6 s
 |
5 d; степени окислени€: 0, 1+, 2+, 3+;
¬озбужденное состо€ние
 |
 6 р
6 р


6 s








5 d; степени окислени€: 0, 1+, 2+, 3+, 4+, 5+.
ќксиды: “аќ; “а2ќ3; “а2ќ5
основной амфотерный кислотный.
√идроксиды: “а(ќЌ)2; “а(ќЌ)3.
ќкислительно-восстановительные свойства:
“а+ может отдать 4 электрона и присоединить 1 электрон;
“а2+ может отдать 3 электрона и присоединить 2 электрона;
“а3+ может отдать 2 электрона и присоединить 3 электрона;
“а4+ может отдать 1 электрон и присоединить 4 электрона;
следовательно, все они могут быть восстановител€ми и окислител€ми;
|
|
|
“а5+ Ц безусловный окислитель, так как может только принимать электроны.
ѕример 7.2. «апишите предполагаемое уравнение химической реакции взаимодействи€ меди с азотной кислотой (концентри-рованной). ”равн€йте стехиометрические коэффициенты ионно-электронным методом. –ассчитайте Δ G 0 химической реакции и сделайте вывод о ее термодинамической веро€тности без учета перенапр€жени€.
– е ш е н и е
n 
 |
Cu + HNO3 (конц) = Cu(NO3)2 + H2O + NO2↑
восст. окисл.
Cu0 Ц 2   Cu2+
(NO3) Ц + 2H+ + 1 Cu2+
(NO3) Ц + 2H+ + 1   NO2 + H2O NO2 + H2O
| |
 Cu + 2(NO3) Ц + 4H+ = Cu2+ + 2NO2 + 2H2O Cu + 2(NO3) Ц + 4H+ = Cu2+ + 2NO2 + 2H2O
|
2NO 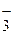 2NO
2NO 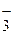
Cu + 4HNO3 = Cu(NO3)2 + 2NO2↑ + 2H2O.
φ  = + 0,34 ¬; φ
= + 0,34 ¬; φ  = +0,77 ¬; ≈ 0 = φ
= +0,77 ¬; ≈ 0 = φ  Ц φ
Ц φ  ;
;






