Для того чтобы наиболее точно зафиксировать точку эквивалентности титрования, необходимо выбрать такой индикатор, который резко меняет свою окраску при значении рН, максимально близком к эквивалентной точке. Изменение рН при нейтрализации различных по степени диссоциации кислот и оснований обычно изображают графически. Такие графики постепенного изменения рН при кислотно-основном титровании называют кривыми нейтрализации, или кривыми титрования кислотно – основного метода. По таким кривым можно выбрать нужный индикатор. В качестве титрантов в кислотно – основном методе обычно применяют сильные кислоты или сильные основания, поэтому здесь возможны следующие 3 случая:
1) титрование сильной кислоты сильным основанием (и наоборот),
2) титрование слабой кислоты сильным основанием,
3) титрование слабого основания слабой кислотой.
Рассмотрим ход кривой титрования более подробно на примере (1), при нейтрализация сильной кислоты сильным основанием. Предположим, что мы титруем раствор HCl 0,1 н концентрации раствором NaOН той же концентрации. Сильные основания и сильные кислоты практически полностью диссоциированны на ионы, поэтому концентрацию ионов [Н+] можно принять численно равной общей концентрации кислоты. Таким образом, до начала реакции нейтрализации раствор HCl будет иметь концентрацию ионов
[H+]=10–1г-ион/л, а рН=1. После нейтрализации 90% кислоты, ее осталось 10%от первоначального количества. Следовательно, количество кислоты уменьшилось в 10 раз. Для упрощения вычисления мы пренебрегаем увеличением общего объема раствора.
При уменьшении общей концентрации кислоты в 10 раз, во столько же раз уменьшится концентрация ионов водорода, т.е. [H+] = 10–2, а рН= 2.
После нейтрализации 99% кислоты общая ее концентрация уменьшилась в 100 раз по сравнению с первоначальной концентрацией. Следовательно, концентрация ионов водорода [H+] = 0,001 = 10–3,в этом случае рН=3. После нейтрализации 99,9% кислоты [H+] = 0,0001 = 10–4, рН=4. Когда же будет нейтрализована вся кислота (100%), в растворе будет содержаться только соль NaCl, не подвергающаяся гидролизу. Концентрация [Н+] и [ОН–] при этом одинаковы, а рН =7 и точка эквивалентности совпадает с точкой нейтральности (таблица 2).
Если и далее приливать щелочь к титруемому раствору, концентрация ионов [ОН–] будет увеличиваться в том же порядке, как шло уменьшение ионов Н+. При добавлении 1% избытка щелочи концентрация ионов будет
[OH–] = 10–3моль/л, а [Н+] = 10–11моль/л, значит рН =11. При добавлении 10% избытка NaOH концентрация ионов [OH–] = 10–2моль/л, в этом случае
[H +] = 10–12моль/л и рН = 12.
Если добавить 100% избытка щелочи, то концентрация ионов гидроксила в растворе будет равна 10–1моль/л, [H+] = 10–13моль/л, а рН = 13.
Таким образом, при нейтрализации 99,9% кислоты рН изменяется всего на три единицы от 1,0 до 4,0, а переход от 0,1% избытка кислоты к 0,1% избытку щелочи изменяет рН раствора от 4,0 до 10,0. В конце титрования 1–2 капли щелочи снижают концентрацию [Н+] в растворе от 10–4 до 10–10, т.е. в миллион раз. Такое резкое изменение величины рН вблизи эквивалентной точки, т.е. в конце титрования называется скачкомтитрования, при этом резко меняется окраска индикатора. Это говорит о том, что при выборе индикатора нужно руководствоваться не столько величиной рН в точке эквивалентности, сколько интервалом перехода окраски индикатора. В рассмотренном выше случае можно использовать любой кислотно-основной индикатор, интервал перехода окраски которого лежит в пределах значений рН от 4 до 10. В данном интервале для фиксирования точки эквивалентности можно использовать и фенолфталеин и метилоранж (таблица 2).
Таблица 2
Результаты титрования сильной кислоты сильным основанием
| Изменение [Н+] и рН при нейтрализации 0,1н. раствора сильной кислоты | ||||
| Ход нейтрализации | Н+ | рН | Примечания | |
| Нейтрализовано кислоты, % | ||||
| 0,0 | 10 | 1,0 | ||
| 90,0 | 10 | 2,0 | ||
| 90,0 | 10 | 3,0 | ||
| 99,9 | 10 | 4,0 | ||
| 100,0 | 10 | 7,0 | Раствор нейтральный. Точка эквивалентности | |
| Прибавлен избыток щелочи, % | ||||
| 0,1 | 10 | 10,0 | ||
| 1,0 | 10 | 11,0 | ||
| 10,0 | 10 | 12,0 | ||
| 100,0 | 10 | 13,0 | ||
Изменение рН раствора от количества прибавленного раствора сильного основания к сильной кислоте можно выразить графически. На кривой титрования (рис.2) четко видно, что рН раствора в процессе титрования изменяется с разной скоростью. В начале титрования при добавлении щелочи от 0 до 99,9 % рН медленно изменяется всего на 3,3 единицы. При дальнейшем прибавлении щелочи около 100,1% рН резко меняется на 5,6 единицы. Такое резкое изменение рН вблизи точки эквивалентности и есть скачок титрования. Дальнейшее изменение рН происходит медленно. Точка эквивалентности на рисунке совпадает с точкой нейтральности, т.е. находится при рН = 7. Скачок титрования в данном случае не зависит от концентрации сильной кислоты и щелочи.
§5. Лабораторный практикум
«Кислотно-основное титрование»
Цель работы:
1. Приобрести навыки титрования.
2. Научиться стандартизировать растворы с приготовленным титром.
3. Овладеть методикой титрования реакционной смеси в присутствии двух индикаторов.
Реактивы и оборудования:
Бюретки на 25 мл в штативе, химические воронки, пипетки Мора на 5мл и 10мл, колбы конические для титрования на 100мл, резиновые груши, стаканчики для слива; стандартный раствор тетрабората натрия 0,1N концентрации, раствор НС1, индикаторы: метилоранжевый и фенолфталеин; исследуемый раствор, состоящий из смеси гидрокарбоната натрия и карбоната натрия.
Задание 1. «Стандартизация титранта – раствора НС l»
Полученный раствор НСl, имеет приблизительную концентрацию. Стандартизацию проводят с помощью тетрабората натрия. Реакция протекает по следующему уравнению:
Na2B4O7 + 2HCl + 5H2O → 4H3BO3 + 2NaCl
Ход работы:
1. Готовят бюретку к работе. Для этого в чистую бюретку через воронку приливают приготовленный раствор HCl до “0” отметки.
2. Приготавливают раствор тетрабората натрия для титрования. В коническую колбу для титрования вносят пипеткой Мора 5мл, приготовленного раствора буры и 1–2 капли метилоранжа.
3. Титруют приготовленный раствор буры раствором хлороводородной кислотой из бюретки, добавляя в колбу небольшими порциями (0,2 – 0,3мл).
Титрование при изменении окраски индикатора от желтой окраски до красной. Результаты титрования заносят в таблицу.
Первое титрование ориентировочное. Второе и последующие титрования проводят более точно, добавляя в конце титрования по каплям кислоту и все результаты титрования заносят в таблицу 3.
 Таблица 3.
Таблица 3.
Результаты опыта
| № | 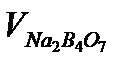
| 
| 
| 
| 
|
| 1. | |||||
| 2. | |||||
| 3. | |||||
| Ср. |
Рассчитывают концентрацию раствора хлороводородной кислоты по среднему объёму кислоты.
Задание 2. «Определение карбоната и гидрокарбоната при совместном присутствии в растворе»
Гидрокарбонат натрия (питьевая сода) может содержать примеси карбоната натрия. Определение содержания обоих веществ в пробе осуществляется титрованием хлороводородной кислотой.
В основе анализа протекают следующие реакции.
1.Na2CO3 + 2HCl = 2NaCl + H2O
CO32– + H+ = HCO–3
2.NaHCO3 + HCl = NaCl + CO2 + H2O
HCO3– + H+ = CO2 + H2O
Реактивы: Титрованный раствор HCl – 0,1N, исследуемые растворы гидрокарбоната натрия разной концентрации, индикатор метилоранжевый и фенолфталеин.
Ход работы:
1. Бюретку заполняют раствором HCl.
2.В колбу для титрования вносят мерной пипеткой 5мл исследуемого раствора, добавляют 1–2 капли фенолфталеина и титруют до обесцвечивания. Отмечают объём на бюретке – V1 это объём кислоты – эквивалентный половине, содержащегося в растворе карбоната.
3.Добавляют в ту же колбу 1–2 капли индикатора метилоранжевого и титруют кислотойдоперехода жёлтой окраски в бледно-розовую. Отметить объём на бюретки – V2 – объём, эквивалентный – гидрокарбонату и половине карбоната, т.к. часть его была уже оттитрована. Следовательно, объём кислоты, идущий на титрование всего карбоната равен 2V1, а объём, идущий на титрования гидрокарбоната, равен V2 – V1.
Титрование с пробой повторить ещё 2–3 раза. Данные титрования поместить в таблицу 4.
Таблица 4.
Результаты опыта
| № | Объем анализируемого р-ра, V. | Первый отсчёт по бюретке, V1. | Второй отсчёт по бюретке, V2 | Объем кислоты затраченный на титрование карбоната, 2V1 | Объем кислоты, затраченный на титрование гидрокарбоната (V2 – 2V1) | Нормальность кислоты, NHCl |
| 1 | ||||||
| 2 | ||||||
| 3 |
Расчёты N, T и m каждого компонента смеси проводить по средней величине объёмов.
§6. Задачи для самостоятельного решения
Пример 1.
Чему равна молярная масса эквивалента серной кислоты при титровании раствором KOH?
Решение.
При титровании H2SO4 раствором KOH протекает реакция
H2SO4 + 2КOH → К2SO4 + 2H2O
Разделим левую и правую части уравнения на 2
½H2SO4 + КOH → ½К2SO4 + H2O
Становится очевидным, что f э(H2SO4)=1/2. Тогда молярная масса эквивалента серной кислоты равна
M (1/2H2SO4)=1/2 M (H2SO4)=1/2×98,07=49,035 г/моль
Пример 2.
Навеску H2C2O4×2H2O массой 0,6000 г растворили в мерной колбе вместимостью 100,0 мл. На титрование аликвоты 20,00 мл полученного раствора израсходовано 18,34 мл NaOH. Определить молярную концентрацию раствора NaOH, его титр и титр этого раствора по H2C2O4.
Решение.
В соответствии с принципом эквивалентности число молей эквивалента титранта равно числу молей эквивалента определяемого вещества
n (NaOH)= n (1/2H2C2O4×2H2O),
где n (NaOH) – число молей эквивалента щелочи, затраченное на титрование аликвоты раствора щавелевой кислоты.
Запишем уравнение реакции, протекающей при титровании
H2C2O4 + 2NaOH → Na2C2O4 + 2H2O
Каждая молекула щавелевой кислоты содержит 2 иона водорода, поэтому f э(H2C2O4×2H2O)=1/2. Из условия задачи следует, что
 ,
,
где V к – объем мерной колбы, в которой растворена навеска щавелевой кислоты; V а – объем аликвоты полученного раствора, отобранного пипеткой на титрование.
Если обозначить молярную концентрацию раствора гидроксида натрия C (NaOH), то количество вещества эквивалента NaOH можно выразить как

Объединяя полученные выражения, получаем


Титр T (NaOH) и титр по щавелевой кислоте T (NaOH/H2C2O4) в г/мл можно рассчитать по формулам
 ;
;
 ,
,
где M (NaOH) и M (1/2H2C2O4×2H2O) – молярные массы эквивалента NaOH и щавелевой кислоты соответственно.
 ;
;
 .
.
1. Сколько грамм буры (Na2B4O7·10Н2О) следует взять для приготовления 250мл 0,1N раствора.
2. Рассчитайте, какой объем хлороводородной кислоты с плотностью, равной 1,180г/мл, потребуется для приготовления 200мл раствора с концентрацией кислоты в 0,05моль/л.
3. На титрование 5,00мл раствора серной кислоты израсходовано 4,12мл 0,1020М раствора NaOH. Вычислите нормальность и рН раствора кислоты.
4. При титровании 30 мл раствора гидроксида натрия для его нейтрализации потребовалось 60 мл 0,5 н. раствора соляной кислоты. Вычислите нормальность и молярность раствора гидроксида натрия.
5. Какую навеску щавелевой кислоты H2C2O4*2Н2О нужно взять, чтобы на ее титрование израсходовать 20 мл 0,1н NaOH?
6. 10 мл анализируемого раствора HCl разбавили в мерной колбе на 200 мл. На титрование 20 мл полученного раствора HCl пошло 10,40 мл 0,1 н раствора NaOH. Определить содержание (в гр.) кислоты в объеме 10 мл, взятом для разбавления.
§7. Тестовые задания для самостоятельного решения
Выберите один правильный ответ
001. ТИТРИМЕТРИЧЕСКИЙ АНАЛИЗ – ЭТО…
1) метод количественного анализа, основанный на точном измерении объема раствора определяемого вещества
2) метод качественного анализа, основанный на измерении объема раствора титранта
3) метод количественного анализа, основанный на точном измерении объема раствора реагента, необходимого для эквивалентного взаимодействия с определяемым веществом
4) метод количественного анализа, основанный на точном измерении объема индикатора
5) метод количественного анализа, основанный на точном измерении массы раствора реагента
002. ТОЧКА ЭКВИВАЛЕНТНОСТИ СООТВЕТСТВУЕТ рН = 7 ПРИ ТИТРОВАНИИ
1) NH3+HCl
2) KOH + HCl
3) HNO2+ KOH
4) NH3 + HNO2
5) H2C2O4 + KOH
003. НАВЕСКА H2C2O4*2H2O, ДЛЯ ПРИГОТОВЛЕНИЯ 25 МЛ 0,05Н РАСТВОРА, РАВНА
1) 80 г
2) 0,08 г
3) 0,8 г
4) 160 г
5) 16 г
004. В ОСНОВЕ РАСЧЕТОВ В АЦИДИМЕТРИИ ЛЕЖИТ ЗАКОН:
1) сохранения заряда,
2) эквивалентов
3) сохранения массы вещества
4) сохранения энергии
5) постоянства состава вещества
005. ИНДИКАТОР - ЭТО
1) растворимые неорганические соединения, способные изменять окраску раствора в зависимости от pH раствора,
2) растворимые сложные органические соединения, способные изменять собственную окраску в зависимости от pH раствора,
3) растворимые сложные органические соединения, способные изменять собственную окраску в зависимости от присутствия других веществ
4) не растворимые неорганические соединения, способные изменять окраску раствора
5) растворимые неорганические и органические соединения, способные изменять окраску раствора под действием света
006. ЕСЛИ НА ТИТРОВАНИЕ 0,4217Г. БУРЫ ИЗРАСХОДОВАНО 17, 5МЛ HCL, ТО НОРМАЛЬНОСТЬ ЭТОЙ КИСЛОТЫ РАВНА
1) 0,0074 н
2) 0,2500 н
3) 0,1267 н
4) 0,0125 н
5) 1,024 н
007. В АЛКАЛИМЕТРИИ В КАЧЕСТВЕ ТИТРАНТА ВЫСТУПАЕТ:
1) KOH
2) NaCl
3) KMnO4
4) AgNO3
5) HCl
Занятие №10
«ОКИСЛИТЕЛЬНО-ВОССТАНОВИТЕЛЬНОЕ ТИТРОАНИЕ. ПЕРМАНГАНАТОМЕТРИЯ»
Вопросы к занятию
1. Окислительно-восстановительное титрование. Классификация методов.
2. Окислительно-воостановительный потенциал, уравнение Нернста.
3. Метод перманганатометрии. Рабочие растворы.
4. Применение перманганатометрии в медицинской практике и фармации.






