ѕример 1. ѕри сгорании 6,048 г водорода в кислороде при 298,15 в стандартных услови€х выдел€етс€ 857,5 кƒж теплоты. ќпределить стандартную энтальпию образовани€ H2O(ж) при 298,15 .
–ешение. ¬одород сгорает в кислороде согласно уравнению:
H2 (ж) + 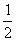 O2 (ж) = H2O(ж)
O2 (ж) = H2O(ж)
¬се вещества наход€тс€ в стандартных состо€ни€х, поэтому энтальпи€ сгорани€ 1 моль Ќ2 (2,016 г) и будет стандартной энтальпией образовани€ Ќ2ќ (ж) при 298,15 ; найдем эту величину:
6,048 Ц 857,5
2,016 Ц х, †††††††††††††††††††††††† откуда х = 258,8;
DHoбp.H2O(ж) = Ц 285,8 кƒж/мoль.
ѕример 2. Ёнтальпи€ какого процесса отвечает термину Устандартна€ энтальпи€ образовани€ ЌNO3 при 298,15 Ф:
а) 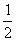 H2(г) +
H2(г) + 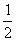 N2(г) +
N2(г) +  O2(г) = HNO3(ж);
O2(г) = HNO3(ж);
б) 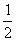 H2(г) +
H2(г) + 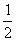 N2(г) + O3(г) = HNO3(ж);
N2(г) + O3(г) = HNO3(ж);
в) 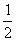 H2(г) +
H2(г) + 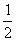 N2(г) +
N2(г) + 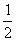 O2(г) = HNO3(ж);
O2(г) = HNO3(ж);
г) 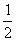 H2(г) +
H2(г) + 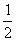 N2(г) +
N2(г) +  O2(г) = HNO3(к);
O2(г) = HNO3(к);
д.) 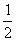 N2O5(к) +
N2O5(к) + 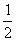 H2O(ж) = HNO3(ж)?
H2O(ж) = HNO3(ж)?
–ешение. ѕри 298,15 стандартными состо€ни€ми дл€ простых веществ будут H2 (г), O2 (г) и N2 (г), а дл€ азотной кислоты - жидкость (хот€ можно говорить и о стандартных термодинамических функци€х, например, и HNO3 (г) при этой температуре). ѕоэтому лишь процесс "в" строго отвечает названному термину.
ѕример 3. ќпределить величину стандартной энтальпии образовани€ C3H8 (г)*, если стандартные энтальпии образовани€ CO2(г) и Ќ2ќ (ж) равны соответственно Ц393,5 и Ц285,8 кƒж/моль, а стандартна€ энтальпи€ сгорани€ —3Ќ8(г) составл€ет Ц2219,9 кƒж/моль.
–ешение. «апишем уравнение процесса, соответствующего термину "стандартна€ энтальпи€ образовани€ —3Ќ8(г)":
3C(к, графит) + 4H2(г) = C3H8(г);  .
.
ƒл€ определени€ DЌ воспользуемс€ 2-м следствием из закона √есса (соотношение 1.12):
 =3DH0сгор— (к, графит) + 4DH0сгорH2(г) Ц DH0сгор.C3H8 (г).
=3DH0сгор— (к, графит) + 4DH0сгорH2(г) Ц DH0сгор.C3H8 (г).
ƒалее обратим внимание, что стандартные энтальпии образовани€ —ќ2(г) и Ќ2O(ж) есть не что иное, как стандартные энтальпии сгорани€ графита
и водорода. —ледовательно:
 =3DH0обр—O2(г) + 4DH0обрH2O(ж) Ц DH0сгорC3H8 (г) =
=3DH0обр—O2(г) + 4DH0обрH2O(ж) Ц DH0сгорC3H8 (г) =
= 3×(Ц393,5) + 4×(Ц285,8) Ц (Ц2219,9) = Ц 103,8 кƒж/моль.
ѕример 4. Ёнтальпи€ растворени€ Na2SO4 (к) в воде с образованием раствора состава Na2SO4 (к) × 400 H2O составл€ет Ц1,5 кƒж/моль, а энтальпи€ присоединени€ воды к Na2SO4 (к)c образованием Na2SO4 ×10H2O(к) равна Ц82,0 кƒж/моль. ќпределить энтальпию растворени€ Na2SO4×10H2O(к) в воде
†с образованием раствора состава Na2SO4 ×400 H2O.
–ешение. «апишем условие задачи в виде термохимических уравнений:
а) Na2SO4(к) + 400 H2O (ж) =Na2SO4 (р-р, Na2SO4×400H2O); DH1;
б) Na2SO4(к) + 10 H2O (ж) = Na2SO4×10H2O(к); DH2;
в) Na2SO4×10H2O(к) + 390 H2O (ж) = Na2SO4 (р-р, Na2SO4×400H2O); DH’.
ак видно, искомое (третье) уравнение получаетс€ путем вычитани€ второго термохимического уравнени€ из первого, следовательно:
DH’ = DH1 Ц DH2 = 1,5 Ц (Ц82,0) = 80,5 кƒж.
ѕример 5. Ќа основе справочных данных определить, в каком случае в стандартных услови€х выделитс€ больше теплоты Ц при сгорании 2 моль CH4 или 1 моль C2H6?
|
|
|
–ешение. “ермохимические уравнени€ процессов горени€:
—H4 (г) + 2O2 (г) = CO2 (г) + 2H2O (ж);
—2H6 (г) + 3,5O2 (г) = 2CO2 (г) + 3H2O (ж).
¬ыписываем из справочных данных стандартные энтальпии образовани€ —ќ2(г), H2O(ж), CH4(г) и C2H6(г), пользу€сь 1-м следствием из закона √есса, вычисл€ем энтальпии написанных процессов, то есть стандартные энтальпии сгорани€ CH4(г) и C2H6(г):
DH0сгор—H4 (г) = DH0обрCO2(г) + 2DH0обрH2O(ж) Ц DH0обр—H4 (г) Ц 2DH0обрO2(г) =
= Ц 393,5 + 2(Ц285,8) Ц (Ц74,8) Ц 2×0 = Ц890,3 кƒж;
DH0сгор—2H6 (г) = 2DH0обрCO2(г) + 3DH0обрH2O(ж) ЦDH0обр—2H6 (г) Ц
Ц 3,5DH0обрO2(г) = 2(Ц393,5) + 3(Ц285,8) Ц (Ц84,7) Ц 3,5×0 = Ц 1273,9 кƒж.
††† »з полученных величин следует, что при сгорании 2 моль CH4(г) выдел€етс€ теплоты больше, чем при сгорании 1 моль C2H6(г).
ѕример 6. ¬ычислить среднюю энтальпию св€зи —ЦЌ в метане на основе следующих термохимических данных:
1) C (к,графит) + 2H2 (г) = CH4 (г),  = Ц74,8 кƒж;
= Ц74,8 кƒж;
2) H2 (г) = 2H (г),  †= 436,0 кƒж;
†= 436,0 кƒж;
3) C(к,графит) = C(г),  =715,1 кƒж.
=715,1 кƒж.
–ешение. —огласно определению средн€€ энтальпи€ св€зи —ЦЌ в —Ќ4 Ц это 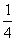 ††часть энтальпии следующего процесса:
††часть энтальпии следующего процесса:
CH4(г) = C (г) + 4H (г); 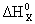 = 4
= 4  CЦH.
CЦH.
Ёто уравнение может быть получено путем сложени€ третьего уравнени€ с удвоенным вторым уравнением и вычитанием из этой суммы первого уравнени€, следовательно:
 = 715,1 + 2(436,0)Ц(Ц74,8)=1661,9 кƒж.
= 715,1 + 2(436,0)Ц(Ц74,8)=1661,9 кƒж.
ќтсюда:
DH0ср.—ЦH =  =415,5 кƒж.
=415,5 кƒж.
ѕример 7. ќпределить энтальпию св€зи —Ц— в этане на основе следующих данных:
DЌобр—2Ќ6(г) = Ц84,7 кƒж/моль; DЌсублимации—(к,графит) = 715,1 кƒж/моль;
DЌдисЌ2на атомы= 436,0 кƒж;DЌср.—ЦЌ = 415,5 кƒж/моль св€зи.
–ешение. «апишем исходные данные в виде термохимических уравнений:
1) C (к,графит) + 3H2(г) = C2H6(г),  обрC2H6(г);
обрC2H6(г);
2) C (к,графит) = C (г),  сублимацииC (к,графит);
сублимацииC (к,графит);
3) H2 (г) = 2H (г),  диссоциацииH2на атомы.
диссоциацииH2на атомы.
ќсновой дл€ нахождени€ искомой величины €вл€етс€ термохимическое уравнение
C2H6 (г) = 2C (г) + 6H (г); 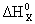
ѕри распаде на атомы 1 моль —2Ќ6(г) будет разорвано 1 моль св€зей —Ц— и 6 моль св€зей —ЦЌ, следовательно:
 =
=  + 6
+ 6  .
.
— другой стороны,  †может быть найдена на основе исходных термохимических данных, а именно Ц искомое уравнение получаетс€ путем сложени€ удвоенного второго уравнени€ с утроенным третьим уравнением и вычитани€ из полученной суммы первого уравнени€, отсюда:
†может быть найдена на основе исходных термохимических данных, а именно Ц искомое уравнение получаетс€ путем сложени€ удвоенного второго уравнени€ с утроенным третьим уравнением и вычитани€ из полученной суммы первого уравнени€, отсюда:
 =2
=2  +3
+3  Ц
Ц  =2×715,1 + 3(436,0) Ц (Ц84,7) = 2822,9 кƒж.
=2×715,1 + 3(436,0) Ц (Ц84,7) = 2822,9 кƒж.
Ќаходим теперь энтальпию св€зи —Ц— в этане:
 = 2822,9 Ц 6(415,5) = 329,9 кƒж/моль св€зи.
= 2822,9 Ц 6(415,5) = 329,9 кƒж/моль св€зи.
Ёнтропи€
ѕример 1. ¬ каком из следующих процессов следует ожидать наибольшего и наименьшего изменени€ энтропии:
1) H2(г) + 0,5O2 (г) = H2O(ж);
2) H2 (г) + 0,5 O2 (г) = H2O (г);
3) C2H6 (г) + 3,5O2 (г) = 2 CO2 (г) + 3 H2O (г);
4) KClO3 (к) = KCl (к) + 1,5 O2 (г).
–ешение. «нак и величину DS процесса можно оценить, не производ€ вычислений, если вспомнить, что энтропи€ газа существенно больше энтропии жидкости и тем более Ц кристаллического вещества. ѕоэтому, определ€€, насколько число молей газообразных веществ в правой части уравнени€ больше числа молей газообразных веществ в левой части уравнени€, приходим к выводу, что наибольша€ положительна€ энтропи€ характеризует четвертый процесс. Ёнтропии первого и второго процессов отрицательны, наибольша€ отрицательна€ энтропи€ соответствует первому процессу.
|
|
|
ѕример 2. ѕользу€сь справочными данными, определить стандартную энтропию образовани€ K2Cr2O7(к).
–ешение. »скома€ величина Ц это DSо следующего процесса:
2K(к) + 2Cr(к) + 3,5 O2(г) = K2Cr2O7(к).
Ёта величина может быть найдена на основе соотношени€ (1.18):
DS0 = S0K2Cr2O7 (к) Ц 2S0 K (к) Ц 2S0 Cr (к) Ц 3,5S0 02 (г) =
= 291 Ц 2×64,48 Ц 2×23,6 Ц 3,5×205,0 = Ц 603,1 ƒж/ .
ѕример «. ѕользу€сь справочными данными, определить возможность самопроизвольного протекани€ при 298,15 следующих процессов в изолированных системах:
1) KClO3 (к) = KCl (к) +1,5 O2 (г);
2) 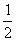 N2 (г) + 2H2 (г) +
N2 (г) + 2H2 (г) + 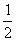 Cl2 (г) = NH4Cl (к).
Cl2 (г) = NH4Cl (к).
–ешение. ƒл€ изолированных систем критерием самопроизвольного течени€ процесса €вл€етс€ положительное значение энтропии. ѕоэтому, пользу€сь справочными данными, вычисл€ем дл€ указанных процессов
 †= S0KCl(к) + 1,5S0O2 (г) Ц S0KClO3 (к) =
†= S0KCl(к) + 1,5S0O2 (г) Ц S0KClO3 (к) =
= 82,6 + 1,5×205,0 Ц 143,0 = 247,1 кƒж/ ;
 = S0NH4Cl(к) Ц 0,5S0N2 (г) Ц 2S0 H2 (г) Ц 0,5S0Cl2 (г) =
= S0NH4Cl(к) Ц 0,5S0N2 (г) Ц 2S0 H2 (г) Ц 0,5S0Cl2 (г) =
= 95,8 Ц 0,5×191,5 Ц 2×130,5 Ц 0,5×223,0 = Ц 372,5 ƒж/ .
¬ результате делаем вывод, что в изолированной системе первый процесс разрешен в самопроизвольном течении, а второй - запрещен.
Ёнерги€ √иббса






