јналогичные по форме соотношени€ можно получить при расчЄте равновеси€ диссоциации слабого основани€, например, гидроксида аммони€ NH4OH:
NH4OH Û NH4+ + OH-
aї 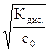 и [OH-] = [NH4+] = a × с 0 =
и [OH-] = [NH4+] = a × с 0 = 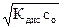 , (5.10)
, (5.10)
где с 0 Ц мол€рна€ концентраци€ аммиака в его растворе, а дис . Ц справочное значение константы диссоциации гидроксида аммони€; константу диссоциации основани€ обозначают b (от английского слова base Ц основание).
ƒиссоциаци€ слабого электролита Ц обратимый процесс, в котором положение равновеси€ можно смещать влево (подавление диссоциации) за счЄт добавлени€ в раствор сильных электролитов, при диссоциации которых образуетс€ ион, одноимЄнный с ионом слабого электролита. —мещение равновеси€ вправо достигаетс€ за счЄт св€зывани€ продуктов диссоциации в менее диссоциирующие соединени€. Ќапример, диссоциацию уксусной кислоты можно подавить за счЄт добавлени€ в еЄ раствор ацетата натри€ или сильной кислоты, поставл€ющей в раствор Ќ+.
ќсобый интерес представл€ет диссоциаци€ воды Ц слабого амфотерного электролита:
H2O Û H+ + OH-,
где равновесные концентрации ионов св€заны друг с другом через константу диссоциации:
дис . = 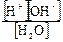 ;
;
ее величина может быть вычислена при любой температуре на основании данных измерени€ электропроводности чистой воды. ѕри 22 0— дис . равна 1,8Ј10-16; пренебрега€ ничтожно малой степенью диссоциации воды, концентрацию молекул воды как в чистой воде, так и в разбавленных водных растворах можно считать величиной посто€нной:
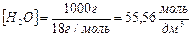 .
.
“огда выражение дл€ константы диссоциации можно переписать:
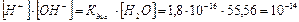
произведение [Ќ+]Ј[ќЌ-]=10-14 называют ионным произведением воды и обозначают w: [OH-]Ј[H+]= w =10-14 (5.11)
ƒл€ чистой воды и дл€ разбавленных водных растворов любых веществ ионное произведение воды w при данной температуре €вл€етс€ величиной посто€нной и равно 10-14. — повышением температуры w возрастает, с понижением Ц уменьшаетс€. “ак, например, при 100 0— ионное произведение воды w =10-12.
»спользу€ величину ионного произведени€ воды w и известную концентрацию одного из ионов воды, исход€ из формулы (5.11), можно вычислить концентрацию ионов Ќ+ и ќЌ- в любом водном растворе:
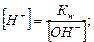
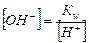 .
.
¬ чистой воде из услови€ электронейтральности:
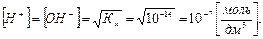
–астворы, в которых [Ќ+]=[ќЌ-] =10-7 моль/дм3, называютс€ нейтральными.
≈сли [H+] > [OH-] > 10-7 моль/дм3 (>10-6...10-1моль/дм3) растворы называютс€ кислыми, если [H+] < [OH-] < 10-7моль/дм3 Ц щелочными растворами (<10-8...10-14 моль/дм3).
Ќа практике кислотность раствора в водных средах прин€то характеризовать не мол€рной концентрацией ионов водорода или гидроксила, а безразмерной величиной Ц водородным показателем рЌ.
¬одородный показатель рЌ Ц количественна€ характеристика кислотности среды Ц величина, равна€ отрицательному дес€тичному логарифму равновесной концентрации ионов водорода в растворе:
рЌ = -lg[H+].
јналогично гидроксильный показатель рќЌ:
рќЌ = -lg[OH-].
¬ любом водном растворе [OH-]Ј[H+]= w=10-14 (при 220—). Ћогарифмиру€ это выражение и учитыва€ выражени€ дл€ рЌ и рќЌ, получаем:
|
|
|
рЌ + рќЌ = 14 (5.12)
¬ нейтральной среде рЌ = рќЌ = 7,0; в кислой среде рЌ < 7,0; в щелочной Ц рЌ > 7,0. ¬ разбавленных водных растворах различных веществ величина рЌ мен€етс€ от 0 до 14.
ќЅ”„јёў»≈ «јƒј„»
1.¬ычислить ионную силу в растворе 0,1 моль/дм3 сульфата натри€.
–ешение:
Na2SO4→2Na+ + SO42-
c [ 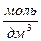 ]: 0,1 0,2 0,1
]: 0,1 0,2 0,1
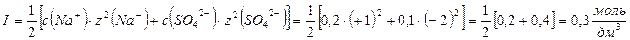 ќтвет: ионна€ сила 0,1 моль/дм3 Na2SO4 равна 0,3 моль/дм3.
ќтвет: ионна€ сила 0,1 моль/дм3 Na2SO4 равна 0,3 моль/дм3.
2. ¬ычислить концентрацию ионов ќЌ-, рќЌ и рЌ дл€ 0,1 моль/дм3 раствора аммиака ( b =1,76Ј10-5).
–ешение: NH3 + H2O Û NH4OH Û NH4+ + OH-
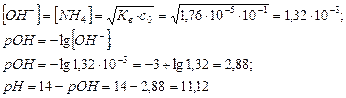
ќтвет: [ќЌ-]=1,32Ј10-3; рќЌ=2,88; рЌ=11,12.
3. онстанта диссоциации уксусной кислоты равна 1,75 10-5. ќпределите массу ацетата натри€, которую необходимо добавить к 300 см3 0,1 моль/дм3 раствора уксусной кислоты, чтобы понизить концентрацию ионов водорода в растворе в 100 раз.
ЌайдЄм равновесную концентрацию ионов водорода в 0,1 моль/дм3 растворе CH3COOH:
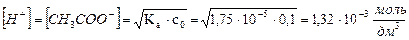 .
.
ѕри добавлении CH3COONa концентраци€ ионов H+ должна быть в 100 раз меньше (диссоциаци€ частично подавлена), т.е. 1,32 ∙ 10-5 моль/дм3. ѕусть необходимо внести в 1 дм3 исходного раствора кислоты х моль соли, что соответствует добавке x моль ацетат- ионов, тогда равновесные концентрации равны: [H+] = 1,32×10-5 моль/дм3
[CH3COO-] = (x + 1,32 ∙ 10-5) моль/дм3ї x моль/дм3
[CH3COOH] = с 0 Ц [H+] = 0.1 Ц 1.32× 10-5ї 0,1 моль/дм3
и, подставив значени€ равновесных концентраций в выражение дл€ константы диссоциации уксусной кислоты:
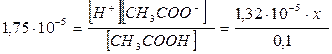
находим x = 0,133 моль, что соответствует концентрации соли с (CH3COONa) = 0,133 моль/дм3.
ћасса добавки ацетата натри€ к 300 см3 раствора равна:
m (CH3COONa) = с (CH3COONa) × V р-р × M (CH3COONa) = 0,133 × 0,3 × 82 = 3,27 г
ќтвет: m (CH3COONa) = 3,27 г.






