ѕиррал ‘уран “иофен


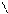
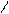



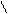
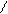



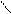

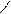


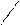
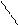

 ......
......
 N O S
N O S
H
Ёти соед.,€в-с€ аромат.,т.к. отвечают всем критери€м ароматически:
1. имеют плоский замкн. δ-скелет.
2. налич.ед.сопр€ж. π-электр.облака, вклдючающее неподеленную пару е.
3. выполн.правило ’юккел€
Hn+2=6; n-целое число-правило выполн€етс€.
Ёто π-избыточные системы, степень ароматичности ниже, чем у бензола.
Ёлектроотрицат.у S меньше, чем у N и ќ.
ƒол€ участие непод.пары S в образ-ии аромат.секстета в молекуле тиофена больше, чем атом N в пирроле и ќ-в фуране.
— учетом стойкости к кислотам, окислител€м степени легкости элктрофильного замещени€ по ароматичности распологаютс€ в р€д.
“иофен > пиррол > фуран.
пособы получени€:
** циклизаци€ 1,4 Ц дикарбонильных соединении.





 P2S5 R R
P2S5 R R

 —H2-CH2 S
—H2-CH2 S
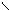
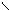
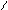








 R-C C-R CH-CH
R-C C-R CH-CH
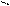


 O O R-C C-R NH3 R R
O O R-C C-R NH3 R R


 OH OH N
OH OH N
H


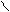
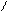
 H2SO4, to R
H2SO4, to R
 O
O
** взаимные превращени€ (цикл реак.ёрьева)
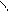
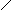








 H2O
H2O
 N NH3 O
N NH3 O
H



 H2O Al2O3 H2S
H2O Al2O3 H2S
NH3 t=450o H2O
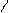
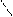
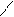

 |  |
S
—пецифические способы получени€:
**нагрев.аммонильной соли слизевой кислоты.
 ѕиррол
ѕиррол
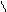
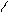




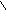
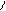 HO-CH-CH-OH to
HO-CH-CH-OH to
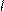

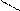


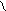
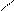 HC CH COONH≠4 -H2O N
HC CH COONH≠4 -H2O N


 H4NOOH OH OH - CO2 H
H4NOOH OH OH - CO2 H
 -NH3
-NH3
** перегонка сукцин имида с Zn-ой пылью.
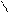
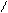

 H2C CH2
H2C CH2

 O= =O Zn
O= =O Zn
 NH N
NH N
H

 ** ацетилен + NH3
** ацетилен + NH3
 HCaCH+NH3 300o-400o
HCaCH+NH3 300o-400o
 Al2O3 N
Al2O3 N
H
 |

 ** HCaCH+2HC=O to [HOCH2ЦCaCЦCH2OH]+ ЦNH3, toЃ
** HCaCH+2HC=O to [HOCH2ЦCaCЦCH2OH]+ ЦNH3, toЃ
 H N
H N
H
‘уран
** суха€ перегонка слизевой к-ты.


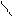
 HO-CH-CH-OH
HO-CH-CH-OH


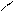
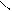
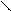

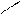

 HOOC-CH CH ЦCOONH HOOC - - COOH - COOH to
HOOC-CH CH ЦCOONH HOOC - - COOH - COOH to
OH OH O O -CO2
ƒегидрослизева€ пирослиз.
-та к-та.
** нагревание альдопентоз с водоотним.св-ми.

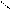
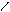
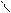








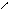
 HO-CH-CH-OH -C=O [O] -COOH to =
HO-CH-CH-OH -C=O [O] -COOH to =

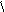

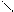
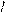 HCH CH C=O O H O -CO2 O
HCH CH C=O O H O -CO2 O
OH OH H
√иофен
** ѕарафазна€ циклизаци€ бутана с серой
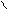
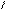
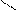
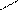
 —4H10 + HS 700oC
—4H10 + HS 700oC
-3H2S S
 ** по „ичибабину
** по „ичибабину
2HC aCH+H2S 400o-450oC
 H2O3 S
H2O3 S
 |
** 2CH3C=O + H2S 300o
 H Al2O≠3 S
H Al2O≠3 S
’имические свойства:
** кислотно-основные
“.к.неподел.пара е- гетероатомов вт€нуты в общи секстед, поэтому гетероциклы не про€вл€ют основных св-в.
ѕиррол и фуран ацидофобные в кисл.среде тер€ют ароматичность и пол€ризуютс€ с обр.смол.
+
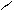

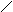
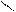
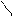
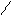



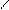
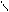
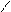
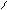
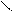
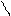
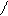

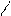

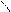 .. +H+ H H полимер-и€
.. +H+ H H полимер-и€
’ X H X+ H
’=0; NH
ѕиррол облад.слаб.кисл.св-ами реагирует с сильн.основ.с Me, MeOH и др.
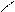
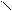
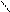
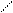

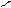
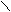



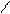
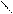
 N + KOH (тв) N пирролат кали€
N + KOH (тв) N пирролат кали€
H -H2O K
 |  |


 N + NaNH2 N- ≠ пирролнатрии
N + NaNH2 N- ≠ пирролнатрии
H -NH3 Na+
 | |||
 | |||
N +CH3MgBr N- пирролмагний бромид
 H -CH4 MgBr +
H -CH4 MgBr +
** реак.электрофильного замещ.
яв-сь ѕ-избыт.ароматич.системами они легче чем бензол вступают в реак. S≈
ѕо активности они располаг. в р€д:
|
|
|
ѕиррол> фуран > тиофен

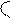
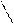
 (B) 4 3(B) реак. S≈ идут преимущественно в полож. α(2)
(B) 4 3(B) реак. S≈ идут преимущественно в полож. α(2)
 |  | ||
 (α)5 (б-)2(α)
(α)5 (б-)2(α)
..
’
√алогенирование
“иофен можно: Hal Ц (Cl2 или Br2) на холоде

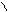
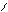
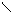
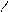


 Cl Cl
Cl Cl
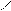
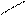
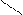 S + Cl2 S -Cl +Cl≠2 Cl S - Cl + 2Cl Cl Cl
S + Cl2 S -Cl +Cl≠2 Cl S - Cl + 2Cl Cl Cl

 -HCl -HCl -2HCl S
-HCl -HCl -2HCl S
— J2 р-и€ идет труднее, в присутств. Hgo.
ѕирроз с Hal Цреагир.легко с образованием сразу тетрагалогенпиррала.
ƒл€ полдг.моно/ди исп-с€ сульфохлорид
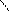
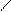
 Cl Cl
Cl Cl
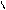
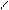
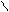
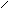
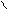

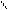
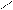
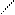
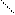
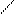

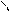
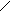

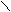
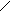
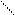



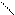

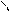
 SO2Cl2 - Cl SOCl2 Cl - Cl SOCl2 Cl- - Cl
SO2Cl2 - Cl SOCl2 Cl - Cl SOCl2 Cl- - Cl






 N N N N
N N N N
H H H H
√алогенир.фурана происх.сложно, т.к. с замещ.водорода на Hal Ц идет 2,5 Ц присоединение



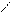
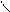
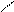

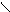


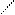
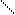
 O +Cl2 Cl - O -Cl
O +Cl2 Cl - O -Cl
Ќитрование
”читыва€ ацедовобность пиррола и фурана дл€ нитр.исп.ацетилнитрат.
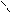
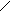


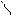
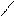
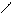
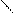 ..
..



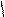 ’ + —–3-—=O X -NO2 +CH3COOH
’ + —–3-—=O X -NO2 +CH3COOH
ONO2
X=0; NH
“иофен можно нитрировать азотной к-ой в м€гк.усл.
—ульфирование
¬ виду ацедофобности пиррола и фурана дл€ их сульфировани€ исп.перидиксульфотриоксид.(“ерентьев).
 | |||||||
 | |||||||
 |  | ||||||


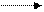 + - SO3H +
+ - SO3H +
 | |||
 | |||
 ’ N X
’ N X
SO3H
X=0; NH
“иофен легко сульфир. H2SO4 (k)
јцилирование
»спользуют ангидриды к-т в присутств.к-т Ћьюиса (ZnCl2, SnCl4)
 |  |
+ (Cu3CO)2O SnCl4 -COCH3 + CH3COOH

X S
“иофен можно ацилир.хлорангидр.в присут. AlCl3
** ¬осстановление

 CH2 CH2
CH2 CH2
+2H2 to, 140-150 атм. Ќ2—- CH2
 ат(ћ)
ат(ћ)
O O тетрагидрофуран
‘уран

 H2C CH2
H2C CH2
 + 2H2 to, 2-4 атм.Pd H2C CH2
+ 2H2 to, 2-4 атм.Pd H2C CH2

S S тетрагидротиофен
“иофен

 N
N
H
 пирролин
пирролин

 [H] CH2 CH2
[H] CH2 CH2
 (Zn+CH3COOH) +H2, Pd/Pt H2C CH2
(Zn+CH3COOH) +H2, Pd/Pt H2C CH2

 N N
N N
H H
** ќкисление
ѕиррол и фуран очень чувств.к действ.окис-ей.
ќкисл-с€ даже O2 (возд), при этом происходит разрыв кольца.
¬ присут.катал.об-с€
 |  |
 + O2 V2O5, to O= = O
+ O2 V2O5, to O= = O
O O малеиновый ангидрид
 |  |
 + O2 V2O5 O= = O
+ O2 V2O5 O= = O

 O N
O N
H малеинимид
—пецифич.реакции



 CH3J
CH3J
 ’олод -KJ CH3J, to - CH3 + KJ
’олод -KJ CH3J, to - CH3 + KJ
јлк-ие
 N N
N N
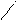
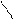
 CH3 H
CH3 H


 |  |
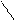
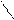
 CH3COCl N- CH3COCl, to - COCH3 + KCl
CH3COCl N- CH3COCl, to - COCH3 + KCl
 ’олод K+ ацил-ие
’олод K+ ацил-ие
 N N
N N
CH3-C=O H
ѕиррол легко вступает в реак. азосочетани€.

 NaN
NaN
 | |||||||
 |  |  | |||||
 + Cl - N=N -
+ Cl - N=N -
 -HCl
-HCl
N N
H H бензолазопиррол
‘уран занима€ промежуток полож.м/у аромат.соед. и 1,3 Ц сопр€ж.диенами вступает в характерную дл€ сопр€ж.диенов реак ƒисса јлодера.



 O 6 O
O 6 O




 5 1
5 1

 O + O O O
O + O O O




 4 2
4 2
O 3 O
ћалеиновый ангидрид 1.2.3.6-тетрагидро-3-6-эпоксифталевый ангидрид
ѕроизводные
**
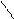

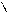 -C=O
-C=O
H
O фурфурол
1) дегидр.пентоз (лекци€ моносах)
2) формирование фурана


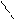
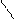

 1)HCN,HCl -C=O
1)HCN,HCl -C=O
2)HOH H
O O
¬ хим-ом поведении сходен с бензоальгедридом
2 реакци€ центра:
1) альдегидо группа
|
|
|
2) аромат. ѕ-содерж.кольцо.
**


 O2N - CH=N=NH-C-NH семикарбозон-5-нитрофурфурола
O2N - CH=N=NH-C-NH семикарбозон-5-нитрофурфурола
O фурацилин
O
**
»ндал (бензпиррол)
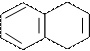
 N
N
H

 H
H


 HC
HC



 + + H2
+ + H2

 N-H HC
N-H HC
 H N
H N
H
¬ хим-ом повед.харак-ен пиррол, но в отл.от него реак SE идут в β-положении.
»ндол входит:
1)в состав амк-триптофана
2) в состав биогенных аминов-серотонина, триптамина
3)в состав β-индальуксусной к-ты(гетероауксин)
4)в состав противовосп.препарата индомитацина
5)в состав красителей индиго.






