ОГЛАВЛЕНИЕ
| Таблица определения вариантов письменных домашних заданий............... | 3 | |
| Тема 1: | Введение: правила работы в химической лаборатории. Техника безопасности и оказание первой помощи. Номенклатура неорганических веществЛабораторная посуда, реактивы.................. | 4 |
| Тема 2: | Элементы термодинамики. Закон Гесса.......................................... | 8 |
| Тема 3: | Химическая кинетика. Химическое равновесие............................. | 13 |
| Тема 4: | Растворы, способы выражения состава растворов. Приготовление растворов с заданным содержанием растворенного вещества..……………..….............................................................................. | 18 |
| Тема 5: | Электролитическая диссоциация. Слабые и сильные электролиты. Водородный показатель............................................................... | 22 |
| Тема 6: | Равновесные процессы в растворах электролитов. Теории кислот и оснований. Произведение растворимости. Гидролиз солей. Буферные растворы............................................................... | 28 |
| Тема 7: | Окислительно-восстановительные реакции. Окислительно-восстановительные процессы, определение возможности самопроизвольного осуществления химической реакции. Мембранный потенциал.................. | 32 |
| Тема 8: | Комплексные соединения. Строение, классификация и устойчивость комплексных соединений........................................................ | 35 |
| Приложение........................................................................................................ | 40 | |
Таблица определения вариантов
Письменных домашних заданий для самостоятельной
внеаудиторной подготовки студентов направления "Лечебное дело" и "Педиатрия" к занятиям по дисциплине "Химия"
| № фамилии студента по журналу | № варианта письменного задания |
| 1, 11 | 1 |
| 2, 12 | 2 |
| 3, 13 | 3 |
| 4, 14 | 4 |
| 5, 15 | 5 |
| 6, 16 | 6 |
| 7, 17 | 7 |
| 8, 18 | 8 |
| 9, 19 | 9 |
| 10, 20 | 10 |
Тема 1.
Вводное занятие: правила работы в химической лаборатории.
Техника безопасности и оказание первой помощи.
Номенклатура неорганических веществ. Лабораторная посуда,
реактивы.
Вопросы для самостоятельной внеаудиторной подготовки к занятию:
1. Правила работы в химической лаборатории. Техника безопасности и оказание первой помощи.
Химические виды веществ. Физические и химические свойства веществ.
3. Что такое номенклатура химических соединений, из чего она складывается?
4. Что означает аббревиатура ИЮПАК?
5. Чем отличается адаптированный вариант номенклатуры неорганических соединений от варианта ИЮПАК?
6. Как в соответствие с принципами адаптированного варианта номенклатуры ИЮПАК образуются названия различных классов неорганических соединений, ионов?
Изучив эти вопросы, выполните письменно следующие задания:
Вариант № 1
1. Назовите химические соединения; укажите, к какому классу они относятся: Fe2O3, Fe(OH)3, Fe2(SO4)3, Fe(HSO4)3, FeOHSO4, FeSO4∙7H2O, FeOCl, H2Cr2O7.
Назовите ионы: Fe3+, FeOH+, Fe(OH)  , HSiO3-.
, HSiO3-.
2. Напишите формулы химических соединений и ионов: фосфат гидроксомарганца(II), гидрокарбонат кальция, сульфат аммония, оксид хрома(VI), пентагидрат сульфата меди(II), ион дигидроксохрома(III), дигидрофосфат–ион.
Вариант № 2
1. Назовите химические соединения; укажите, к какому классу они относятся: MnO, MnSO4∙7H2O, (MnOH)2CO3, Mn(OH)2, HMnO4, Mn(HCO3)2, Mn3(PO4)2, SbOCl.
Назовите ионы: Sn2+, MgOH+, MnO 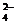 -, HPO
-, HPO 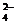 .
.
2. Напишите формулы химических соединений и ионов: оксид свинца(IV), гидроксид свинца(II), гептагидрат сульфата железа(II), гидроксид хрома (III), хлорид оксожелеза(III), ион гидроксомеди(II), гидросульфат-ион.
Вариант № 3
1. Назовите химические соединения; укажите, к какому классу они относятся: PbO2, Pb(OH)2, (PbOH)2SO4, Pb(MnO4)2, Pb(HCO3)2, Pb(NO3)2∙4H2O, H2MnO4, FeONO3.
Назовите ионы: Mn2+, MnOH+, MnO 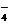 , HCO
, HCO  .
.
2. Напишите формулы химических соединений и ионов: оксид железа(III), гидроксид цинка, фосфат никеля(II), гексагидрат хлорида кальция, ион аммония, ион гидроксоалюминия, дихромат-ион.
Вариант № 4
1. Назовите химические соединения; укажите, к какому классу они относятся: Cl2O7, Co(OH)2, H4P2O7, NiSO4∙7H2O, Cr(OH)2NO3, Cr2(HPO4)3, ZnCl2, CrONO3.
Назовите ионы: Pb2+, Fe(OH)  , HSO
, HSO  , CrO
, CrO 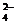 .
.
2. Напишите формулы химических соединений и ионов: перхлорат кальция, оксид свинца(II), кремниевая кислота, карбонат гидроксомарганца(II), гидрокарбонат магния, ион гидроксовисмута(III), дигидродифосфат-ион.
Вариант № 5
1. Назовите химические соединения; укажите, к какому классу они относятся: Cu2О, CuOH, CuSO4∙5H2O, (CuOH)2SO4, Cu(HCO3)2, HPO3, Cu3(PO4)2, MgO2.
Назовите ионы: Cu2+, CuOH+, HPO 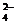 , H2PO
, H2PO 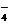 .
.
2. Напишите формулы химических соединений и ионов: дигидрофосфат кальция, сульфат гидроксохрома(II), гидроксид виcмута(III), декагидрат сульфата натрия, оксид фосфора(V), ион гидроксосвинца(II), ион хрома(III).
Вариант № 6
1. Назовите химические соединения; укажите, к какому классу они относятся: Al2O3, Ba(HCO3)2, CrONO3, HNO3, CaO2, ZnOHСl, FeCl3∙6H2O, Pb(OH)2.
Назовите ионы: Al3+, CrOH2+, HSO 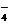 , Cl─.
, Cl─.
2. Напишите формулы химических соединений и ионов: гексагидрат нитрата железа(III), оксид виcмута(III), фосфат никеля(II), пероксид водорода, сульфат хрома (II), ион гидроксомарганца(II), нитрит-ион.
Вариант № 7
1. Назовите химические соединения; укажите, к какому классу они относятся: SnO2, SnOHCl, Pb(HSO4)2, HNO2, KI, Na2SO4∙10H2O, Ba(OH)2, SrO2.
Назовите ионы: Al(OH)  , Cr3+, BiOH2+, HCO
, Cr3+, BiOH2+, HCO  .
.
2. Напишите формулы химических соединений и ионов: пероксид калия, гидроксид висмута(III), хлороводородная кислота, дифосфат калия, оксид-ион, ион гидроксожелеза(III), гидросиликат-ион.
Вариант № 8
1. Назовите химические соединения; укажите, к какому классу они относятся: Na2O2, Cu(OH)2, (CoOH)2CO3, Ca(HCO3)2, H4P2O7, FeSO4∙7H2O, MnO2, CrOCl.
Назовите ионы: Li+, ZnOH+, ClO 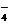 , H2PO
, H2PO 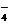 .
.
2. Напишите формулы химических соединений и ионов: оксид мышьяка(V), сернистая кислота, гидроксид никеля(II), дигидрат сульфата кальция; гидрокарбонат висмута(III), нитрат гидроксомеди(II), дифосфат-ион.
Вариант № 9
1. Назовите химические соединения; укажите, к какому классу они относятся: Fe2O3, Fe(OH)3, Fe2(SO4)3, Fe(HSO4)3, FeOHSO4, FeSO4∙7H2O, FeOCl, H2Cr2O7.
Назовите ионы: Sn2+, MgOH+, MnO 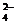 , HPO
, HPO 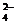 .
.
2. Напишите формулы химических соединений и ионов: перхлорат кальция, оксид свинца(II), кремниевая кислота, карбонат гидроксомарганца(II), гидрокарбонат магния, ион гидроксовисмута(III), дигидродифосфат-ион.
Вариант № 10
1. Назовите химические соединения; укажите, к какому классу они относятся: Al2O3, Ba(HCO3)2, CrONO3, HNO3, CaO2, ZnOHСl, FeCl3∙6H2O, Pb(OH)2.
Назовите ионы: Pb2+, Fe(OH)  , HSO
, HSO  , CrO
, CrO 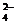 .
.
2. Напишите формулы химических соединений и ионов: дигидрофосфат кальция, сульфат гидроксохрома(II), гидроксид виcмута(III), декагидрат сульфата натрия, оксид фосфора(V), ион гидроксосвинца(II), ион хрома(III).
Образец выполнения письменных заданий:
1. Назовите химические соединения; укажите, к какому классу они относятся:
CdO, Cd(OH)2, H2CrO4, Cd(HSO3)2, CdCl2∙6H2O, Al(OH)2NO3, Fe(NO3)2, BiONO3.
Назовите ионы: Fe2+, PbOH+, PO  , HS ─.
, HS ─.
Ответ:
CdO – оксид кадмия, класс – оксиды, основный оксид;
Cd(OH)2 – гидроксид кадмия, класс - гидроксиды, основание;
H2CrO4 – хромовая кислота, класс - гидроксиды, оксокислота;
Cd(HSO3)2 – гидросульфит кадмия, класс - соли, кислая соль;
CdCl2∙6H2O – гексагидрат хлорида кальция, класс - соли, средняя соль, кристаллогидрат;
Al(OH)2NO3 – нитрат дигидроксоалюминия, класс - соли, основная соль;
Fe(NO3)2 – нитрат железа(III), класс - соли, средняя соль;
BiONO3 – нитрат оксовисмута(III), класс - соли, оксосоль;
Sr2+ – ион стронция;
PbOH+ – ион гидроксосвинца(II);
PO  – фосфат-ион;
– фосфат-ион;
HS─ – гидросульфид-ион.
2. Напишите формулы химических соединений и ионов: фосфат аммония; хлорат калия; дигидрат сульфата кальция; хлорид гидроксомарганца(II); гидрофосфат цинка; ион гидроксоалюминия; дифосфат-ион.
Ответ:
(NH4)3РO4; KClO3; CaSO4∙2H2O; MnOHCl; ZnHPO4; AlOH2+; P2O  .
.
Тема 2.
Элементы термодинамики. Закон Гесса.
Вопросы для самостоятельной внеаудиторной подготовки к занятию:
1. Что изучает термодинамика, химическая термодинамика?
2. Что называется системой? Что такое система открытая, закрытая, изолированная? Какая из этих систем используется в термодинамике в качестве модели?
3. Что называется состоянием системы?
4. Что называется процессом? Изобарный, изохорный и изотермический процесс.
5. Что изучает термохимия? Что называется тепловым эффектом химической реакции, каким символом он обозначается, в каких единицах измеряется? В чём отличие термохимических уравнений, от химических и термодинамических?
6. Закон Гесса. Следствия из закона Гесса.
7. Что такое термодинамические параметры системы? Какие термодинамические характеристики называются функциями состояния системы?
8. Что называется внутренней энергией системы? Каким символом она обозначается, в каких единицах измеряется?
9. Что называется энтальпией, каким символом она обозначается, в каких единицах измеряется? Чему равна стандартная энтальпия образования простого вещества?
10. Что называется энтропией, каким символом она обозначается, в каких единицах измеряется? Что называется микро- и макросостоянием системы? Энтропия как мера термодинамической вероятности системы?
11. Изобарно-изотермический потенциал системы (энергия Гиббса). Уравнение Гиббса. Анализ уравнения. Условия, при которых процесс идет самопроизвольно.
Вариант № 1
1. Стандартная энтальпия образования хлорбензола (С6Н5Сl(к)) равна (кДж/моль): -10,7. Напишите термохимические уравнения реакции, к которой относится этот тепловой эффект.
2. Для реакции: ВаСО3(тв) → ВаО(тв) + СО2 (газ)
∆Н0 (кДж/моль) -1202,0 -553,9 -394,0
∆S0 (Дж/моль-К) 112,2 70,5 213,8
Рассчитайте ∆Н0, ∆S0, ∆G0 область температур, при которых она будет протекать самопроизвольно. Зависимостью от температуры пренебречь. Ответ (кДж): 254; 0,17; 203; Т > 1477 К
3. Определите величину энтропии следующих реакций:
а) Br2(г) + H2(г) ↔2HBr(г)
б) Fe2O3(к) + 3H2(г) ↔ 2Fe(к) + 3H2O(г)
в) 2SO2(г) + O2(г) ↔ 2SO3(г)
Вариант № 2
1. Стандартная энтальпия сгорания ацетона (С3Н6О(ж)) равна -1832 кДж/моль. Напишите термохимическое уравнение реакции, к которой относится этот тепловой эффект.
2. Метаболизм глюкозы возможен в следующем направлении:
С6H12O6 (p.p) → СН3(СН2)2СООН(ж) + 2 СО2(г) + 2Н2 (г)
∆G0298 (кДж/моль) -916 -400 -384
∆Н0298 (кДж/моль) -1264 -532 -394
Используя данные задачи, рассчитайте ∆G0, ∆Н0 и ∆S0 для этого процесса. При каких температурах (высоких, низких, любых) возможно самопроизвольное протекание этого процесса?
Ответ: -252 кДж, -56 кДж; 658 Дж/К
3. Определите величину энтропии следующих реакций:
а) N2(г) + 3H2(г) ↔ 2NH3(г)
б) 2Fe(к) + 3H2O(г) ↔ Fe2O3(к) + 3H2(г)
в) 2CO(г) + O2(г) ↔ 2CO2(г)
Вариант № 3
1. При образовании 1,7 г аммиака из азота и водорода выделилось 4,62 кДж тепла. Рассчитайте стандартную энтальпию образования аммиака.
Ответ: 46,2 кДж/моль.
2. Рассчитайте ∆G0 для следующего процесса: креатинфосфат + АДФ ↔ креатин + АТФ, если известно, что для реакций: креатинфосфат + Н2О ↔ креатин + фосфат, ∆G0= -43 кДж/моль; АТФ + Н2О ↔ АДФ + фосфат, ∆G0 = -30,5 кДж/моль.
Ответ: -12,5 кДж/моль.
3. Определите величину энтропии следующих реакций:
а) 2NO(г) + O2(г) ↔ 2NO2(г)
б) СS2(г) + 2H2S(г) ↔ CH4(г) + 4S(т)
в) 2NH3(г) ↔N2(г) + 3H2(г)
Вариант № 4
1. При сгорании 2,24 л (н.у.) пропана выделилось 216 кДж в стандартных условиях. Рассчитайте стандартную энтальпию сгорания пропана.
Ответ: -2160 кДж/моль.
2. Вычислите ∆G0 процесса: АТФ + аланин + глицин → АДФ + фосфат + аланилглицин, если известно, что для реакций: а) АТФ + Н2О → АДФ + фосфат; ∆G0 = -30,5 кДж/моль; б) аланин + глицин → аланилглицин + Н2О; ∆G0 = 17,2 кДж/моль.
Ответ: -13,3 кДж/моль.
3. Определите величину энтропии следующих реакций:
а) NO(г) + NO2(г) + O2(г) ↔2N2O5(т)
б) CO(г) + Cl2(г) ↔СОCl2(г)
в) Cl2(г) + H2(г) ↔ 2HCl(г)
Вариант № 5
1. Рассчитайте тепловой эффект реакции С2Н2(газ) + 2 Н2(газ) → С2Н6(газ) двумя способами: а) с использованием стандартных энтальпий образования веществ (данные таблицы); б) с использованием стандартных энтальпий сгорания веществ.
Ответ: -312 и -314 (в кДж).
2. При 37°С и физиологическом значении pH величины ∆Н0 и ∆G0 для гидролиза АТФ равны -20,0; -29,3 кДж/моль, соответственно. Вычислите величину ∆S0 для тех же условий.
Ответ: 30 Дж/моль К.
3. Определите величину энтропии следующих реакций:
а) 2CO(г) ↔CO2(г) + C(к)
б) 2NO(г) + Cl2(г) ↔2NOCl (г)
в) 2SO2(г) + О2(г) ↔2SО3(г)
Вариант № 6
1. Энергетическую ценность топлива и пищевого продукта принято оценивать его теплотой сгорания (калорийностью) и выражать в кДж/г (ккал/г). Вычислите калорийность (в кДж/г) жира тристеарина (С57Н110О6, М = 890г/моль), если энтальпия сгорания жира равна -37760кДж/моль.
Ответ: 42,4 кДж/г.
2. Рассчитайте стандартное значение энергии Гиббса при 25°С реакции гидратации этилена: С2Н4(газ) + Н2О(ж) → С2Н5ОН(Ж), если для нее ∆Н0298 = -44 кДж и ∆S0298 = 127 Дж/К. Оцените роль энтальпийного и энтропийного факторов для этого процесса.
Ответ: -6,2 кДж.
3. Определите величину энтропии следующих реакций:
а) CO2(г) + C(к) ↔2CO(г)
б) 2Н2(г) + O2(г) ↔2Н2O(г)
в) 3H2(г) + N2(г) ↔ 2NH3(г)
Вариант № 7
1. Энергетическую ценность топлива и пищевого продукта принято оценивать его теплотой сгорания (калорийностью) и выражать в кДж/г (ккал/г). Вычислите калорийность (в кДж/г): сахарозы (С12Н22О11, М = 342 г/моль), если энтальпия сгорания сахарозы равна -5653 кДж/моль.
Ответ: 16,5 кДж/г.
2. Используя табличные данные, рассчитайте стандартное изменение энергии Гиббса реакции фотосинтеза глюкозы 6СО2(газ) + 6Н2О(ж) → С6H12O6(тв) + 6О2(газ); Возможно ли самопроизвольное протекание каждой из реакций?
Ответ (кДж): 2871.
3. Определите величину энтропии следующих реакций:
а) 2NO(г) + SO2(г) ↔ N2O(г) + SO3(г)
б) Br2(г) + H2(г) ↔2HBr(г)
в) С(тв) + 2Н2(г) ↔СН4(г)
Вариант № 8
1. Рассчитайте стандартную энтальпию реакции сгорания ацетилена (С2Н2), исходя из стандартных энтальпий образования веществ. Какой объем ацетилена сгорел, если в результате реакции выделилось 260 кДж теплоты?
Ответ: -1300 кДж/моль, 4.48 л.
2. Используя табличные данные, рассчитайте стандартное изменение энергии Гиббса реакции гликолиза: С6Н12О6(р-р) → 2С3Н6О3(р-р). Возможно ли самопроизвольное протекание реакций?
Ответ (кДж): -161.
3. Определите величину энтропии следующих реакций:
а) NH3(г) + HCl(ж) ↔NH4Cl(ж)
б) Fe3O4(к) + 4Н2(г) ↔3Fe(к) + 4Н2О(г)
в) 2NO(г) + O2(г) ↔2NO2(г)
Вариант № 9
1. Для реакции С6Н12О6(тв) + 6 О2(газ) → 6СО2(газ) + 6Н2О(ж), ∆Н0298 = -2817кДж рассчитайте стандартную энтальпию образования глюкозы используя стандартные энтальпии образования СО2(газ) и Н2О(ж).
Ответ: -1260 кДж/моль.
2. Установить путем расчета ∆G0 возможность протекания в стандартных условиях при 25°С следующей реакции:
СО(газ) + 3Н2(газ) → СН4(газ + Н2О(газ),
3. Определите величину энтропии следующих реакций:
а) 2NO(г) + O2(г) ↔2NO2(г)
б) СS2(г) + 2H2S(г) ↔CH4(г) + 4S(т)
в) 2NH3(г) ↔N2(г) + 3H2(г)
Вариант № 10
1. При сгорании 1 кг жира выделяется 32000 кДж теплоты. Допустив, что избыточный вес человека обусловлен отложением жира, определите расстояние, которое нужно пробежать человеку, чтобы "сбросить" 1 кг веса, если при умеренном беге затрачивается 400 кДж/км. Определите время такой пробежки со скоростью 8 км/час и оцените, насколько реально уменьшение веса тела человека в результате подобных физических упражнений.
Ответ: 10 час, 80 км.
2. Установить путем расчета ∆G0 возможность протекания в стандартных условиях при 25°С следующей реакции:
4НСl(газ) + О2 (газ) → 2H2O + 2О2 (газ).
3. Определите величину энтропии следующих реакций:
а) 2NO(г) + SO2(г) ↔N2O(г) + SO3(г)
б) Br2(г) + H2(г) ↔2HBr(г)
в) С(тв) + 2Н2(г) ↔СН4(г)
Тема 3.
Химическая кинетика. Химическое равновесие.
Вопросы для самостоятельной внеаудиторной подготовки к занятию:
1. Что изучает химическая, формальная и молекулярная кинетика? Значение химической кинетики для фармации.
2. Гомо- и гетерогенные, простые и сложные реакции. Что такое "молекулярность" и "порядок" реакции?
3. Скорость химической реакции. Факторы, влияющие на скорость гомо- и гетерогенной реакции.
4. Закон действующих масс для скорости реакции. Физический смысл константы скорости, факторы, влияющие на константу скорости.
5. Правило Вант-Гоффа. Температурный коэффициент.
6. Понятие обратимые и необратимые реакции. Абсолютно необратимые и практически необратимые реакции.
7. Химическое равновесие. Константа равновесия.
8. Понятие "смещение" или "сдвиг" химического равновесия. Факторы, влияющие на смещение химического равновесия. Принцип Ле Шателье.
Изучив эти вопросы, выполните письменно следующие задания:
Вариант № 1
1. Определите, во сколько раз увеличится скорость реакции, если температура возрастет от 20оС до 50оС. Температурный коэффициент равен 2.
Ответ: скорость реакции увеличится в 8 раз.
2. Обоснуйте, при изменении каких факторов равновесие можно сместить в сторону образования продуктов в реакции:
N2(г) + 3H2(г) ↔2NH3(г); ΔH ˚ <0.
3. Напишите выражение закона действующих масс для скорости прямых реакций и химического равновесия и рассчитайте Кравн при превращении 40% исходных веществ:
а) Br2(г) + H2(г) ↔2HBr(г)
б) Fe2O3(к) + 3H2(г) ↔ 2Fe(к) + 3H2O(г)
в) 2SO2(г) + O2(г) ↔ 2SO3(г)
Вариант № 2
1. Определите, на сколько градусов необходимо увеличить температуру, чтобы скорость реакции увеличилась в 16 раз. Температурный коэффициент равен 2.
Ответ: температуру необходимо увеличить на 40оС.
2. Обоснуйте, при изменении каких факторов равновесие можно сместить в сторону образования продуктов реакции:
2SO2(г) + O2(г) ↔2SO3(г); ΔH ˚ <0.
3. Напишите выражение закона действующих масс для скорости прямых реакций и химического равновесия и рассчитайте Кравн при превращении 30% исходных веществ:
а) N2(г) + 3H2(г) ↔ 2NH3(г)
б) 2Fe(к) + 3H2O(г) ↔ Fe2O3(к) + 3H2(г)
в) 2CO(г) + O2(г) ↔ 2CO2(г)
Вариант № 3
1. Рассчитайте, температурный коэффициент химической реакции, если при увеличении температуры на 50оС скорость реакции увеличилась в 32 раза.
Ответ: температурный коэффициент равен 2.
2. Обоснуйте, при изменении каких факторов равновесие можно сместить в сторону образования исходных веществ в системе: CO(г) + Cl2(г) ↔ СОCl2(г); ΔH ˚ >0.
3. Напишите выражение закона действующих масс для скорости прямых реакций и химического равновесия и рассчитайте Кравн при превращении 50% исходных веществ:
а) 2NO(г) + O2(г) ↔ 2NO2(г)
б) СS2(г) + 2H2S(г) ↔ CH4(г) + 4S(т)
в) 2NH3(г) ↔N2(г) + 3H2(г)






