| Кислота | Формула | Кдис |
| Азотистая | HNO2 | 4∙10-4 |
| Азотистоводородная | НN3 | 2,6 ∙10-5 |
| Азотная | HNO3 | 4,36∙10 |
| Алюминиевая (мета) | HAIO2 | 6∙10-13 |
| Борная (мета) | HBO2 | 7,5∙10-10 |
| Борная (орто) | Н3ВО3 | (I) 5,8∙10-10 (II) 1,8∙10-13 (III) 1,6 ∙10-14 |
| Кислота | Формула | Кдис |
| Бромоводородная | HBr | 1 ∙109 |
| Бромноватая | HВrO3 | 2∙10-1 |
| Бромноватистая | HBrO | 2,06 ∙10-9 |
| Водорода пероксид | H2О2 | (I) 2,63 ∙10-12 (II) 1,0∙10-25 |
| Галлия гидроксид | H3GaO3 | (II) 5∙10-11 (III) 2∙10-12 |
| Германиевая | H2GeO3 | (I) 1,7∙10-9 (II) 1,9∙10-13 |
| Дитионистая | H2S2O4 | (I) 5∙10-1 (II) 4∙10-3 |
| Железистосинеродистая | H4[Fe(CN)6] | (III) 1,0∙10-3 (IV) 5,6 ∙10-2 |
| Иодоводородная | HI | 1∙1011 |
| Иодная (орто) | H5IO6 | (I) 3,09∙1012 (II) 7,08∙10-9 (III) 2,5∙10-13 |
| Йодная (мета) | H1О3 | 1,7∙10-1 |
| Кремниевая (мета) | H2SiO3 | (I) 2,2∙10-10 (II) 1,6∙10-12 |
| Марганцовая | HМnО4 | 2∙102 |
| Молибденовая | H2MoО4 | (II) 1∙10-6 |
| Мышьяковая (орто) | H3AsО4 | (I) 5,9810-3 (II) 1,05∙10-7 (III) 3,89∙10-12 |
| Мышьяковистая (орто) | H3AsО3 | (I) 6∙10-10 (II) 1,7∙10-14 |
| Мышьяковистая (мета) | HasО2 | 6∙10-10 |
| Оловянистая | Н2SnO2 | 6∙10-18 |
| Оловянная | H2SnО3 | 4∙10-10 |
| Родановодородная | HSCN | 1,4∙10-1 |
| Пирофосфорная | Н4Р2О7 | (I) 1,4∙10-1 (II) 1,1∙10-2 (III) 2,1∙10-7 (IV) 4,1∙10-10 |
| Кислота | Формула | Кдис |
| Свинцовистая | H2PbO2 | 2∙10-16 |
| Селенистая | H2SeO3 | (I)3,5∙10-3 (II) 5,0∙lO-8 |
| Селеноводородная | H2Se | (I)1,7∙10-4 (II) 1,0∙10-11 |
| Селеновая | H2SeO4 | (I) 1,0∙103 (II) 1,2∙10-2 |
| Серная | H2SO4 | (I)1∙103 (II) 1,2∙10-2 |
| Сернистая | H2SO3 | (I)1,58∙10-2 (II) 6,31∙10-8 |
| Сероводородная | H2S | (I)6∙10-8 (II) 1∙10-14 |
| Сурьмяная (орто) | H3SbO4 | 4∙10-5 |
| Сурьмянистая (мета) | HSbO2 | 1∙10-11 |
| Теллуристая | H2TeO3 | (I)3∙10-3 (II) 2∙10-3 |
| Теллуроводородная | H2Te | 1∙10-8 |
| Теллуровая | H2TeO4 | (I)2,29 ∙10-8 (II) 6,46∙10-12 |
| Тетраборная | H2B4O7 | (I)1,8∙10-4 (II) 1,5∙10-5 |
| Тиосерная | H2S2О3 | (I)2,2∙10-1 (II) 2,8∙10-2 |
| Угольная | H2CО3 | (I)4,45∙10-7 (II) 4,69∙10-11 |
| Фосфористая(орто) | H3PО3 | (I)1,6∙10-3 (II) 6,3∙10-7 |
| Фосфорная (орто) | H3PO4 | (I)7,52∙10-3 (II) 6,31∙10-8 (III) 1,26∙10-12 |
| Фосфорноватистая | H3PO2 | 7,9 ∙10-2 |
| Фтороводородная | HF | 6,61∙10-4 |
| Хлористая | HCIO2 | 5∙10-3 |
| Хлороводородная(соляная) | HC1 | 1 ∙107 |
| Кислота | Формула | Кдис |
| Хлорноватистая | HC1О | 5,01∙l0-8 |
| Хромовая | H2CrO4 | (I) 1,8∙10-1 (II) 3,16∙10-7 |
| Циановодородная | HCN | 7,9∙I0-10 |
| Муравьиная | HCOOH | 1,77∙10-4 |
| Уксусная | CH3COOH | 1,75∙10-5 |
КОНСТАНТЫ ДИССОЦИАЦИИ НЕОРГАНИЧЕСКИХ ОСНОВАНИЙ В ВОДНЫХ РАСТВОРАХ
| Основание | Формула | Кдис |
| Гидроксид | ||
| алюминия | A1(OH)3 | (III) 1,38∙10-9 |
| аммония | NH4ОH | 1.79∙10-5 |
| бария | Ba(OH)2 | 2,3∙10-1 |
| галлия | Ga(OH)3 | (II) 1,6∙10-11 (III) 4∙10-12 |
| железа (II) | Fe(OH)2 | (II) 1,3∙10-4 |
| железа (III) | Fe(OH)3 | (II) 1,82∙10-11 (III) 1,35∙10-12 |
| кадмия | Cd(OH)2 | (II) 5,0∙10-3 |
| кальция | Ca(OH)2 | (II) 4,3∙10-2 |
| кобальта | Co(OH)2 | (II) 4∙10-5 |
| лития | Li(OH) | 6,75∙10-1 |
| магния | Mg(OH)2 | (II) 2,5∙10-3 |
| марганца | Mn(OH)2 | (II) 5,0∙10-4 |
| меди | Cu(OH)2 | (II) 3,4∙10-7 |
| натрия | NaOH | 5,9 |
| никеля | Ni(OH)2 | (II) 2,5 ∙10-5 |
| ртути | Hg(OH)2 | (I) 3,0∙10-11 (II) 5,0∙10-12 |
| свинца | Pb(OH)2 | (1) 9,6∙10-4 |
| cepeбpa | AgOH | 1,1∙ 10-4 |
| стронция | Sr(OH)2 | (II) 1,5∙10-1 |
| xpoмa | Cr(OH)3 | (III)1,02∙10-10 |
| цинка | Zn(OH)2 | (11) 4∙10-5 |
| таллия | TlОH | > 10-1 |
КОНСТАНТЫ НЕСТОЙКОСТИ КОМПЛЕКСНЫХ ИОНОВ
| Комплексный ион | Кнест | Комплексный ион | Кнест |
| Ag(NH3)2+ | 9,31∙ 10-8 | Cu (NH3)42+ | 2,14∙10-13 |
| Ag(CN)2- | 8,0∙ 10-22 | Cu(CN)2- | 1,0∙10-24 |
| Ag(CN)43- | 2,1 ∙10-21 | Cu(CN)43- | 5,0∙10-31 |
| Ag(SCN)2- | 2,7 ∙l0-8 | Cu(CN)42- | 5,0∙10-28 |
| Ag(SCN)43- | 1,3∙10-11 | CuCl42- | 6,3∙10-6 |
| Ag(SО4)23- | 0,59 | CuBr2- | 1,3∙10-6 |
| AgCl2- | 1,76∙10-5 | Cul2- | 1,75∙10-9 |
| AgCl32- | 4,0 ∙10-6 | Cu(OH)42- | 7,6∙10-17 |
| AgCl43- | 1,2∙ 10-6 | Cu(SO3)23- | 3,1∙10-9 |
| AgBr2- | 7,8∙10-8 | Fe(CN)64- | 1,0∙10-24 |
| Agl32- | 1,4∙10-14 | Fe(CN)63- | 1,0∙10-31 |
| Agl43- | 1,8∙10-14 | Fe(SO4)2- | 1,05∙10-3 |
| AgBr43- | 6,3∙10-10 | Hg(CN)42- | 4,0∙10-42 |
| Ag(S2О3)23- | 2,5∙10-14 | HgCl42- | 8,5∙10-16 |
| Ag(SО3)23- | 4,5 ∙10-9 | HgBr42- | 2,0∙10-22 |
| A1F63- | 1,44∙l0-20 | Hgl42- | 1,48∙10-30 |
| A1F4- | 1,8∙10-18 | Hg(SCN)42- | 5,9∙10-22 |
| Au(CN)2- | 5,0 ∙10-39 | Hg(S2O3)22- | 3,6∙10-30 |
| Au(SCN)2- | 1,0∙10-23 | Hg(NH3)42+ | 5,3∙10-20 |
| Au(SCN)4- | 1,0∙l0-42 | Mg(NH3)42+ | 10,9 |
| AuCl4- | 5,0 ∙10-22 | Ni(NH3)42+ | 1,12∙10-8 |
| AuBr2- | 4,0 ∙10-13 | Ni(NH3)62+ | 1,86∙10-9 |
| Cd(NH3)42+ | 7,56 ∙10-8 | Ni(CN)42- | 1,8∙10-14 |
| Cd(NH3)62+ | 7,3∙10-6 | PbCl42- | 7,1∙10-3 |
| Cd(CN)42- | 1,41∙ 10-19 | PbBr42- | 1,0∙10-3 |
| Cd(SCN)64- | 1,03 | Pbl42- | 1,4∙10-4 |
| CdCl42- | 9,3∙10-3 | PtCl42- | 2,5∙10-17 |
| CdCl64- | 2,6∙ 10-3 | PtBr42- | 4,0∙10-21 |
| CdBr42- | 2,0∙10-4 | PtI42- | 2,5∙10-30 |
| Комплексный ион | Кнест | Комплексный ион | Кнест |
| Cdl42- | 8,0∙10-7 | PdCl42- | 6,3∙10-13 |
| Cdl64- | 1,0∙l0-6 | PdBr42- | 8,0∙10-17 |
| Co(NH3)62+ | 7,75∙10-6 | Zn(NH3)42+ | 3,46∙10-10 |
| Co(NH3)63+ | 3,1∙10-33 | Zn(CN)42- | 1,3∙10-17 |
| Co(CN)64- | 1,0∙l0-19 | Zn(SCN)42- | 5,0∙10-2 |
| Co(CN)63- | 1,0∙10-64 | Zn(OH)42- | 3,6∙10-16 |
| Co(SCN)42- | 5,5∙10-3 | ZnCl42- | |
| Cu(NH3)2+ | 1,35∙10-11 | Znl42- |
17. ДИАГРАММЫ РАСЩЕПЛЕНИЯ d - ОРБИТАЛЕЙ В ОКТАЭДРИЧЕСКОМ И ТЕТРАЭДРИЧЕСКОМ ПОЛЯХ ЛИГАНДОВ
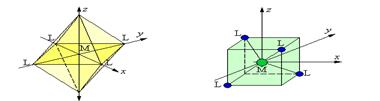
dg dg
Ион в сферическом поле









 ∆окт ∆тетр
∆окт ∆тетр







 de de
de de
свободный ион
РАСТВОРИМОСТЬ НЕОРГАНИЧЕСКИХ ВЕЩЕСТВ В ВОДЕ
ПРИ КОМНАТНОЙ ТЕМПЕРАТУРЕ
| Ионы | Br- | CH3COO- | CN- | CO32- | Cl- | F- | I- | NO3- | OH- | PO43- | S2- | SO42- | SO32- | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Zn2+ |
P М Р |
P Р Р |
P Р Н |
Н Р - |
Р М Р |
Н Р М |
Р Н Р |
Р Р Р |
М Р Н |
Н М Н |
Р Н Н |
Н Р |
Н Р Н |
р– хорошо растворимый (> 1 г на 100 г H2O); м – малорастворимый; н – практически нерастворимый (< 0,1 г на 100 г H2O); (-) – вещество не существует или разлагается водой; ∞ - неограниченная растворимость;? – нет данных






