Review Article
Gut-Microbiota and Mental Health: Current and Future Perspectives
Ajit Kumar Thakur1, Anshul Shakya2, Gulam Mohammed Husain3, Mila Emerald4 and Vikas Kumar1*
Recently, there is a growing interest of research on the relationship of gut-microbiota and neurological disorders. Increasing number of findings suggests the broader role of gut-microbiota in the modulation of various physiological and pathological conditions and it is now well recognized that a bidirectional communication between brain and gut-microbiota is essential to maintain homeostasis. The gut-brain axis includes central nervous system (CNS), the neuroendocrine and neuroimmune systems, autonomic nervous system, enteric nervous system, and intestinal microbiota. Probiotics (i.e., live microorganisms similar to beneficial microorganisms found in the human gut) are reported to modulate a number of disorders including metabolic disorders, behavioral conditions and cognitive functions. This review covers the significance of gut-brain axis in relation to the overall mental well-being. Apart from the recent studies highlighting the importance of gut-brain axis, here we also reviewed the interaction of few herbal medicines with gut-brain axis. Animal studies have indicated that some herbs or their isolated constituents alter the normal gut flora and have prominent effect on behavioral condition such as anxiety depression and cognition. Thus alteration of gut-brain axis by traditional medicines will be a potential strategy for the management of comorbid CNS disorders and gastrointestinal problems.
INTRODUCTION
The concept of the gut-brain axis, a term which describes the complex bidirectional communication system that exists between the central nervous system and the gastrointestinal tract and which is vital for maintaining homeostasis [1,2]. The gut-brain axis is involved in a multitude of physiological processes including satiety, food intake, regulation of glucose and fat metabolism, insulin secretion and sensitivity, bone metabolism [3,4], and lifespan [5].
Emotional or physical stressors may cause disturbances at every levels of the brain-gut axis including the central, autonomic and enteric nervous systems and affect regulation of visceral perception and emotional response to visceral events [6]. Brain communicates with the gut through multiple parallel pathways including autonomic nervous system, the hypothalamic pituitary-adrenal axis, and other connections, which were termed the brain-gut axis [7,8]. Based on previous studies there is strong evidence that exposure to stress, and release of catecholamines and norepinephrine into the GI tract during stress [9], may be responsible for the dysregulation of the gut-brain axis, via changing the GI motility, secretion of mucus and epithelial cells, thus leading to the different diseases of the gut [10]. It is also found that stress doing the early maturity life in animal, produces microbiota changes associated with inflammatory cytokines and increased levels of corticosterone [11]. Epidemiological studies have implicated stress of psychosocial, physical or immune origin as a trigger of first onset or exacerbation of irritable bowel syndrome symptoms [12-14]. In adult irritable bowel syndrome patients, acute stress episodes, chronic social stress, anxiety disorders, and maladaptive coping style determine the illness experience, health care-seeking behavior as well as treatment outcome [15,16]. Stress-related psychosocial factors such as somatization, neuroticism, and hypochondriasis are also important predictors in the development of post-infectious irritable bowel syndrome [17,18]. Microbiota provides the significant protection against incoming bacterial pathogens [19]. It has been shown that microbiota helps and protects the host against the viruses indirectly via activation of the inflammasome which is crucial for defense against influenza [20,21]. Interestingly, in spite of the fact that microbiota help the host to fight viruses (for example in case of influenza), it may also equally enhance viral infection via influence on virus replication by stimulating the proliferation or activation of target cells [22]. There is a growing appreciation of the critical role played by the commensally microbiota, both in our general wellbeing and in the specific functioning of the brain-gut axis. Interestingly, bacteria may respond directly to stress-related host signals because of interplay between stress and gut microbiota. Thereby, stress may influence the outcome of infections by these bacteria in many hosts [23].
Microbiota and host-metabolism
The human gastrointestinal microbiota represents a complex ecosystem that consists of bacteria, archaea, yeasts, planctomycetes and filamentous fungi and viruses, such as Senegal virus [24-30]. The human gastrointestinal tract typically comprises more than 10 times microbial cells that of the number of human cells in our bodies and containing 150 times as many genes as our genome [31,32]. The gut microbiota is therefore often referred to as the forgotten organ. The estimated number of species in the gut microbiota varies greatly, but it is generally accepted that the adult microbiota consists of more than 1,000 species which are belong to a few bacterial phyla [33], and more than 7,000 strains [32,34,35]. Interestingly, the gut microbiota modulated the expression of genes involved in immunity, nutrient absorption, energy metabolism and intestinal barrier function in human or mouse intestine [15].
The microbiota and host have mutually beneficial symbiotic relationships, which assure balanced habitat [36]. The compositions of the microbiota play an important role in the maintenance of intestinal homeostasis and host health [37]. Through the cooperative action of different functional microbial groups, the gut microbiota synthesizes essential amino acids and vitamins. In addition, by deploying an array of glycosidehydrolases and polysaccharide lysases, the microbiota facilitates utilization of otherwise indigestible food compounds [34,38]. Fermentation of saccharides by gut microbiota is the main source of energy for intestinal epithelial cells [26]. Microbial de-polymerization of complex carbohydrates and proteins gives rise to mono- and oligomeric compounds that are subsequently fermented into short-chain fatty acids (SCFAs) as well as to carbon dioxide and molecular hydrogen [39]. Carbohydrate fermentation and short-chain fatty acid production significantly improve the absorption of calcium, magnesium, and phosphorus [40].
For more than 50 years we have known that the administration of low doses of antibacterial agents promotes the growth of farm animals, consequently, in the United States, the largest use of antibiotics and related antimicrobial substances is within farms, with low doses fed to large numbers of animals used for food production to increase weight gain [41]. There are two main mechanisms by which it can maximize nutrient availability, either by the release of calories from otherwise unavailable oligosaccharides or by modulating absorption [42]. Alteration of gut microbiota can cause number of diseases for example, it has been shown that an increased ratio of the phylum Firmicutes to the genus Bacteroides is linked to obesity [43].
As might be expected given the importance of the microbiota in supporting host digestion and metabolism, obesity has been considered as an illness with a potential microbial basis [44]. Till 2004, there is over 138 data publications and 60 reviews for obesity and microbiota [45]. A perturbation of the resident flora in the accumulation of excess fat, microbial influences should not be considered in isolation because obesity is a multifactorial condition that also involves strong genetic factors, hypothalamic dysfunction, and an increase in the consumption of energy-dense food [46]. A significant energy source for humans is the bacterial metabolism of dietary fiber to short-chain fatty acids (SCFAs) [47]. SCFAs can modulate the host energy balance through Gpr41, a G protein coupled receptor that binds SCFA, and is dependent upon the gut microbiome. It is thought that interaction between SCFAs produced by the gut bacteria, and Gpr41 increases circulating levels of PYY, an enteroendocrine hormone that reduces gut motility and thus increases absorption of SCFAs [48]. A pictorial representation of Microbiota host metabolism is depicted in Figure 1.
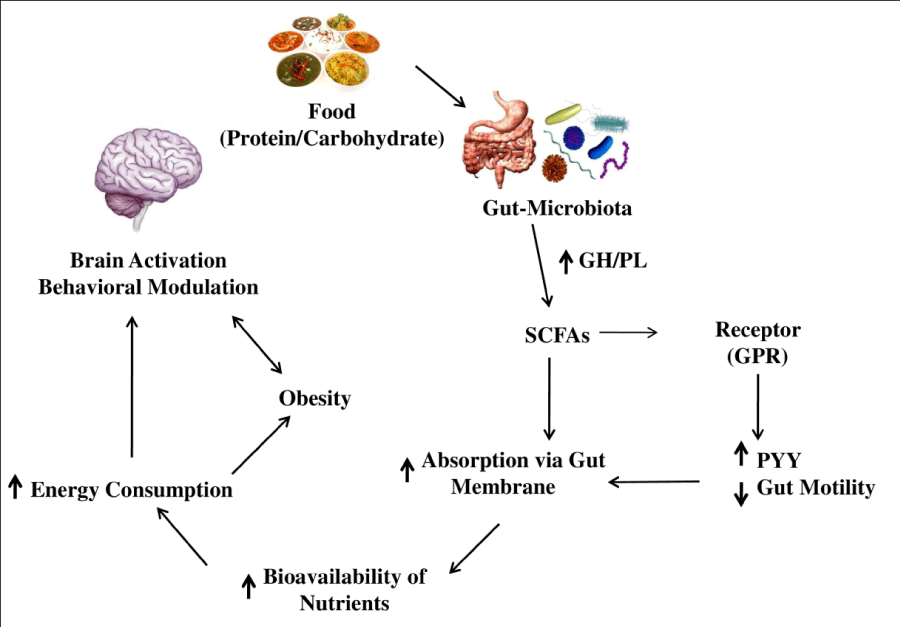 Figure 1 Microbial-host metabolism and the effect on behavioral function of brain. GH- Glycoside Hydrolases; PL- Polysaccharide Lysases; SCFAs-Short-Chain Fatty Acids; GPR- G-Protein Coupling Receptors; PYY- Peptide YY.
Figure 1 Microbial-host metabolism and the effect on behavioral function of brain. GH- Glycoside Hydrolases; PL- Polysaccharide Lysases; SCFAs-Short-Chain Fatty Acids; GPR- G-Protein Coupling Receptors; PYY- Peptide YY.

Several nutrients, including L-glutamine, L-glutamate, glucose, and sucrose, have physiological effects such as protecting the gastric mucosa, improving emotional state, and supplying energy in the subconscious state. These nutrients can also modulate subsequent behavior, such as brain activation and behavioral modulation resulting from internal signaling through the gut-brain axis [49]. Ingested nutrients are digested and absorbed in the gastrointestinal tract. The afferent vagus nerve, which innervates the entire gastrointestinal tract and projects to the nucleus of the solitary tract, is then activated, or peripheral humoral factors such as insulin and glucagon like peptide-1 (GLP-1) are released. In addition to absorption and metabolism, recent studies have indicated that the stomach, duodenum, and intestine contain chemosensing taste receptors and some kind of the G-protein coupling receptors (GPRs). The T1R receptor, which is related to the chemoreception of the sweet and the umami taste, and the T2R receptor, which is related to the chemoreception of the bitter taste, are both expressed in the gut [50,51]. In addition, GPR120 exists in both the oral cavity and the gastroinestinal tract in rodents. Fatty acids interact with GPR120 to induce the release of circulating GLP-1 [52]. Free fatty acids also interact with GPR40 in the gastrointestinal tract and promote the secretion of GLP-1 [53], and CCK [54]. GLP-1 and CCK evoke c-fos positive immunoreactivity in several brain regions, including the amygdala and the periaqueductal gray matter [55-57]. Intragastric infusion of glucose solution increases blood glucose, GLP-1, and insulin, and circulating GLP-1 acts on neurons in the nucleus of the solitary tract. Recently, it has been demonstrated that fluctuations in insulin following the intragastric administration of glucose correlate with the blood oxygenation level-dependent response in the amygdala, ventromedial hypothalamus, and nucleus accumbens [49,58].
The GI tract also is a locus of hormone production, including those involved in energy homeostasis (such as insulin, glucagon, leptin and ghrelin) and growth (for example, glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide 1 (GLP-1) [59]. Scientists working in this field develop a model of obesity by treating mice at their early life time by administering mid-range of US FDA-approved sub-therapeutic level of various antibiotics in their drinking water, and reported that subtherapeutic antibiotic treatment (STAT) in early life growth mice results in significant increase in adiposity, bone mineral density and GIP level. The increase GIP level was in supports with other existing models of obesity. But no significant differences for fasting insulin-like growth factor (IGF)-I, insulin, peptide YY, leptin, or ghrelin levels between control and STAT mice. Various dynamic phases of growth in young animals, STAT alterations of the microbiome may affect pluripotent cells that can become osteoblasts, adipocytes, or myocytes. Postulated that STAT exposures selected for microbiota with increased metabolic activity that were able to extract a higher proportion of calories from dietary complex carbohydrates that were relatively indigestible in the control mice. The increased SCFA concentrations are the metabolic products of this activity, which then may be delivered in increased quantities through the portal circulation to the liver, enabling enhanced lipogenesis. Enhanced caloric absorption has been implicated as a mechanism for increased weight gain in other murine obesity models [41].
Microbiota-gut-brain axis
In general the brain–gut–enteric microbiota axis includes the CNS, the neuroendocrine and neuroimmune systems, the sympathetic and parasympathetic arms of the autonomic nervous system (ANS), the enteric nervous system (ENS), and of course the intestinal microbiota. During the feeding, the gut released peptides which are affecting hypothalamic pathways, and especially arcuate nucleus involved in the regulation of satiety and metabolism. Put simply, through this bidirectional communication network, signals from the brain can influence the motor, sensory, and secretory modalities of the GIT and conversely, visceral messages from the GIT can influence brain function [60]. The vagus nerve is the direct communication observed between the bacteria and the brain [61]. A pictorial representation of the bidirectional communication represented in Figure 2. The cross talk between gut microbiota, the immune system and the brain-gut axis plays an important role in the modulation of the stress response of the gut in the context of the development of different gut disorders as microbiota communicate with the gut-brain axis through different mechanisms viz. direct interaction with mucosal cells (endocrine message), via immune cells (immune message), and via contact to neural endings (neuronal message) [2].
 Figure 2 Bidirectional communications between Gut-Microbiota and Gut-Brain Axis (GBA) in the modulation of the stress response. Microbiota communicate with the gut-brain-axis through different mechanisms viz. direct interaction with mucosal cells (endocrine message), via immune cells (immune message), and via contact to neural endings (neuronal message) to influence brain development and behavior. Stress through GBA effect on Gut-Microbiota which is responsible for functional GI disorders and dysbiosis. Similarly dysbiosis effect synthesis of several microbial by-product and precursor that gain access to the brain via the bloodstream and the area postrema, via cytokine release from mucosal immune cells, via the release of gut hormones such as 5‑hydroxytryptamine (5‑HT) from entero-endocrine cells, or via afferent neural pathways, including the enteric nervous system.
Figure 2 Bidirectional communications between Gut-Microbiota and Gut-Brain Axis (GBA) in the modulation of the stress response. Microbiota communicate with the gut-brain-axis through different mechanisms viz. direct interaction with mucosal cells (endocrine message), via immune cells (immune message), and via contact to neural endings (neuronal message) to influence brain development and behavior. Stress through GBA effect on Gut-Microbiota which is responsible for functional GI disorders and dysbiosis. Similarly dysbiosis effect synthesis of several microbial by-product and precursor that gain access to the brain via the bloodstream and the area postrema, via cytokine release from mucosal immune cells, via the release of gut hormones such as 5‑hydroxytryptamine (5‑HT) from entero-endocrine cells, or via afferent neural pathways, including the enteric nervous system.

Microbiota also interacts with host gut-brain axis through neurohumoral communication to influence brain development and behavior [62]. For example, alteration in gastrointestinal function is communicated to the brain bringing about the perception of visceral events such as nausea, satiety, and pain or when, in turn, stressful experiences lead to altered gastrointestinal secretions and motility [63]. The neuroendocrine, neuroimmune, the sympathetic and parasympathetic arms of the autonomic nervous system and the enteric nervous system are the key pathways through which they communicate with each other [64]. This might influence a broad spectrum of diseases, psychiatric conditions and other disorders [65]. Putative mechanisms by which microbes access the brain and influence behavior include microbial products that gain access to the brain, via cytokine release from the mucosal immune cells, via the release of gut hormones such as 5-HT from endocrine cells, or via afferent neural pathways, including the vagus nerve. Stress and emotions can also influence the microbial composition of the gut through the release of stress hormones or sympathetic neurotransmitters (GABA, 5-HT precursors etc.) that influence gut physiology and alter the habitat of the microbiota and also these catecholamine alter the growth, motility and virulence of pathogenic and commensally bacteria. Alternatively, host stress hormones such as noradrenalin might influence bacterial gene expression or signaling between bacteria, and this might change the microbial composition and activity of the microbiota [66].
Some of the earliest indications of a critical role of the gut microbiota in stress responses are well recognized. Germ-free animals were identified as having exaggerated hypothalamic–pituitary–adrenal (HPA) axis activation in response to stress. This hyper responsiveness was reversed by reconstitution with feces from animals kept in a pathogen- free environment or with a single bacterial strain, Bifidobacterium infantis [67]. More recently, two studies have indicated that germ-free conditions during early growth of mice results in decreased anxiety-like behavior compared to conventional animals [68,69]. Addressing neural correlates of reduced anxiety in germ-free animals, Diaz et al. demonstrated that NGFI-A mRNA expression was significantly lower in various sub-regions of the prefrontal cortex, including the orbital frontal cortex and the striatum, hippocampus dentate gyrus, and amygdala, compared with specific pathogen-free mice [68]. Germ-free mice also had significantly lower BDNF mRNA expression in the hippocampus, amygdala, and cingulate cortex, which are important components of the neural circuitry underlying anxiety and fear [70, 71]. In addition to altered neurotrophin levels, changes have been reported in NMDA receptor subunit expression with decreased NR1 and NR2A in the hippocampus, decreased NR2A in the cortex, and decreased NR2B in the amygdala, but not in the hippocampus [67,69]. Enhanced turnover rate of noradrenaline, dopamine, and 5-HT has also been demonstrated in the striatum of germ-free mice compared with specific pathogen-free mice [68].






