ѕрограмн≥ питанн€
ѕоширен≥сть та форми знаходженн€ метал≥в у природ≥. ћетоди добуванн€ метал≥в: п≥рометалург≥€, електрометалург≥€, г≥дрометалург≥€.
≈лектронна будова атом≥в метал≥в. ¬≥дновн≥ властивост≥ метал≥в. ¬заЇмод≥€ метал≥в з простим≥ речовинами, водою, розчинами луг≥в та кислот.
ЌагадаЇмо, що х≥м≥чн≥ елементи розпод≥л€ютьс€ на дв≥ групи: метали ≥ неметали. « ф≥зичноњ точки зору ще з час≥в ћ.¬.Ћомоносова (1748 р.) метали мають сукупн≥сть загальних властивостей: так званий металевий блиск, тобто, с≥рий кол≥р (виключенн€ Ц м≥дь ≥ золото); висок≥ значенн€ тепло- ≥ електропров≥дностей; €к правило, - високу механ≥чну м≥цн≥сть та висок≥ температури плавленн€; пластичн≥сть Ц зд≥бн≥сть метал≥в п≥д впливом механ≥чних навантажень деформуватись без руйнуванн€ (ковк≥сть). «агальн≥сть ф≥зичних властивостей метал≥в обумовлена специф≥чн≥стю метал≥чного звТ€зку та особливост€ми будови метал≥чноњ реш≥тки. ” неметал≥в такий сукупност≥ загальних властивостей немаЇ.
“акож в≥домо, €кщо провести д≥агональ в≥д Be до At в пер≥одичн≥й таблиц≥ х≥м≥чних елемент≥в ƒ.≤.ћенделЇЇва, то головних п≥дгрупах зл≥ва в≥д ц≥Їњ д≥агонал≥ будуть знаходитись метали, а з права Ц неметали. Ќеметал≥в нал≥чуЇтьс€ всього 21 Ц H, B, C, N, O, F, Si, P, S, Cl, As, Se, Br, Te, I, а також ≥нертн≥ елементи - He, Ne, Ar, Kr, Xe, Rn. ќднак, в багатьох крањнах вид≥л€ють ще одну групу елемент≥в Ц металоњд≥в. —≥м елемент≥в ц≥Їњ групи знаход€тьс€ вздовж д≥агонал≥, утворюючи так зван≥ Усх≥дцiФ: B, Si, Ge, Sb, As, Te, At. ’≥м≥чн≥ властивост≥ сполук цих елемент≥в характерн≥ €к дл€ сполук метал≥в, так ≥ неметал≥в.
« точки зору електронноњ будови атоми метал≥в мають невелику к≥льк≥сть електрон≥в на зовн≥шн≥м (валентн≥м) р≥вн≥: ns1-2. ÷≥ електрони слабо взаЇмод≥ють з €дром атома (низки значенн€ енерг≥њ ≥он≥зац≥њ та стандартних окисно-в≥дновних потенц≥ал≥в) ≥ тому вони легко в≥дриваютьс€ в≥д нього. јтом метала, таким чином, про€вл€Ї властивост≥ в≥дновника ≥ окислюЇтьс€:
ће0 - ne = ћеn+
¬загал≥ до метал≥в, кр≥м s елемент≥в - ns1-2 (виключенн€ Ц H, He), в≥днос€тьс€ також вс≥ d- ≥ f- елементи (d1-10 ≥ f1-14) без виключень, нав≥ть частина р-елемент≥в Ц з 1-2 електронами на цьому п≥др≥вн≥. як вже було нагадано, метали, вступаючи в реакц≥њ, окислюютьс€. “ому значенн€ њх окисно-в≥дновних ( red-ox або електродних ) потенц≥ал≥в, (€к правило, стандартн≥ значенн€) мають бути меншими за в≥дпов≥дн≥ значенн€ окисник≥в:
 ће <
ће <  окисника
окисника
ƒл€ студент≥в енергетик≥в, електроенергетик≥в, студент≥в любого техн≥чного напр€мку дуже важливо знати, €к той чи ≥нший метал реагуЇ з речовинами, в середовище €ких в≥н ≥снуЇ Ц пов≥тр€м, киснем, воднем, азотом, з водою ≥ водневими розчинами луг≥в, кислот ≥ таке ≥нше. « цими знанн€ми також звТ€зани питанн€, €ки стосуютьс€ повед≥нки метал≥в та њх сплав≥в ≥ конструкц≥й, вироблених з р≥зних метал≥в, у вод≥ та в вологому пов≥тр≥ ≥ водневих середовищах р≥зних речовин Ц питанн€ електрох≥м≥чноњ короз≥њ.
¬заЇмод≥€ метал≥в з водою. јтомом-окисником в реакц≥€х з водою Ї кат≥он г≥дрогену, Ќ+, води:
|
|
|
ће  - ne = Me
- ne = Me 
2 Ќ2ќ + 2e = H  + 2ќЌ-;
+ 2ќЌ-;  = -0,41 ¬
= -0,41 ¬
“обто, принципово повинн≥ реагувати з водою т≥льки метали,  котрих Ї меншим за -0,41 ¬ольт (дивись табл. є). Ќаприклад:
котрих Ї меншим за -0,41 ¬ольт (дивись табл. є). Ќаприклад:
2Na + 2 Ќ2ќ = 2 NaOH + H2
Na0 - 1e = Na+ x2
2H+ + 2x1e = H2 x1
“обто, продуктом в≥дновленн€ Ї газопод≥бний водень, продуктом окисленн€ Ц розчинн≥ у вод≥ г≥дроксиди метал≥в. ќднак, б≥льш≥сть метал≥в з такими значенн€ми потенц≥ал≥в мають на своЇњ поверхн≥ нерозчинн≥ у вод≥ захисн≥ пл≥вки оксид≥в. “ому вони не розчин€ютьс€ Ц Mg, Al, Zn, Cr, FeЕ
¬заЇмод≥€ метал≥в з розчинами луг≥в. якщо ц€ захисна пливка оксиду маЇ амфотерний характер, то вона буде розчин€тись в розчин≥ лугу, ≥ метал буде взаЇмод≥€ти з водою. “акими металами Ї: Be, Zn, Sn, Pb (т≥льки ц≥ двовалентн≥), Al, Cr,Е(та ≥нш≥ трьохвалентн≥).
ѕосл≥довн≥сть стад≥й взаЇмод≥њ в таких випадках буде наступною:
1. –озчиненн€ захисноњ оксидноњ пл≥вки з утворенн€м розчинних у вод≥ гидроксокомплекс≥в металу Ц
ZnO + 2NaOH + H2O = Na2[Zn(OH)4 ]
Al2O3 + 6NaOH + 3H2O = 2Na3[ Al(OH)6]
в≥дпов≥дно дл€ дво- ≥ трьохвалентних метал≥в.
2. ¬заЇмод≥€ металу з водою (окисно-в≥дновна реакц≥€) -
Zn + 2H2O =  Zn(OH)2 + H2
Zn(OH)2 + H2
2Al + 6H2O =  2Al(OH)3 + 3H2
2Al(OH)3 + 3H2
3. –озчиненн€ осаду гидроксиду у надлишку лугу Ц
Zn(OH)2 + 2NaOH = Na2[Zn(OH)4 ]
Al(OH)3 + 3NaOH = Na3[ Al(OH)6].
“ак €к перша ступ≥нь реакц≥њ даЇ лише незначну к≥льк≥сть продукт≥в, то складанн€ р≥вн€нь двох останн≥х реакц≥й приводить до сумарного р≥вн€нн€ процесу взаЇмод≥њ металу з розчином лугу:
Zn + 2NaOH +2H2O = Na2[Zn(OH)4 ] + H2
Zn - 2e = Zn2+ x1
2H2O + 2x1e = H2 + 2OH- x1
2Al(OH)3 + 6NaOH + 6H2O = 2Na3[ Al(OH)6] + 3H2
Al0 - 3e = Al3+ x2
2H2O + 2x1e = H2 + 2OH- x3
“обто, продуктом в≥дновленн€ Ї газопод≥бний водень, продуктом окисленн€ Ц розчинн≥ у вод≥ г≥дроксокомплекси ц≥х метал≥в.
“≥ж сам≥ метали взаЇмод≥ють ≥з розтопленими лугами (при нагр≥ванн≥) в в≥дсутност≥ води. ≤оном-окисником в цих випадках буде Ќ+ лугу, а продуктом окисненн€ Ц сол≥ кислот, €ки в≥дпов≥дають амфотерним г≥дроксидам Ц ће(ќЌ)2 та ће(ќЌ)3:
Zn + 2KOH = K2ZnO2 + H2;
2Al + 6KOH = 2K3AlO3 + 3H2;
¬заЇмод≥€ метал≥в з розчинами кислот СТ неокисник≥в СТ (HCl, H  SO
SO 
 , H
, H  PO
PO  , CH
, CH  COOHЕ), тобто розчинами кислот - слабких окисник≥в в пор≥вн€нн≥ з розчинами кислот Ц сильних окисник≥в. јтомом-окисником знову ж таки Ї кат≥он г≥дрогену, але з б≥льшим значенн€м окисно-в≥дновного потенц≥алу, н≥ж у вод≥ -
COOHЕ), тобто розчинами кислот - слабких окисник≥в в пор≥вн€нн≥ з розчинами кислот Ц сильних окисник≥в. јтомом-окисником знову ж таки Ї кат≥он г≥дрогену, але з б≥льшим значенн€м окисно-в≥дновного потенц≥алу, н≥ж у вод≥ - 
 0. “ому в таких розчинах повинн≥ розчин€тись метали, стандартн≥ red-ox потенц≥али менш≥ нул€ ¬ольт, тобто т≥ метали, €ки знаход€тьс€ в р€ду напруг метал≥в до г≥дрогену:
0. “ому в таких розчинах повинн≥ розчин€тись метали, стандартн≥ red-ox потенц≥али менш≥ нул€ ¬ольт, тобто т≥ метали, €ки знаход€тьс€ в р€ду напруг метал≥в до г≥дрогену:
Fe + H  SO
SO 
 = FeSO4 + H2
= FeSO4 + H2  ;
;  = -0,44 B
= -0,44 B
2Al + 3H  SO
SO 
 = Al2(SO4)3 + 3H2
= Al2(SO4)3 + 3H2  ;
;  = -1,66 B
= -1,66 B
Cu + HCl =, так €к  = + 0,34 B
= + 0,34 B 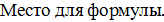
ќднак, в де€ких випадках (Pb, Sn,Е), реакц≥€, котра принципово мала б переб≥гати, не зд≥йснюЇтьс€, або переб≥гаЇ з дуже малими швидкост€ми по причинам поган≥й розчинност≥ сол≥ ≥ таке ≥нше.
¬заЇмод≥€ метал≥в з розчинами кислот Ц сильних окисник≥в. ѕри реакц≥€х метал≥в з водними розчинами кислот-окисник≥в, таких €к H  SO
SO 
 , та ЌNO
, та ЌNO 
 , ЌNO
, ЌNO 
 , потр≥бно памТ€тати, що атомами окисниками в цих випадках Ї атоми кислото-утворюючих елемент≥в - S
, потр≥бно памТ€тати, що атомами окисниками в цих випадках Ї атоми кислото-утворюючих елемент≥в - S  , N
, N  . ѕродуктами окисленн€ будуть, €к правило, сол≥ метал≥в. —клад продукт≥в в≥дновленн€ залежить в≥д активност≥ метал≥в (дивись р€д стандартних red-ox потенц≥ал≥в метал≥в, таблЕ..). ƒуже активн≥ метали (Li Ц Mg) в≥дновлюють концентровану сульфатну кислоту до Ќ
. ѕродуктами окисленн€ будуть, €к правило, сол≥ метал≥в. —клад продукт≥в в≥дновленн€ залежить в≥д активност≥ метал≥в (дивись р€д стандартних red-ox потенц≥ал≥в метал≥в, таблЕ..). ƒуже активн≥ метали (Li Ц Mg) в≥дновлюють концентровану сульфатну кислоту до Ќ  S; активн≥ (¬е Ц Cr) Ц до S; малоактивн≥ (Fe Ц Hg) Ц до SO
S; активн≥ (¬е Ц Cr) Ц до S; малоактивн≥ (Fe Ц Hg) Ц до SO  . ѕод≥бним же чином д≥Ї розведена н≥тратна кислота. ¬≥дпов≥дними продуктами в≥дновленн€ н≥трогену будуть: NH4NO3, N
. ѕод≥бним же чином д≥Ї розведена н≥тратна кислота. ¬≥дпов≥дними продуктами в≥дновленн€ н≥трогену будуть: NH4NO3, N  O; N
O; N  ; NO. Ќеобхидно пам'€тати, що в б≥льшост≥ випадк≥в вид≥л€ютьс€ вс≥ продукти в≥дновленн€, але в р≥зн≥х сп≥вв≥дношенн€х. ћи же пишимо т≥льк≥ основной з них.
; NO. Ќеобхидно пам'€тати, що в б≥льшост≥ випадк≥в вид≥л€ютьс€ вс≥ продукти в≥дновленн€, але в р≥зн≥х сп≥вв≥дношенн€х. ћи же пишимо т≥льк≥ основной з них.
|
|
|
¬≥дпов≥дн≥ схеми:
Li

 H2S
H2S  + с≥ль металу + H2O
+ с≥ль металу + H2O
+8e Mg
+6e Be
H  S+6 O
S+6 O  конц.
конц. 
 S
S  + с≥ль металу + H2O
+ с≥ль металу + H2O
Cr
+ 2e Fe

 SO2
SO2  + с≥ль металу + H2O
+ с≥ль металу + H2O
Hg
Ќеметали окислюютьс€, €к правило, до вищих оксид≥в або до в≥дпов≥дних кислот.
Li
дл€ дуже розведеноњ 
 N-3H4N+5O3 + с≥ль металу + H2O
N-3H4N+5O3 + с≥ль металу + H2O
+8e 5е х2 Mg  N2
N2  + с≥ль металу + H2O
+ с≥ль металу + H2O
+4e x2 Be
HN+5 O3розвед. 
 N2O
N2O  + с≥ль металу + H2O
+ с≥ль металу + H2O
Cr
+3e Fe

 SO2
SO2  + с≥ль металу + H2O
+ с≥ль металу + H2O
Hg
онцентрована н≥тратна кислота незалежно в≥д активност≥ металу в≥дновлюЇтьс€ з утворенн€м NO  :
:
1 Mg + HNO3 конц.  2 NO2 + 1 Mg(NO3)2 + H2O
2 NO2 + 1 Mg(NO3)2 + H2O
N+5 +e = N+4 x2
Mg0 - 2e = Mg2+ x1
¬сього з права Ї 4 атома н≥трогену, тому перед н≥тратною кислотой ставимо коеф≥ц≥Їнт 4. «л≥ва Ц 4 атом≥в г≥дрогену, сл≥д перед водой Ц 2:
1 Mg + 4 HNO3 конц. = 2 NO2 + 1 Mg(NO3)2 + 2 H2O
ѕерев≥рка по к≥лькост€м оксигену даЇ по 12. оеф≥ц≥Їнти розставлен≥ в≥рно.
онцентрован≥ н≥тратна та сульфатна кислоти СТна холоду СТ (без нагр≥ванн€) пасивують де€к≥ метали (Al, Cr, Fe, Co, Ni, Ti, Zr,Е), тобто перевод€ть њх в неактивний стан, утворюючи на поверхн€х метал≥в захисну оксидну пл≥вку, €ка не розчин€Їтьс€ у розчин≥ ≥ запоб≥гаЇ переб≥гу реакц≥њ:
Al + 6 HNO3 конц.  Al2O3
Al2O3  + 6 NO2
+ 6 NO2  + 3 H2O
+ 3 H2O
2Al0 - 3e x2 = Al3+ x1
N+5 + 1e = N+1 x6
¬ обох частинах р≥вн€нн€ по 18 атом≥в оксигену, тому коеф≥ц≥Їнти розставлени в≥рно.
ѕотр≥бно також памТ€тати, що розчини кислот - сильних окисник≥в зд≥бн≥ окислювати метали, що сто€ть в р€ду напруг метал≥ т≥льки по Hg (меркур≥й) включно. ћенш активн≥ метали (Au, Pt, Е.) окислюютьс€ т≥льки сум≥шами розчин≥в кислот.






