’имическа€ кинетика Ц раздел химии, изучающий скорость и механизм химических реакций. –азличают гомогенные и гетерогенные химические реакции.
√омогенные реакции протекают в объеме одной фазы Ц в жидком (водном) или газовом растворе. Ќапример, 2—ќ(г) + ќ2(г) = 2—ќ2(г) (8)
√етерогенные реакции протекают на границе раздела фаз: г Ц ж, г Ц т,
т Ц т и т.д. Ќапример, 2—(т) + ќ2(г) = 2—ќ(г) (9)
—корость химической реакции (υ ) Ц это изменение количества вещества одного из реагентов за единицу времени в единице реакционного пространства. ќна зависит от природы реагирующих веществ, температуры (“), давлени€ (–), концентрации (—) реагирующих веществ и других факторов. «ависимость υ от — выражаетс€ законом действующих масс: ѕри посто€нной температуре скорость химической реакции пр€мо пропорциональна произведению концентраций реагирующих веществ, вз€тых в степен€х, равных стехиометрическим коэффициентам в уравнении реакции.
Ќапример, дл€ гомогенной реакции (8) υ = k∙—2—ќ ×—ќ2, где k Ц константа скорости химической реакции, котора€ зависит от природы реагирующих веществ, температуры, присутстви€ катализатора, но не зависит от концентрации реагирующих веществ; — Ц мол€рные концентрации веществ (моль / л). ƒл€ гетерогенной реакции (9) υ = k×—ќ2 , так как концентрации твердых веществ посто€нны и прин€ты равными единице, т.е. —— = 1.
«ависимость скорости химической реакции от температуры выражает правило ¬ант-√оффа: при повышении температуры на каждые 10º скорость химической реакции увеличиваетс€ примерно в 2 Ц 4 раза. ћатематическое выражение закона ¬ант-√оффа:

 , (10)
, (10)
где υ“1 и υ“2 Ц скорости реакции при температурах “1 и “2; γ - температурный коэффициент скорости химической реакции. ќн показывает, во сколько раз увеличиваетс€ скорость реакции при повышении температуры на 10º.
ѕо полноте протекани€ химических процессов различают необратимые и обратимые химические реакции. Ќеобратимые реакции протекают только в одном направлении и завершаютс€ полным превращением исходных веществ в продукты реакции. ќбратимые реакции при одной и той же температуре протекают как в пр€мом, так и в обратном направлении и завершаютс€ установлением в системе химического равновеси€. ќни не идут до конца, в системе всегда остаютс€ исходные вещества:
N2(г) + 3H2(г) ⇄ 2NH3(г) (11)
’имическое равновесие Ц такое состо€ние химического взаимодействи€, при котором скорости пр€мой и обратной реакций равны между собой, т.е.  .
.
ѕосле наступлени€ состо€ни€ равновеси€ концентрации реагирующих веществ при данных услови€х остаютс€ неизменными и называютс€ равновесными концентраци€ми, обозначаютс€ в квадратных скобках [ ]. оличественной характеристикой химического равновеси€ €вл€етс€ константа химического равновеси€ Ц величина, определ€ема€ соотношением равновесных концентраций продуктов реакции и исходных веществ.≈сли в соотношение дл€ константы равновеси€ вход€т равновесные концентрации, то ее обозначают символом с, а если используютс€ парциальные давлени€ газов (–i), то Ц символом р. Ќапример, дл€ реакции (11):
|
|
|
 ,
, 
—осто€ние химического равновеси€ устанавливаетс€ и сохран€етс€ лишь при определенных услови€х (температуре, давлении и концентрации реагирующих веществ). ѕри изменении хот€ бы одного из этих условий, т.е. при каком-либо внешнем воздействии, равновесие в системе нарушаетс€, и система переходит в новое состо€ние равновеси€. Ётот переход называетс€ смещением химического равновеси€.
¬ли€ние внешних воздействий (изменение “, – или —) на состо€ние химического равновеси€ можно предсказать, пользу€сь принципом Ће Ўателье - Ѕрауна: если на систему, наход€щуюс€ в равновесии, оказать внешнее воздействие, то равновесие смещаетс€ в том направлении, которое ослабл€ет эффект внешнего воздействи€, т.е на внешнее воздействие система отвечает противодействием. “ак увеличение концентрации какого-либо вещества вызовет противодействие системы Ц стремление уменьшить концентрацию этого вещества, что возможно путем смещени€ равновеси€ в направлении, при котором концентраци€ этого вещества будет уменьшатьс€.
ѕовышение –общ или –i; приводит к смещению равновеси€ в сторону протекани€ той реакции, котора€ вызывает уменьшение –общ и –i и наоборот, понижение –общ или Pj смещает равновесие в направлении той реакции, котора€ приводит к увеличению этих параметров.
ѕовышение температуры вызывает смещение равновеси€ в направлении протекани€ той реакции, котора€ сопровождаетс€ поглощением теплоты, т.е. эндотермической (∆Ќ > 0). ѕонижение температуры будет вызывать смещение равновеси€ в сторону экзотермической реакции, сопровождающеес€ выделением теплоты (∆Ќ < 0).
ѕримеры решени€ задач
ѕример 1. ак изменитс€ скорость реакции 2Nќ(г) + ќ2(г) ⇄ 2NO2(г) , если
одновременно концентрацию Nќ увеличить в 2 раза, а концентрацию ќ2 уменьшить в 8 раз?
– е ш е н и е. 1) «ависимость скорости химической реакции от концентрации реагирующих веществ выражаетс€ законом действующих масс, поэтому записываем математическое выражение этого закона дл€ данной реакции в начальный момент времени: 
—огласно условию концентрацию NO увеличили в 2 раза, т.е. —'NO=2— NO, а концентрацию ќ2 уменьшили в 8 раз, т.е. —'o2 = —o2 /8.
2) «аписываем выражение дл€ скорости реакции в конечный момент времени: υ' = k × (—' NO)2 × —'o2 или 
3)  ќпредел€ем, как измен€етс€ скорость реакции. ƒл€ этого рассматриваем отношение
ќпредел€ем, как измен€етс€ скорость реакции. ƒл€ этого рассматриваем отношение 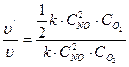 = 1/2
= 1/2
ќтвет: при одновременном увеличении концентрации Nќ в 2 раза и уменьшении концентрации ќ2 в 8 раз скорость реакции уменьшаетс€ в 2 раза.
ѕример 2. ќпределите, во сколько раз измен€тс€ скорости пр€мой и обратной реакций в системе 2SO2 (г) + ќ2 (г) ⇄ 2Sќ3 (г), если объем газовой смеси уменьшить в 3 раза. аково направление смещени€ равновеси€ в этой системе?
– е ш е н и е. 1) «аписываем выражени€ дл€ скоростей пр€мой и обратной реакций, использу€ закон действующих масс:  ,
, 
2) –ассматриваем, что происходит в реакционной системе при уменьшении ее объема. ”меньшение объема гомогенной системы в 3 раза приводит к увеличению концентрации каждого из реагирующих веществ также в 3 раза, т.е. математически это записываем в виде: —'so2 = 3— so2, —'о2 = 3—o2 и —'so3 = 3— so3.
3) «аписываем выражение дл€ скоростей пр€мой и обратной реакций после изменени€ объема системы:


4) ќпредел€ем, во сколько раз измен€ютс€ скорости пр€мой и обратной реакций: 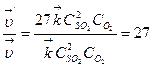 ;
; 
|
|
|
“аким образом, при уменьшении объема газовой смеси в 3 раза скорость пр€мой реакции увеличитс€ в 27 раз, а скорость обратной реакции Ц в 9 раз.
5) ”станавливаем направление смещени€ равновеси€ в этой системе. ¬ св€зи с тем, что скорость пр€мой реакции увеличилась в 27 раз, а обратной Ц только в 9 раз, равновесие сместитс€ в направлении протекани€ пр€мой реакции, т.е. в сторону образовани€ SO3.
ѕример 3. –ассчитайте температурный коэффициент скорости химической реакции, если известно, что константа скорости этой реакции при 140∞— равна 5,5 × 10-4, а при 185∞— Ц 9,2 × 10-3.
ƒано: “1 =140∞ —, k1 =5,5 × 10 -4; “2= 185∞—, k2 = 9,2∙× 10-3. Ќайти γ.
– е ш е н и е. 1) «ависимость скорости химической реакции от температуры описываетс€ правилом ¬ант-√оффа в форме: 
Ќо дл€ проведени€ расчетов это уравнение необходимо прологарифмировать, т.е. 

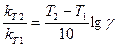
2) –ассчитываем температурный коэффициент скорости этой реакции.
а) вычисл€ем температуру по абсолютной шкале:
T1 = 140 + 273 = 413 , “2 = 185 + 273 = 458 ;
б) рассчитаем численное значение γ:
 , lg 16,7 = 4,5 lgγ
, lg 16,7 = 4,5 lgγ
отсюда  , т.е. lg γ = 0,27, и тогда γ = 1,87.
, т.е. lg γ = 0,27, и тогда γ = 1,87.
ќтвет: температурный коэффициент этой реакции равен 1,87.
ѕример 4. ѕри синтезе аммиака N2 + «Ќ2 ⇄ 2NЌз равновесие установилось  при следующих концентраци€х реагирующих веществ (моль/л) [ N2 ] = 4; [ H2 ] = 2 и [ NH3 ] = 6. –ассчитайте константу равновеси€ и исходные концентрации Ќ2 и N2.
при следующих концентраци€х реагирующих веществ (моль/л) [ N2 ] = 4; [ H2 ] = 2 и [ NH3 ] = 6. –ассчитайте константу равновеси€ и исходные концентрации Ќ2 и N2.
ƒано: [ Ќ2 ]=2 моль/л, [ N2 ] = 4 моль/л, [ NH3 ] = 6 моль/л. Ќайти с, —исх (Ќ2), Cиcx (N2).
– е ш е н и е. 1) «аписываем выражение дл€ константы равновеси€ в этой системе и рассчитываем ее значение:  ;
; 



2) ќпредел€ем исходные концентрации Ќ2 и N2.
ƒл€ обратимых реакций ни одно из реагирующих веществ не расходуетс€ полностью. ѕоэтому исходна€ концентраци€ вещества ј складываетс€ из равновесной концентрации этого вещества [ј] и концентрации этого вещества, котора€ была израсходована на реакцию к моменту установлени€ равновеси€ (условно обозначим ее X (ј). “аким образом, выражение дл€ исходной концентрации вещества ј может быть представлено в виде: —исх(ј) = [ј] + ’(ј).
а) ƒл€ определени€ —исх(N2) и —исх(Ќ2) обращаемс€ к уравнению реакции, согласно которому на образование 2 моль NH3 требуетс€ 1 моль N2 и 3 моль Ќ2. Ќо поскольку равновесна€ концентраци€ NH3 равна 6 моль/л, то при этом было израсходовано 6/2 = 3 моль N2 и 3×(6 / 2) = 9 моль Ќ2.
б) –ассчитаем —исх(N2) и —исх(Ќ2):
—исх(Ќ2) = [Ќ2] + ’(Ќ2) = 2 + 9 = 11 моль/л,
Cисх(N2) = [N2] + X(N2) = 4 + 3 = 7 моль/л.
ќтвет: константа равновеси€ реакции равна 1,1. »сходные концентрации Ќ2 и N2 составл€ют 11 моль/л и 7 моль/л соответственно.
ѕример 5. ”становите направление смещени€ равновеси€ в системе
2Ќ2ќ(г)+ 2ќ2(г) ⇄ 2Ќ2ќ2 (г), ∆ Ќ = Ц 483,7 кƒж
при понижении температуры и уменьшении общего давлени€.
– е ш е н и е. ¬ли€ние внешнего воздействи€ на состо€ние химического равновеси€ в системах, в которых протекают обратимые реакции, оцениваетс€ с помощью принципа Ће Ўателье-Ѕрауна.
1) –ассматриваем вли€ние температуры на состо€ние равновеси€ в данной системе. “ермохимическое уравнение реакции показывает, что ∆Ќ < 0, следовательно, пр€ма€ реакци€ экзотермическа€. ѕри понижении температуры согласно принципу Ће Ўателье-Ѕрауна система будет стремитьс€ ослабить внешнее воздействие (т.е. повысить “), а это возможно за счет реакции, протекающей с выделением теплоты, т.е. экзотермической реакции. —ледовательно, понижение температуры приведет к смещению равновеси€ в направлении протекани€ пр€мой реакции.
2) ќцениваем вли€ние изменени€ давлени€ на состо€ние равновеси€ в данной системе. »з уравнени€ реакции видно, что пр€ма€ реакци€ сопровождаетс€ уменьшением объема системы 2V (Ќ2ќ) + 2V (ќ2) ≠ 2V (Ќ2ќ2), т.е. ∆V ≠ 0. ѕоэтому общее давление (–общ) будет вли€ть на состо€ние равновеси€ в данной системе. ”меньшение –общ согласно принципу Ће Ўателье-Ѕрауна вызовет противодействие системы (увеличение –общ) за счет смещени€ равновеси€ в сторону протекани€ реакции, сопровождающейс€ увеличением –общ, т.е. в сторону обратной реакции. ƒействительно, обратна€ реакци€ приводит к увеличению числа моль газообразных веществ в системе, а это значит, что при протекании обратной реакции –общ в системе увеличиваетс€.
|
|
|
1.7. Ёлектролитическа€ диссоциаци€. –еакции ионного обмена
Ёлектролитическа€ диссоциаци€ Ц это процесс распада молекул электролита на ионы под действием пол€рных молекул растворител€.
Ёлектролиты Ц это вещества, расплавы или водные растворы которых провод€т электрический ток. ним относ€тс€ растворы кислот, расплавы и растворы щелочей и солей. Ќеэлектролиты Ц это вещества, которые не провод€т электрический ток. ним относ€тс€ многие органические вещества.
Ёлектролиты, которые практически полностью диссоциируют на ионы, называютс€ сильными; электролиты, которые частично диссоциируют на ионы, называютс€ слабыми. ƒл€ количественной оценки полноты диссоциации введено пон€тие степени диссоциации. —тепенью диссоциации электролита называют отношение числа молекул, распавшихс€ на ионы, к общему числу молекул, наход€щихс€ в растворе.
ќбычно степень диссоциации (α) выражают в дол€х единицы или %:
 , (12)
, (12)
где n Ц число частиц, подвергшихс€ электролитической диссоциации;
n 0 Ц общее число частиц в растворе.
—ильные электролиты Ц почти все соли, растворимые основани€ (NaOH, KOH, Ba(OH)2 и др.), неорганические кислоты (H2SO4, HCl, HNO3, HBr, HI и др).
—лабые электролиты Ц нерастворимые основани€ и NH4OH, неорганические кислоты (H2CO3,, H2S, HNO2, H3PO4 и др.), органические кислоты и вода H2O.
—ильные электролиты диссоциируют на ионы практически нацело (т.е. процесс диссоциации €вл€етс€ необратимым) и одностадийно:
HCl = H+ + ClЦ H2SO4 = 2H+ + SO42Ц
—лабые электролиты диссоциируют частично (т.е. процесс диссоциации€вл€етс€ обратимым) и ступенчато. Ќапример, дл€ многоосновных кислот на каждой стадии происходит отрыв одного иона водорода:
1. H2SO3 ⇄ H+ + HSO3- 2. HSO3- ⇄ H+ + SO32-
“аким образом, число стадий многоосновных кислот определ€етс€ основностью кислоты (числом ионов водорода), а число стадий многокислотных оснований будет определ€тьс€ кислотностью основани€ (или числом гидроксильных групп): NH4OH ⇄ NH4+ + OHЦ. ѕроцесс электролитической диссоциации завершаетс€ установлением в системе состо€ни€ химического равновеси€, которое характеризуетс€ константой равновеси€: 
онстанта равновеси€ процесса электролитической диссоциации называетс€ константой диссоциации Ц ƒ. онстанта диссоциации зависит от природы электролита, природы растворител€, температуры, но не зависит от концентрации электролита.
ћежду ƒ и α существует количественна€ св€зь:
 (13)
(13)
—оотношение (13) называют законом разбавлени€ ќствальда: степень диссоциации слабого электролита возрастает с разбавлением раствора.
ƒл€ слабых электролитов, когда α << 1, ƒ = α2—.
¬ода €вл€етс€ слабым электролитом, поэтому диссоциирует обратимо:
H 2O ⇄ H+ + OHЦ ∆H = +56,5кƒж/моль
онстанта диссоциации воды: 
—тепень диссоциации воды очень мала (это очень слабый электролит). “ак как вода присутствует в большом избытке, то ее концентраци€ может считатьс€ величиной посто€нной и составл€ет  , тогда
, тогда
ƒ [ H2O] = [H+]∙[OH-] = 55,6∙1,8∙10-16 = 10-14
[ H+]∙[OH-] = 10-14 = KW Ц ионное произведение воды
“ак как в воде концентрации катионов водорода и гидроксид-ионов равны, то: [ H+] = [OH-] =  .
.
–астворение в воде других веществ (кислот, оснований, солей) измен€ет концентрацию ионов Ќ+ или ќЌЦ, а их произведение всегда остаетс€ посто€нным и равным 10-14 при “=250—. онцентраци€ ионов Ќ+ может служить мерой кислотности или щелочности раствора. ќбычно дл€ этой цели используетс€ водородный показатель: pH = -lg[H+]. “аким образом, водородный показатель Ц это дес€тичный логарифм концентрации ионов водорода, вз€тый с обратным знаком.
|
|
|






