ѕ–»ћ≈–џ –≈Ў≈Ќ»я «јƒј„
ѕ 5. Ёнергетические эффекты химических процессов.
“ермохимические расчеты. –асчет теплоты сгорани€ топлива.
ѕример 1. “ермохимическое уравнение реакции. Ёнтальпии образовани€ вещества.
Ќапишите термохимическое уравнение реакции, которое отвечает энтальпии образовани€ H2SO4(ж).
–ешение. ’имические уравнени€ реакций, в которых указаны агрегатные состо€ни€ веществ и тепловые эффекты, называютс€ термохимическими уравнени€ми.
—тандартной энтальпией (теплотой) образовани€ веществаназываетс€ энтальпи€ образовани€ 1 мол€ вещества из простых веществ, если все участники реакции наход€тс€ в стандартных состо€ни€х (с.с. при  и
и 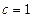 моль/л), (Δ fЌ 0298 см.ѕриложение 2, [1]
моль/л), (Δ fЌ 0298 см.ѕриложение 2, [1]
ѕо определению, искомое уравнение должно состо€ть из исходных простых веществ, устойчивых в стандартном состо€нии:
H2(г) + 2O2(г) + S(к) = H2SO4(ж), Δ fЌ 0298 H2SO4 = −811,3 кƒж/моль.
»з табл. (ѕриложение 2, [1]).
ќтрицательное значение энтальпии образовани€ соединени€ указывает на то, что в результате образовани€ этого соединени€ из простых веществ теплота выдел€етс€.
ѕример 2. ¬заимосв€зь изменени€ энтальпии и внутренней энергии в результате реакции.
–ассчитайте изменение внутренней энергии системы при изохорно-изотермическом протекании реакции
—(к) + —ќ2(г) = 2—ќ(г)
при с.с. и 298 .
–ешение. ¬нутренн€€ энерги€ системы U Ц одна из термодинамических функций состо€ни€ системы. »зменение внутренней энергии в результате какого-либо процесса (например, химической реакции) Δ rU равно тепловому эффекту этого процесса, протекающего в изохорно-изотермических услови€х (QV,T), если при этом не совершаетс€ никакой работы:
QV,T = - Δ rU.
Ёнтальпи€ Ќ Ц одна из термодинамических функций состо€ни€ системы.
»зменение энтальпии в результате какого-либо процесса (химической реакции) Δ rЌ равно тепловому эффекту этого процесса, протекающего в изобарно-изотермических услови€х (Qp,T), когда единственным видом работы €вл€етс€ работа расширени€ газа:
Qp,T = - Δ rH“.
»зменение энтальпии и внутренней энергии в результате реакции св€заны соотношением:
Δ rЌ =Δ rU + ∆ν RT,
где ∆ν Ц разница между числом молей газообразных продуктов и числом молей газообразных исходных веществ.
≈сли Δ rЌ < 0, то процесс идет с выделением тепла в окружающую среду и реакци€ называетс€ экзотермической; если Δ rЌ > 0, теплота в результате реакции поглощаетс€ и реакци€ называетс€ эндотермической.
—огласно данным (ѕриложение 2, [1]):
¬ещество Δ fЌ 0298,кƒж/моль
—(графит) 0
—ќ2 (г) −393,5
—ќ (г) −110,5
ƒл€ расчета энтальпии химической реакции при с.с. и 298 воспользуемс€ следствием из закона √есса:
Δ rЌ 0298= ∑ν i Δ f Ќ 0298, продуктов − ∑ν j Δ f Ќ 0298, исх веществ ;
Δ rЌ 0298 = 2Δ fЌ 0298—ќг − Δ fЌ 0298—к − Δ fЌ 0298—ќ2г = 2(−110,5) Ц 0 Ц (−393,5) =
|
|
|
= 172,5 кƒж. “ак как Δ rЌ 0298 > 0, данна€ реакци€ эндотермическа€.
»зменение энтальпии и внутренней энергии в результате реакции св€заны соотношением:
Δ rЌ =Δ rU + ∆ν RT.
“огда ∆ rU 0298 = ∆ rH 0298 Ц ∆ν RT =
= 172,5 Ц 1Ј8,31Ј298Ј10−3 = 170,02 кƒж. (–азница между числом молей газообразных продуктов и исходных веществ равна ∆ν = 2 − 1 = 1 моль).
”ниверсальна€ газова€ посто€нна€ R =8,31 ƒж/моль. .
ѕример 3. «акон √есса. ћетод циклов.
¬ычислите тепловой эффект образовани€ NH3 из простых веществ при стандартном состо€нии по тепловым эффектам реакций:
2H2 + O2 = 2H2O(ж);; D r H0 1 = - 571,68 кƒж (1)
4 NH3 + 3 O2 = 6 H2O(;ж) + 2 N2 ; D r H0 2 = - 1530,28 кƒж (2).
–ешение. «акон √есса: “епловой эффект реакции зависит от природы и состо€ни€ исходных веществ и конечных продуктов, но не зависит от пути реакции, т.е. от числа и характера промежуточных стадий.
«апишем уравнение реакции, тепловой эффект которой необходимо определить:
½N2 + 3/2Ќ2 = NH3; D r H0 3 = Df H0NH3=?;(3).
¬ уравнени€ (1) и (2) вход€т Ќ2ќ (ж) и ќ2, которые не вход€т в уравнение (3), поэтому, чтобы исключить их из уравнений (1) и (2), умножим уравнение (1) на 3 и вычтем из него ур (2):
6Ќ2 + 3ќ2 Ц 4NЌ3 Ц 3ќ2 = 6Ќ2ќ (ж) Ц 6Ќ2ќ (ж) Ц 2N2 (г) (4).
ѕосле преобразовани€ уравнени€ (4) и делени€ его на 4 получаем искомое уравнение (3). јналогичные действи€ проделаем с тепловыми эффектами:
(3D rH0 1Ц D r H02)/4 = D rH0 3. ¬ результате получаем:
D r H0 3 = [3(Ц571,68) Ц (Ц 1530,28)] / 4 = Ц 46,19 (кƒж);
т.е. ∆ f Ќ0 NH3 = Ц 46,19 кƒж/моль.
ѕример 4. «акон √есса. ћетод циклов.
ќпределите стандартный тепловой эффект реакции:
—(к) + —ќ2(г) = 2—ќ(г); (1),
если известны тепловые эффекты двух других реакций:
2—(к) + ќ2(г) = 2—ќ(г); ∆ rH 0298 = −221 кƒж (2)
и
2—ќ(г) + ќ2(г) = 2—ќ2(г); ∆ rH 0298 = −566,1 кƒж (3).
–ешение. «акон √есса: “епловой эффект реакции зависит от природы и состо€ни€ исходных веществ и конечных продуктов, но не зависит от пути реакции, т.е. от числа и характера промежуточных стадий.
ƒл€ того, чтобы получить уравнение реакции (1), нужно из уравнени€ реакции (2) вычесть уравнение реакции (3) и разделить полученное выражение на 2. “е же действи€ следует проделать и с величинами ∆ rH 0.
»меем ∆ rH 01 = (∆ rH 02 − ∆ rH 03)/2 = [−221 Ц (−566,1)]/2 = 172,5 кƒж.
ѕример 5. –асчет стандартной энтальпии химической реакции при различных температурах.
–ассчитайте стандартный тепловой эффект изобарно-изотермической реакции
—(к) + —ќ2(г) = 2—ќ(г)
при 298 и при 1000 , счита€ посто€нными теплоемкости реагентов в данном температурном интервале.
–ешение. «апишем значени€ стандартной энтальпии образовани€ и стандартной теплоемкости веществ согласно данным (ѕриложение 2, [1]):
¬ещество Δ fЌ 0298,кƒж/моль — 0 р 298, ƒж/моль.
—(графит) 0 8,54
—ќ2 (г) −393,5 37,41
—ќ (г) −110,5 29,14
ƒл€ расчета энтальпии химической реакции при с.с. и 298 воспользуемс€ следствием из закона √есса:
Δ rЌ 0298= ∑ν i Δ f Ќ 0298, продуктов − ∑ν j Δ f Ќ 0298, исх веществ ;
Δ rЌ 0298 = 2Δ fЌ 0298—ќг − Δ fЌ 0298—к − Δ fЌ 0298—ќ2г = 2(−110,5) Ц 0 Ц (−393,5) =
|
|
|
= 172,5 кƒж. “ак как Δ rЌ 0298 > 0, данна€ реакци€ эндотермическа€.
«ависимость энтальпии реакции от температуры в области 298 ÷ “, в которой нет фазовых переходов, в интегральном виде выражаетс€ уравнением ирхгофа:
“
∆ rH 0 T = ∆ rH 0298 + ∫ ∆ r— 0 pdT,
298
где ∆rЌ0 298 − стандартна€ энтальпи€ реакции при 298 ; ∆r—p0 − изменение стандартной теплоемкости системы в ходе реакции.
≈сли пренебречь зависимостью —р реагирующих веществ от “, то есть
∆r—0p = const, то имеем приближение в виде:
∆rЌ0“ = ∆rЌ0298 + ∆r—0р(“ − 298),
рассчитать ∆—0р можно по справочным данным, как разность стандартных изобарных теплоемкостей продуктов и исходных веществ при 298 .
–ассчитаем изменение теплоемкости системы в результате реакции:
∆ r— 0 р = 2 — 0 р 298—ќг Ц — 0 р 298—к Ц — 0 р 298—ќ2г = 2(29,14) Ц 8,54 Ц 37,41 = 12,33 ƒж/ .
ѕо уравнению
∆rЌ0“ = ∆rЌ0298 + ∆r—0р(“ − 298)
рассчитаем энтальпию химической реакции при с.с. и 1000 :
∆ rЌ 01000 = 172,5 + 12,33Ј10−3(1000 − 298) = 172,5 + 8,66 = 181,16 кƒж.
»з расчетов следует, что при изменении температуры на 702 увеличение теплового эффекта составл€ет всего около 5%.
ѕример 6. –асчет тепла, выделенного в результате химической реакции.
ќпределите энтальпию процесса конденсации воды. –ассчитайте, сколько тепла выделитс€ при образовании 1 м3 жидкой воды из пара при стандартных состо€ни€х вещества.
–ешение. онденсаци€ Ц это процесс перехода из газообразного состо€ни€ в жидкое, который относитс€ к фазовым превращени€м. «апишем уравнение конденсации дл€ воды:
Ќ2ќ(г) = Ќ2ќ(ж).
Ёнтальпию этого превращени€ рассчитываем по следствию из закона √есса, использу€ данные табл. (ѕриложение 2, [1]):
Δ rЌ 0298= ∑ν i Δ f Ќ 0298, продуктов − ∑ν j Δ f Ќ 0298, исх веществ ;
∆ rЌ 0298 = ∆ fЌ 0298Ќ2ќж Ц ∆ fЌ 0298Ќ2ќг = −285,84 Ц (−241,84) = −44 кƒж.
“ак как Δ rЌ 0298 < 0, процесс конденсации экзотермический.
Ёнтальпии химической реакции Δ rЌ равна тепловому эффекту этого процесса, протекающего в изобарно-изотермических услови€х (Qp,T):
Qp,T = - Δ rH“.
ќпределим количество вещества Ќ2ќ, содержащеес€ в 1 м3. “ак как плотность воды ρ = 1 г/см3, то масса 1м3 воды: m = ρ V = 1Ј106 г.
оличество молей Ќ2ќ νЌ2ќ = m /MЌ2ќ = 106/18 = 5,56Ј104 моль.
ѕоскольку при конденсации 1 мол€ воды выдел€етс€ 44 кƒж тепла, то при конденсации 5,56Ј104 молей выделитс€
Q = 44Ј5,56Ј104 = 2,45Ј106 кƒж тепла.
ѕример 7. –асчет удельной теплоты сгорани€ газового топлива.
¬ычислить удельную теплоту сгорани€ газового топлива, содержащего 60% Ќ2 и 40% —Ќ4, если известны теплоты сгорани€ газов:  = Ц286 кƒж/моль;
= Ц286 кƒж/моль;  = Ц890 кƒж/моль.
= Ц890 кƒж/моль.
–ешение. “ермохимические уравнени€ процессов сгорани€ водорода и метана:
Ќ2(г) + ½ќ2(г) → Ќ2ќ(ж);  = Ц286 кƒж/моль;
= Ц286 кƒж/моль;
—Ќ4(г) + 2ќ2(г) → —ќ2(г) + 2Ќ2ќ(ж);  = Ц890 кƒж/моль.
= Ц890 кƒж/моль.
“еплота сгорани€ топлива может быть рассчитана по следствию из закона √есса:
Δ rЌ 0 “ = ∑ν i Δ fЌi 0 продуктов − ∑ν j Δ fЌj 0исх веществ .
”дельна€ теплота сгорани€ газа Q“ равна количеству теплоты, выдел€ющейс€ при сгорании 1м3 газообразного вещества до образовани€ высших оксидов.
ѕо условию задачи 1м3 данного газового топлива содержит 600 л Ќ2 и 400 л —Ќ4, что составл€ет соответственно 600/22,4 молей Ќ2 и 400/22,4 молей —Ќ4. (использовали мол€рный объем газа при нормальных услови€х 22,4 л/моль).
¬ соответствии с уравнением:
Q т = ЦD Ќ сгор×(1000/22,4),
рассчитаем удельную теплоту сгорани€ газового топлива:
Q т = 286×(600/22,4) + 890×(400/22,4) = 7660,7 + 15892,8 = 23550 кƒж/м3.
ѕ 6. –асчет энтропии химической реакции. –асчет энергии √иббса.
ќпределение термодинамической возможности самопроизвольного протекани€ химической реакции в заданном направлении. ќпределение температурной области самопроизвольного протекани€ химической реакции.
|
|
|
ѕример 1. –асчет стандартной энтропии химической реакции при различных температурах.
–ассчитайте стандартную энтропию химической реакции
—(к) + —ќ2(г) = 2—ќ(г)
при 298 и при 1000 , счита€ посто€нными теплоемкости компонентов в рассматриваемом температурном интервале.
–ешение. Ёнтропи€ S Ц функци€ состо€ни€ системы, котора€ служит мерой неупор€доченности этой системы, измер€етс€ в ƒж/(мольЈ ).
—огласно данным табл. (см. ѕриложение IV, [2]; ѕриложение 4, [3]):
¬ещество f S 0298, ƒж/(мольЈK) — 0 р 298, ƒж/(мольЈ )
—(графит) 5,74 8,54
—ќ2 (г) 213,68 37,41
—ќ (г) 197,54 29,14
—тандартна€ энтропи€ химической реакции при с.с. ( и
и 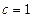 моль/л) и 298 , обозначаетс€ ∆ rS 0298 и рассчитываетс€ по уравнению (следствие из закона √есса):
моль/л) и 298 , обозначаетс€ ∆ rS 0298 и рассчитываетс€ по уравнению (следствие из закона √есса):
Δ rS 0298 = ∑ν i fS 0298, i продуктов − ∑ν j fS 0298, j исх. веществ,
где f S 0298 - стандартна€ энтропи€ образовани€ веществ при 298 (см. ѕриложение IV, [2]; ѕриложение 4, [3]); ν i - стехиометрические коэффициенты уравнени€ химической реакции.
–ассчитаем:
Δ rS 0298 = 2 S 0298,—ќг − S 0298,—к − S 0298,—ќ2г = 2(197,54) Ц 5,74 Ц 213,68 =
= 175,66 ƒж/K.
“о есть данна€ реакци€ при 298 идет с увеличением энтропии, что и следовало ожидать, так как число молей газообразных веществ в результате реакции увеличиваетс€.
«ависимость энтропии реакции от температуры в области 298 ÷ “, в которой нет фазовых переходов, в интегральном виде выражаетс€ уравнением
“
∆ rS 0 T = ∆ rS 0298 + ∫ (∆ r— 0 p/“) dT,
298
где ∆ rS 0298 − стандартна€ энтропи€ реакции при 298 ; ∆ r— 0 p − изменение суммарной стандартной теплоемкости системы в ходе реакции.
–ассчитаем изменение стандартной теплоемкости системы в результате реакции:
∆ r — 0 р = 2 —0р 298—ќг Ц —0р 298—к Ц —0р 298—ќ2г = 2.(29,14)Ц8,54Ц37,41 =12,33 ƒж/ .
ƒл€ многих реакций можно пренебречь зависимостью —р реагирующих веществ от “, то есть ∆ r— 0 p = const. “огда имеем приближение в виде:
∆ rS 0 T = ∆ rS 0298 + ∆ r— 0 р (ln “ − ln298).
ѕо этому уравнению рассчитаем
∆ rS 01000 = 175,66 + 12,33ln(1000/298) = 175,66 + 14,93 = 190,59 ƒж/ .
ак следует из расчетов, увеличение энтропии реакции, обусловленное ростом суммарной теплоемкости системы, при изменении температуры на 702 составл€ет около 8,5%.
ѕример 2. –асчет энергии √иббса химической реакции при стандартном состо€нии и различных температурах. ¬озможное направление самопроизвольного протекани€ процесса.
–ассчитайте стандартную энергию √иббса химической реакции
—(к) + —ќ2(г) = 2—ќ(г)
при 298 и при 1000 . —делайте вывод о возможности самопроизвольного протекани€ этой реакции при указанных температурах и стандартных состо€ни€х всех компонентов.
–ешение. Ќаправление любого процесса определ€етс€ соотношением энтальпийного ∆ rЌ и энтропийного “ ∆ rS факторов реакции.
Ёнерги€ √иббса,или свободна€ энерги€ G - функци€ состо€ни€ системы, учитывающа€ совместное вли€ние этих факторов, G = H Ц TS.
ќднозначный критерий возможности самопроизвольного протекани€ реакции в пр€мом направлении в изобарно-изотермических услови€х:
∆ rG“ = (∆ rH“ Ц T ∆ rS“) < 0.
1). ƒл€ начала, будем считать, чтоэнтальпи€ и энтропи€ реакции не завис€т от температуры. ¬ широком интервале температур в первом приближении можно считать неизменной величину стандартной теплоемкости веществ в ходе химической реакции ∆ r— 0 p = 0, тогда
|
|
|
∆ rЌ 0 T = ∆ rЌ 0298 и ∆ rS 0 T = ∆ rS 0298 .
ƒл€ расчета применим приближенную формулу расчета энергии √иббса
∆ rG 0 “ = ∆ rH 0298 − T ∆ rS 0298.
»спользуем данные, полученные в ѕримере 1. (Δ rS 0298 = 175,66 ƒж/K) и в ѕримере 5. предыдущего практического зан€ти€ (Δ rЌ 0298 = 172,5 кƒж).
∆ rG 0298 = 172,5 Ц 298 Ј 175,66Ј10−3 = 120,15 кƒж, то есть ∆ rG 0298> 0;
∆ rG 01000 = 172,5 Ц 1000 Ј 175,66 Ј 10−3 = −3,16 кƒж, то есть ∆ rG 01000 < 0.
“аким образом, при 298 и стандартных состо€ни€х веществ самопроизвольное протекание указанной реакции в пр€мом направлении невозможно (энтальпийный фактор не способствует самопроизвольному протеканию пр€мой реакции и определ€ет знак энергии √иббса реакции при низких температурах). ѕри высоких температурах определ€ющим становитс€ энтропийный фактор реакции, он определ€ет отрицательное значение энергии √иббса реакции при 1000 и, следовательно, возможность самопроизвольного протекани€ реакции при этой температуре и стандартных состо€ни€х компонентов.
2). ƒл€ более точного расчета ∆ rG 01000сначала рассчитаем изменение стандартной теплоемкости системы в ходе реакции:
∆ r — 0 р = 2 —0р 298—ќг Ц —0р 298—к Ц —0р 298—ќ2г = 2.(29,14)Ц8,54Ц37,41 =12,33 ƒж/ .
«начение стандартной ∆rG 0 “ дл€ любой температуры “ можно в общем случае рассчитать с использованием справочных данных по уравнению:
“ “
∆ rG 0 “ = ∆ rH 0 “ −T ∆ rS 0 “ = ∆ rH 0298 + ∫ ∆ r— 0 pdT + “ ∆ rS 0298 + “ ∫ (∆ r— 0 p/T) dT.
298 298
ƒл€ многих реакций можно пренебречь зависимостью —р реагирующих веществ от “, то есть ∆ r— 0 p = const. “огда имеем приближение в виде:
∆ rG 0 T = ∆ rG 0298 + ∆ r— 0 р (“ − 298) - T . ∆ r— 0 р (ln “ − ln298).
ƒл€ расчета ∆ rG 01000используем это приближение:
∆ rG 01000= ∆ rG 0298 + ∆ r— 0 р (“ − 298) - T . ∆ r— 0 р (ln “ − ln298) =
= 120150 +12,33. (1000-298) Ц 1000.12,33(ln1000 − ln298) =
= -9430 ƒж = -9,43 кƒж, то есть ∆ rG 01000 < 0.
ак видно, имеетс€ различие в значени€х стандартной энергии √иббса ∆ rG 0 T, рассчитанной по разным методикам.
ѕример 3. ќпределение температурной области самопроизвольного протекани€ реакции.
ќпределите температурную область самопроизвольного протекани€ реакции
—(к) + —ќ2(г) = 2—ќ(г)
при стандартных состо€ни€х компонентов.
–ешение. –еакци€ может протекать самопроизвольно при стандартных состо€ни€х компонентов в определенной области температур, дл€ которых ∆rG 0 “ < 0.„тобы найти эту область температур нужно определить граничную температуру (температуру равновеси€), при которой значение ∆rG 0 “ мен€ет знак, то есть необходимо решить неравенство относительно “:
∆ rG 0 “ = ∆ rH 0298 + ∫ “ 298∆ r— 0 pdT + “ ∆ rS 0298 + “ ∫ “ 298(∆ r— 0 p/T) dT < 0.
≈сли пренебречь зависимостью ∆ rH 0 и ∆ rS 0от температуры, то граничную температуру (температуру равновеси€) можно определить из приближенного неравенства:
∆ rG 0 “ = ∆ rH 0298 − T ∆ rS 0298< 0.
ѕодставл€€ в это выражение значени€ Δ rЌ 0298 = 172,5 кƒж и
Δ rS 0298 = 175,66 ƒж/K, (см. ѕример 3), получаем:
∆ rG 0 “ =(172,5 Ц “ Ј 175,66Ј10−3) < 0.
ќткуда “ > 982 . ¬ерхним пределом искомой температурной области €вл€етс€ предел существовани€ наименее устойчивого компонента реакции, который находитс€ из справочных данных.
¬ещество “емпературный интервал,






