¬ариант є1
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) серы; б) марганца.
2. —оставьте формулы оксидов и гидроксидов элементов второго периода, отвечающих их высшей степени окислени€. ак измен€етс€ кислотно-основной характер этих соединений?
3. ƒайте определение электроотрицательности. ак измен€етс€ электроотрицательность р-элементов в V≤≤-ј подгруппе? ак измен€етс€ их окислительна€ активность и почему?
4. Ќапишите электронные формулы атомов Zn и Cl и соответствующих ионов: Zn2+, Cl‾
¬ариант є2
1.»сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) железа; б) хлора.
2. —оставьте формулы оксидов и гидроксидов элементов ≤≤≤-ј подгруппы. ак измен€етс€ кислотно-основной характер этих соединений и почему?
3. ƒайте определение энергии ионизации. ак измен€етс€ восстановительна€ активность элементов ≤≤ - ј подгруппы и р-элементов ≤≤≤ периода.
4. Ќапишите электронные формулы атомов —а и N и соответствующих ионов: Ca2+, N3‾.
¬ариант є3
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) хрома; б) азота.
2. акую низшую и высшую степень окислени€ про€вл€ют углерод, фосфор, сера и йод? —оставьте формулы соединений данных элементов, отвечающих этим степен€м окислени€. ак измен€етс€ их окислительна€ активность?
3. „то характеризует орбитальное квантовое число? акие значени€ оно принимает? акие типы электронных орбиталей вам известны?
4. акое состо€ние атома называетс€ возбужденным? »зобразите распределение электронов по квантовым €чейкам в нормальном и возбужденном состо€нии атома фосфора.
¬ариант є4
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) молибдена; б) углерода.
2. акой из элементов четвертого периода Ц ванадий или мышь€к Ц обладает более выраженными металлическими свойствами? Ќапишите формулы кислородных соединений этих элементов, укажите их характер.
3. „то называетс€ сродством к электрону? ак измен€етс€ окислительна€ активность неметаллов V≤-ј подгруппы?
4. Ќапишите электронные формулы атомов Mn, Cl и соответствующих ионов: Mn4+, S2‾.
¬ариант є5
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) никел€; б) мышь€ка.
2. —оставьте формулы оксидов и гидроксидов элементов третьего периода, отвечающих их высшей степени окислени€. ак измен€етс€ кислотно-основной характер этих соединений?
3. ак измен€етс€ электроотрицательность и окислительна€ активность элементов ≤V-ј подгруппы? —оставьте формулы водородных соединений элементов этой группы.
4. Ќапишите электронные формулы атомов Fe и Se и соответствующих ионов: Fe6+, Se2‾.
¬ариант є6
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) цинка; б) кремни€.
2. ” какого из элементов четвертого периода Ц хрома или селена Ц сильнее выражены металлические свойства? ѕочему? —оставьте формулы оксидов и гидроксидов, укажите их характер.
|
|
|
3. ак измен€етс€ восстановительна€ активность элементов третьего периода? ѕочему? —оставьте формулы оксидов этих элементов.
4. Ќапишите электронные формулы атомов Co и Cl и соответствующих ионов: Co2+, Cl3+.
¬ариант є7
1.»сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) алюмини€; б) германи€.
2. ћарганец образует соединени€, в которых про€вл€ет степени окислени€: 2+, 3+, 4+, 6+, 7+. —оставьте формулы его оксидов и гидроксидов, отвечающих этим степен€м окислени€, укажите их характер.
3. ак измен€етс€ окислительна€ активность элементов ≤V-ј подгруппы, р-элементов V периода? ƒайте мотивированный ответ.
4. Ќапишите электронные формулы ионов: P5+, Cr3+.
¬ариант є8
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) ванади€; б) йода.
2. »сход€ из величины степени окислени€ атомов, определите, какой из двух гидроксидов €вл€етс€ более сильным основанием: а) Fe(OH)2 или Fe(OH)3,
б) Mn(OH)2 или Mn(OH)4, в) Sn(OH)2 или Sn(OH)4?
3. ак измен€етс€ радиус атомов у элементов V-ј подгруппы, р-элементов четвертого периода? ак измен€етс€ их окислительна€ активность?
4. Ќапишите электронные формулы ионов: Br5+, Co2+.
¬ариант є9
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) сурьмы; б) меди.
2. акую высшую и низшую степени окислени€ про€вл€ет кремний, мышь€к, селен, хлор? —оставьте формулы соединений данных элементов, отвечающих этим степен€м окислени€.
3. ак измен€етс€ восстановительна€ активность sЦэлементов первой группы? —оставьте формулы оксидов и гидроксидов элементов этой группы. ак измен€етс€ основной характер этих гидроксидов?
4. Ќапишите электронные формулы атомов и соответствующих ионов: Mn7+, I‾.
¬ариант є10
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) молибдена; б) брома.
2. ” какого из р-элементов п€той группы периодической системы Ц фосфора или сурьмы Ц сильнее выражены неметаллические свойства? —оставьте формулы водородных и кислородных соединений этих элементов.
3. ак измен€етс€ восстановительна€ активность металлов II-ј подгруппы? ак измен€етс€ основной характер оксидов и гидроксидов этих металлов?
4. Ќапишите электронные формулы атомов Ni, P и соответствующих ионов: Ni3+, P3‾.
¬ариант є11
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) железа; б) селена.
2. акую низшую степень окислени€ про€вл€ют: хлор, сера, азот, углерод? —оставьте формулы водородных соединений этих элементов. ак измен€етс€ восстановительна€ способность этих соединений?
3. ”кажите самый активный металл и неметалл п€того периода. Ќапишите формулы их оксидов и укажите их характер.
4. Ќапишите электронные формулы атомов Br, Mn и соответствующих ионов: Br‾, Mn6+.
¬ариант є12
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) вольфрама; б) фосфора.
2. ак измен€ютс€ свойства рЦэлементов V-ј подгруппы? —оставьте формулы соединений, отвечающих их низшей и высшей степен€м окислени€.
3. ’лор образует соединени€, в которых он про€вл€ет степени окислени€ 1+, 3+, 5+, 7+. —оставьте формулы его оксидов и гидроксидов, отвечающих этим степен€м окислени€. ак измен€етс€ сила кислот?
|
|
|
4. Ќапишите электронные формулы атомов Al, As и соответствующих ионов: Al3+, As3‾.
¬ариант є13
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) марганца; б) германи€.
2. ак измен€етс€ кислотно-основной характер оксидов и гидроксидов хрома, соответствующих степен€м окислени€ 2+, 3+, 6+?
3. ак измен€етс€ окислительна€ активность, неметаллические свойства элементов V≤≤-ј подгруппы? —оставьте формулы водородных соединений элементов этой подгруппы.
4. Ќапишите электронные формулы атомов Ca, Sn и соответствующих ионов: Ca2+, Sn4+.
¬ариант є14
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) бари€; б) брома.
2. Ќа каком основании хром и сера, фосфор и ванадий располагаютс€ в одной группе? ѕочему их помещают в разные подгруппы? —оставьте формулы высших оксидов этих элементов и определите их характер.
3. акое состо€ние атома называетс€ возбужденным? »зобразите распределение электронов по квантовым €чейкам в нормальном и возбужденном состо€ни€х атома серы.
4. Ќапишите электронные формулы ионов: Ge4‾, Pb2+.
¬ариант є15
1. »сход€ из положени€ элементов в периодической системе, дайте характеристику атомов: а) олова; б) фосфора.
2. „то характеризует спиновое квантовое число электрона? акова максимальна€ спин-валентность атома с распределением валентных электронов Е5s25p3?
3. ак измен€етс€ электроотрицательность, окислительна€ активность, неметаллические свойства р-элементов п€того периода? —оставьте формулы высших оксидов этих элементов.
4. Ќапишите электронные формулы ионов: Cr3+, Cl‾.
4. “≈ћј: "’имическа€ св€зь, строение молекул"
“еори€ химической св€зи относитс€ к числу основных вопросов курса химии, т. к. свойства веществ и их реакционна€ способность завис€т не только от состава, строени€ молекул, но и от типа химической св€зи между атомами. ѕри изучении этой темы необходимо:
- разобратьс€ в механизме образовани€ ионной, ковалентной и металлической св€зей;
- усвоить свойства и разновидности ковалентной св€зи;
- изучить особенности свойств соединений с различными типами химических св€зей;
- приобрести навыки в составлении электронных схем образовани€ молекул с различными типами химической св€зи.
1. ”кажите тип химической св€зи в молекулах соединений вашего варианта.
2. —оставьте электронные схемы образовани€ молекул с ковалентной св€зью. Ёлектроны, каких орбиталей атомов принимают участие в образовании ковалентной св€зи? ќпределите валентность и степени окислени€ атомов в молекуле.
3. »зобразите электронными уравнени€ми процессы образовани€ соединений с ионной св€зью.
¬арианты контрольных заданий в таблице є2
| єварианта | ‘ормулы соединений | ||||
| I2 | HI | CrCl3 | PbO | (H2O)n | |
| NH4Cl | BaI2 | AsH3 | K2O | F2 | |
| Cl2 | Mn2O3 | CH4 | ZnS | SiO2 | |
| Fe2O3 | N2 | NaBr | NH4+ | SO2 | |
| Al | MgO | N2 | H2S | FeCl3 | |
| PCl3 | HF | Mn2O3 | Cl2 | PI2 | |
| CO2 | H2S | PbCl4 | Fe2S3 | Br2 | |
| KOH | Br2 | ZnO | AlBr3 | H2O | |
| CuCl | HBr | CO2 | Cr2O3 | I2 | |
| O2 | CaS | Na2O | H2S | CCl4 | |
| (HF)n | SiBr4 | Cl2 | CuO | AlI3 | |
| Cu | F2 | Al2O3 | KBr | SiO2 | |
| Br2 | CH4 | NaOH | Cu2O | Al2S3 | |
| NH3 | SiO2 | MgBr2 | CrO | I2 | |
| HBr | Fe2O3 | (H2O)n | Cl2 | FeCl3 |
5. “≈ћј: " омплексные соединени€"
ѕри изучении этой темы необходимо:
- разобратьс€ в механизме донорно-акцепторной св€зи и особенност€х координационных соединений, их структуре, устойчивости и типах лиганд;
- уметь определ€ть координационное число иона-комплексообразовател€, его зар€д, а также зар€д комплексного иона; дать названи€ рассматриваемым комплексным соединени€м.
- приобрести навыки в составлении уравнений реакций образовани€ и диссоциации комплексных соединений;
|
|
|
- иметь представление о значении и применении минеральных, органических и органоминеральных комплексных соединений в сельском хоз€йстве.
1. «аполните пропуски в таблице є3 и напишите выражени€ дл€ констант диссоциации (нестойкости) комплексных ионов.
| є | омлексообразователь | Ћиганды | оординационное число | ¬нутренн€€ сфера комплекса | »оны внешней сферы | ‘ормула комплексного соединени€ |
| 2[Zn(OH)4] | ||||||
| Pt4+ | Cl─ | K+ | ||||
| [Cu(NH3)4]2+ | SO42─ | |||||
| Al3+ | OH─ | Na+ | ||||
| Cr3+ | H2O | Cl─ | ||||
| K[BF4] | ||||||
| [Zn(OH)4]2─ | Na+ | |||||
| Fe3+ | CN─ | K+ | ||||
| Au3+ | Cl─ | H+ | ||||
| K[Ag(CN)2] | ||||||
| [Co(NH3)6]3+ | Cl─ | |||||
| Ni2+ | H2O | SO42─ | ||||
| K2[Ni(CN)4] | ||||||
| Ag+ | NH3 | Cl─ | ||||
| [Al(OH)4]─ | K+ | |||||
| Na3[Co(NO2)6] | ||||||
| Pt2+ | NH3 | Cl─ | ||||
| [Ni(NH3)6]SO4 | ||||||
| [FeF6]4─ | K+ | |||||
| Na3[CoF6] | ||||||
| Al3+ | H2O | Cl─ | ||||
| K3[AlF6] | ||||||
| Sn2+ | OH─ | Na+ | ||||
| [Cu(H2O)4]2+ | NO3─ | |||||
| Na3[CoF6] | ||||||
| W4+ | CN─ | K+ | ||||
| Cu2+ | CN─ | Na+ | ||||
| [Ni(NH3)6]2+ | Br─ | |||||
| Na2[HgI4] | ||||||
| Ca2+ | NH3 | Cl─ |
2.ќпределите зар€д внутренней координационной сферы и составьте формулы двух веществ, в которые она входит. ќпределите координационное число комплексообразовател€:
1. [Co3+(NH3)6]; 9. [Co3+(NH3)2(NO2)4];
2. [Ni3+(H2O)4Cl2]; 10. [Pt4+Cl6];
3. [Zn2+ (OH) 4]; 11. [Fe2+ (CN) 6];
4. [Sb5+ (OH) 6]; 12. [Co3+F6];
5. [Co3+ (NH3)5Br]; 13. [Pt4+Br4];
6. [Fe3+F6]; 14. [Au3+Cl4];
7. [Co3+ (NH3)5SO4]; 15. [Co3+(NO2)6];
8. [Pt4+(NH3)4Cl];
3. —оставьте молекул€рные и ионные уравнени€ реакций, привод€щие к образованию одного малорастворимого вещества:
1. FeCl3+K4 [Fe (CN) 6] →
2. [Pt (NH3)6] Cl4+AgNO3 →
3. K2SO4+Na3 [Co (NO2)6] →
4. [Pt (NH3)3Cl3] Cl+Pb (NO3)2 →
5. FeSO4+K3 [Fe (CN) 6] →
6. NaCl+K [Sb (OH) 6] →
7. [Co (NH3)5SO4] Cl+AgNO3 →
8. [Cu (NH3)4] SO4+BaCl2 →
9. [Co (NH3)5SO4] Br+AgNO3 →
10. [Co (NH3)5Br] SO4+BaCl2 →
11. CuSO4+K4 [Fe (CN) 6] →
12. Na3 [Co (CN) 6] +FeSO4 →
13. K3 [Fe (CN) 6] +AgNO3 →
14. K [Sb (OH) 6] +Na2SO4 →
15. Na3 [Co (CN) 6] +KCl →
4. ѕредставьте указанные в вашем варианте вещества в виде комплексных соединений:
1. KCN∙AgCN; 9. 2KCN∙Ni(CN)2;
|
|
|
2. Co (NO3)3∙6NH3; 10. 2KCl∙PtCl4;
3. 2KCN∙Cu (CN) 2; 11. AgCl∙2NH3;
4. CoCl3∙6NH3; 12. 4KCN∙W (CN) 4;
5. 2KSCN∙Co (SCN) 2; 13. 2NH3∙PtCl4;
6. 4KCN∙Fe (CN) 2; 14. CoF3∙3NaF;
7. KCl∙AuCl3; 15. CrCl3∙6H2O;
8. CuCl2∙4NH3;
6. “≈ћј " Ёнергетика химических процессов "
¬опросы энергетики химических процессов €вл€ютс€ предметом изучени€ отдельного раздела химии, называемого химической термодинамикой. ќсновными задачами этого раздела €вл€ютс€ определение тепловых эффектов химических реакций, возможности самопроизвольного их течени€ в заданных услови€х, а также вы€вление наиболее рациональных условий, обеспечивающих эффективный ход процесса.
ѕри изучении данной темы студент должен:
- ознакомитьс€ с основными термодинамическими пон€ти€ми и величинами;
- пон€ть сущность и практическую значимость первого закона термодинамики, закона √есса, второго закона термодинамики;
- разобратьс€ в практических расчетах изобарного теплового эффекта при стандартных услови€х ∆Ќ0298, изменени€ энтропии ∆S0298 и энергии √иббса ∆G0298 дл€ любой химической реакции;
- научитьс€ на основе расчетов ∆Ќ, ∆S, ∆G судить о выделении или поглощении теплоты в процессе реакции, а также возможности ее самопроизвольного протекани€.
«адание дл€ самосто€тельной расчетной работы
1. »спользу€ справочные данные таблицы є5, определите изобарный тепловой эффект ∆Ќ0298 химической реакции вашего варианта (таблица є4). —делайте вывод о выделении или поглощении теплоты в процессе реакции.
2. ќпределите изменение энтропии ∆S0298 в ходе химической реакции, протекающей при стандартных услови€х в идеальном газообразном состо€нии. ќбъ€сните знак изменени€ ∆S0298 в результате данной реакции.
3. ќпределите изменение энергии √иббса ∆G0298 в ходе химической реакции, использу€ справочные данные таблицы є 5. ѕо знаку изменени€ энергии √иббса ∆G0298 сделайте вывод о возможности самопроизвольного протекани€.
“аблица є4
| є ¬арианта | ’имические реакции |
| 1. | 4NH3(г)+5ќ2(г)↔6Ќ2ќ(г)+4NO(г) |
| 2. | CO(г)+3H2(г) ↔CH4(г)+H2O(г) |
| 3. | 4HCl(г)+ќ2(г) ↔2H2O(г)+Cl2(г) |
| 4. | 2CO(г)+3H2(г) ↔CH4(г)+CO2(г) |
| 5. | 2CH4(г)+ќ2(г) ↔2—O(г)+4H2(г) |
| 6. | CH4(г)+2H2O(г) ↔CO2(г)+4H2(г) |
| 7. | CH4(г)+H2O(г) ↔CO(г)+3H2(г) |
| 8. | 4NH3(г)+3ќ2(г) ↔6Ќ2ќ(г)+2N2(г) |
| 9. | C2H4(г)+3ќ2(г) ↔2—O2(г)+2H2O(ж) |
| 10. | 4NH3(г)+5ќ2(г) ↔6Ќ2ќ(ж)+4NO(г) |
| 11. | CH4(г)+ќ2(г) ↔2—O(г)+2H2O(г) |
| 12. | CO(г)+H2O(ж) ↔CO(г)+H2(г) |
| 13. | CH4(г)+—ќ2(г) ↔2—O(г)+2H2(г) |
| 14. | CH4(г) ↔C2H2(г)+3H2(г) |
| 15. | 2CO(г)+3H2(г) ↔C2H2(г)+3H2O(ж) |
“аблица є5
| ¬ещество | —осто€ние | ∆Ќ0298, 
| ∆G0298, 
| S0298,  
|
| —2Ќ2 | г | 226,750 | 209,200 | 200,820 |
| NO | г | 90,370 | 86,690 | 210,200 |
| —2Ќ4 | г | 52,280 | 68,173 | 219,450 |
| NH3 | г | -45,190 | -16,647 | 192,500 |
| CH4 | г | -74,850 | -50,830 | 186,190 |
| —ќ | г | -110,520 | -137,270 | 197,910 |
| —ќ2 | г | -393,510 | -394,380 | 213,650 |
| H2O | г | -241,830 | -228,590 | 188,720 |
| H2O | ж | -285,950 | -237,190 | 69,940 |
| N2 | г | 0,000 | 0,000 | 200,000 |
| H2 | г | 0,000 | 0,000 | 130,590 |
| ќ2 | г | 0,000 | 0,000 | 205,030 |
| Cl2 | г | 0,000 | 0,000 | 222,950 |
7. “≈ћј " инетика химических реакций. ’имическое равновесие "
”чение о кинетике химических процессов €вл€етс€ одним из важных разделов химии, посв€щенных изучению скоростей химических реакций в зависимости от различных факторов (концентрации реагентов, температуры, давлени€, катализаторов и др.). ѕри рассмотрении обратимых химических реакций с одинаковыми скорост€ми пр€мого и обратного процессов (состо€ние химического равновеси€), большое значение имеет принцип смещени€ химического равновеси€ Ће Ўателье.
ѕри изучении данного раздела необходимо:
- выучить закон действующих масс, правило ¬ант-√оффа, принцип Ће Ўателье;
- уметь составл€ть выражени€ скоростей пр€мой и обратной реакции дл€ любого химического процесса;
- знать, как вли€ют различные факторы: температура, давление, концентраци€, наличие катализатора на скорость химических реакций и особенности действи€ катализаторов;
- уметь составл€ть выражение константы химического равновеси€, грамотно использовать принцип Ће Ўателье при определении смещени€ химического равновеси€.
ѕример: N2 + 3H2  2NH3 + Q
2NH3 + Q
—корость пр€мой реакции:  =
= 
 ;
;
—корость обратной реакции:  =
= 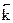
 ;
;
|
|
|
онстанта равновеси€: с = 
ѕри увеличении концентрации Ќ2 равновесие сместитс€ вправо, т.к. по правилу Ће Ўателье преимущество получит та реакци€, в ходе которой водород будет расходоватьс€, т.е. пр€ма€.
ѕри увеличении температуры равновесие сместитс€ влево т.к. по правилу Ће Ўателье преимущество получит та реакци€, в ходе которой температура системы понижаетс€, т.е. обратна€ реакции, идуща€ с поглощением теплоты.
ѕри увеличении давлени€ равновесие сместитс€ вправо т.к. по правилу Ће Ўателье преимущество получит та реакци€, в ходе которой давление в системе понижаетс€, т.е. пр€ма€ реакции, в ходе которой из 4 моль вещества образуетс€ 2 моль вещества.






