Fe(OH)2Cl + NaOH = Fe(OH)3 â + NaCl
(основна€ соль)(основание)
Fe(OH)2+ + OH = Fe(OH)3
¬заимодействие с кислотами:
Na2CO3 + 2Ќ—1 = 2NaCl + —O2 á + H2O
—O32- + 2Ќ+ = —O2 á + Ќ2O
—а—ќз + 2Ќ—1 = —а—12 + —O2 á + Ќ2O
—а—ќз + 2Ќ+ = —а2+ + —O2 + Ќ2O
NaHCO3 + HC1 → CO2 á + H2O +NaC1
Na2S + H2S = 2NaHS
(нормальна€ (кисла€ соль)
соль)
S2- + H2S = 2HS
MgOHCl +HC1 = MgCl2 + Ќ2O
(основна€ соль) (нормальна€ соль)
MgOH ++ Ќ+ = Mg2+ + Ќ2O
AgNO3 +HC1 = AgCl â +HNO3
Ag+ + Cl - = AgCl
¬а—12 + H2SO4 = BaSO4 â + 2Ќ—1
Ba2+ + SO42-= BaSO4
Pb(NO3)2 + H2S = PbS â + 2HNO3
Pb2+ + H2S = PbS + 2H+
¬заимодействие солей друг с другом (возможны дл€ растворимых солей):
AgNO3 + NaCl = AgCl â + NaNO3
Ag+ + CI- = AgCl
Ba(NO3)2 + K2S04 = BaSO4 â + 2KNO3
Ba2+ + SO42- = BaSO4
CaCl2 + Na2CO3 = CaCO3 â + 2NaCl
Ca2+ + CO32- = CaCO3
Zn(NO3)2 + K2S = ZnSâ + 2KNO3
Zn2+ + S2- = ZnS
NH4HCO3 + NaCl = NaHCO3 â + NH4Cl
HCO3 - + Na+ = NaHCO3
2K3[Fe(CN)6] + 3FeSO4 = Fe3[Fe(CN)6]2 â + 3K2SO4
2[Fe(CN)6]3- + 3Fe2+ = Fe3[Fe(CN)6]2
FeCl3 + 3NH4SCN à Fe(SCN)3 + 3NH4C1
ß
Fe3+ + 3SCN à Fe(SCN)3 (слабый электролит)
ß
ћногие обменные реакции €вл€ютс€ качественными реакци€ми на катионы и анионы: реакци€ (2) - на катионы NH4+; реакции (6, 7, 8) - на анион —O32-; реакции (11, 14) - на катион Ag+ и анион —l-; реакции (12,15) - на катион ¬а2+ и анион SO42-; реакци€ (13) - на анион S2-; реакци€ (19) - на катион Fe2+; реакци€ (20) - на катион Fe3.
б) ќкислительно-восстановительные реакции
–еакции, в которых катионы соли играет роль окислител€
а) взаимодействие растворов солей с металлами (более активные металлы восстанавливают менее активные металлы из растворов их солей):
+2 0
CuSO4 + Fe = —u + FeSO4
+2 0
Hg(NO3)2 + —u = Hg + Cu(NO3)2
б) взаимодействие растворов солей с другими восстановител€ми:
+1 0
8 AgNO3 + –Ќ3 + 4Ќ2O = 8Ag + Ќ3–O4 + 8HNO3
–еакции, в которых катион соли играет роль восстановител€
+2 +3
2FeCl2 + —12 = 2FeCl3
+2+3
4—г—12 + 4Ќ—1 +O2 = 4—г—13 + 2Ќ2O
+3 +6
2—г—13 + 3NaBiO3 + 7NaOH + H2O = 2Na2CrO4 + 3Bi (OH)3 + 6NaCl
–еакции, в которых анион соли играет роль окислител€
+6 +3
2—г2O7 + 3H2O2 + 4H2SO4 = Cr2(SO4)3 + 3O2 + K2SO4 + 7H2O
+7 +4
2 ћnO4 + 3—2Ќ4 + 4Ќ2O = 2ћnO2 â + 3—2Ќ4(ќЌ)2 + 2 ќЌ
–еакции, в которых анион соли играет роль восстановител€:
0
2 l - + C12 = l 2 + 2KC1
+3 +6
2 —rO2 + 3–bO2 + 2KOH = 2 2—rO4 + 3–bќ + Ќ2O
–еакции, в которых и катион, и анион соли играют роль восстановителей:
+2 -1 t +3 +4
4FeS2 + 11O2= 2Fe2O3 + 8SO2
+2 -2+3 0
10FeS + 36HNO3 = 10Fe(NO3)3 + 10S + 3N2 + 18H2O
(разб.)
–еакци€ внутримолекул€рного окислени€-восстановлени€:
+5 -2 t, кат -1 0
2 —lO3 = 2 —1 + 3O2á
+5 t + 4 0
2Cu(NO3)2 = 2CuO + 4NO2 á + O2á
+7 -2 t +6 +4 0
2KMnO4 = K2MnO4 + MnO2 + O2 á
в) –еакции разложени€ солей, происход€щие без переноса электронов.
|
|
|
≈сли анион соли не про€вл€ет окислительных свойств, то такие соли, как правило, разлагаютс€ без изменени€ степеней окислени€:
t
—а—O3 = —аќ + —O2 á
t
Mg(HCO3)2 = Mg(OH)2â+ 2—O2 á
t
(CuOH)2CO3 = 2CuO + —O2 á + H2O
(NH4)2SO4 = NH3 á+ NH4HSO4
5. —пособы получени€ солей.
Ќиже представлены способы получени€ солей на примере растворимой соли бескислородной кислоты (хлорид натри€) и нерастворимой соли кислородосодержащей кислоты (сульфат бари€).
| ѕолучение NaCl | |
| осн. + к-та | NaOH + Ќ—1 = NaCl + H2O |
| осн. оке. + к-та | Na2O + 2 Ќ—1 = 2NaCl + H2O |
| Me + к-та | 2Na + 2HCl = 2NaCl + H2 á |
| щелочь + соль | 2NaOH + CuCl2 = 2NaCl + Cu(OH)2 â |
| соль + кислота | Na2CO3 + 2Ќ—1 = 2NaCl + CO2 á+ H2O |
| соль + соль__________ | Na2SO4 + BaCl2 = 2NaCl + BaSO4_________ |
| Me + HeMe | 2Na + Cl2 = 2NaCl |
| гидрид +к-та | NaH + HCl = NaCl + H2á |
| ѕолучение BaSO4 | |
| осн. + к-та | Ba(OH)2 + H2SO4= BaSO4 â + 2H2O |
| осн. оке. + к-та | BaO + H2S04 = BaSO4 â + H2O |
| Me + к-та | Ba + H2SO4 = BaSO4 â + H2 á (разб.) |
| щелочь + соль | Ba(OH)2 + Na2SO4 = BaSO4 â + 2NaOH |
| соль + кислота | BaCl2 + H2SO4 = BaSO4 â + 2Ќ—1 |
| соль + соль__________ | Ba(NO3)2 + Na2SO4 = BaSO4 â + 2NaNO3 |
| осн. оке. + кисл. оке. | BaO + SO3 = BaSO4 |
| ўелочь + кисл. оке. | Ba(OH)2 + SO3 = BaSO4 â + H2O |
ѕервые 6 способов получени€ €вл€ютс€ идентичными дл€ обеих солей.
¬опросы дл€ самоподготовки:
— помощью уравнений химических реакций осуществите генетическую св€зь между классами неорганических веществ

|
Ћекци€ є8
“ема: јмфотерность. ’имические свойства амфотерные оксидов и гидроксидов. √енетическа€ св€зь между классами неорганических соединений.
ѕлан
1. јмфотерные оксиды.
2. јмфотерные гидроксиды; определение и важнейшие представители; физические свойства.
3. ’имические свойства:
а) основные свойства;
б) кислотные свойства;
в) термическое разложение.
4. —пособы получени€.
5. √енетическа€ св€зь между классами неорганических соединений.
1. јмфотерные оксиды.
јмфотерные оксиды, облада€ свойствами и основных и кислотных оксидов, взаимодействуют с сильными кислотами и щелочами:
ZnO (как основный оксид) + 2Ќ—1 (раствор) = ZnCl2 + H2O
ZnO (тв., как кислотный оксид) + 2NaOH (тв.) = Na2ZnO2 (цинкат натри€) + Ќ2ќ
или в растворе щелочи:
ZnO + 2NaOH + H2O = Na2[Zn(OH)4] (тетрагидроксоцинкат натри€)
ZnO + 2 ќЌ - + Ќ2O = [Zn(OH)4]2-
2. јмфотерные гидроксиды; определение и важнейшие представители;
физические свойства.
јмфотерные гидроксиды - слабые электролиты, которые способны диссоциировать как с образованием ионов Ќ+ (по типу кислоты), так и с образованием ионов ќЌ- (по типу основани€).
ƒиссоциаци€ по кислотному типу усиливаетс€ в щелочной среде, а по основному типу - в кислой среде:
ќЌ Ќ+


 Ќ+ + ћеќ- ћеќЌ ће+ + ќЌ Ц
Ќ+ + ћеќ- ћеќЌ ће+ + ќЌ Ц
¬ажнейшие представители:
| ‘орма основани€ | ‘орма кислоты |
| Zn(OH)2 гидроксид цинка | H2ZnO2 ÷инкова€ кислота |
| ј1(ќЌ)3 гидроксид алюмини€ | Ќ3ј1O3 - ортоалюминева€ к-та ЌјlO2 - метаалюминиева€ к-та |
| —r(ќЌ)3 гидроксид хрома (III) | Ќ3—rќз - ортохромиста€ к-та ЌCr02 - метахромиста€ к-та |
| ¬е(ќЌ)2 гидроксид берили€ | Ќ2¬еO2 бериллиева€ к-та |
| Sn(OH)2 гидроксид олова (II) | H2SnO2 олов€нниста€ к-та |
| Sn(OH)4 гидроксид олова (IV) | H4SnO4 - ортоолов€на€ к-та H2SnO3 - метаолов€нна€ к-та |
| –b(ќЌ)2 гидроксид свинца (II) | Ќ2–bO2 - свинцова€ к-та |
| –b(ќЌ)4 гидроксид свинца (IV) | Ќ4–bO4 - ортосвинцова€ к-та Ќ2–bќз - метасвинцова€ к-та |
‘изические свойства: все амфотерные гидроксиды представл€ют собой твердые вещества, плохо растворимые в воде.
|
|
|
3. ’имические свойства.
ѕро€вл€€ свойства очень слабых оснований и очень слабых кислот, амфотерные гидроксиды взаимодействуют с сильными кислотами и с сильными основани€ми (щелочами).
а) ќсновные свойства.
¬ результате реакций с кислотами в раствор переход€т катионы металлов, вход€щих в состав амфотерных гидроксидов; например:
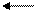
 Zn(OH)2 + 2Ќ—1 ZnCl2 + 2Ќ2O
Zn(OH)2 + 2Ќ—1 ZnCl2 + 2Ќ2O
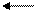
 Zn(OH)2 + 2H+ Zn2+ + 2H2O
Zn(OH)2 + 2H+ Zn2+ + 2H2O
 2Al(OH)3 + 3H2SO4 A12(SO4)3 + 6H2O
2Al(OH)3 + 3H2SO4 A12(SO4)3 + 6H2O

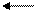 Al(OH)3 + 3H+ A13+ + 3H2O
Al(OH)3 + 3H+ A13+ + 3H2O
 б) кислотные свойства.
б) кислотные свойства.
Co щелочами амфотерные гидроксиды могут взаимодействовать при высокой температуре в расплавах; при этом образуютс€ соли, содержащие кислотные остатки метакислотных форм амфотерных гидроксидов, например:
t
ј1(ќЌ)3 + NaOH = NaAlO2 + 2Ќ2O
метаалюминат
натри€
t
2—r(ќЌ)3 + ¬а(ќЌ)2 = ¬а(—rO2)2 + 4Ќ2O
метахромит бари€
ѕри взаимодействии амфотерных гидроксидов с растворами щелочей образуетс€ гидроксокомплексы с координационным числом, как правило, 4 или 6; например:
Zn(OH)2 + 2NaOH = Na2[Zn(OH)4] - тетрагидроцинкат натри€
Zn(OH)2 + 2OЌ - = [Zn(OH)4]2-
ј1(ќЌ)3 + NaOH = Na[Al(OH)4] - тетрагидроксоалюминат натри€
ј1(ќЌ)3 + ќЌ - = [AL(OH)4] -
или:
ј1(ќЌ)3 + 3NaOH = Na3[Al(OH)6] - гексагидроксоалюминат натри€
ј1(ќЌ)3 + 3ќЌ - = [ј1(ќЌ)6]3 -
в) термическое разложение.
ак и нерастворимые основани€, амфотерные гидроксиды разлагаютс€ при нагревании (дегидратируютс€):
t
2ј1(ќЌ)3 = ј12O3 + 3Ќ2O
4. —пособы получени€.
а) ѕодробно нерастворимым основани€м, амфотерные гидроксиды можно
осаждать щелочами из растворов соответствующих солей, избега€ избытка
щелочи:
ј1—13 + 3NaOH = ј1(ќЌ)3 â + 3NaCl
ј13+ + 3ќЌ - = ј1(ќЌ)3
ZnSO4 + 2KOH = Zn(OH)2 â +K2SO4
Zn2+ + 2OH - = Zn(OH)2
б) јмфотерные гидроксиды выдел€ютс€ также в процессе разрушени€
гидроксокомплексов при действии кислот:
3[—r(ќЌ)6] + «Ќ—1 = —r(ќЌ)3 â + 3 —1 + 3Ќ2O
[Cr(OH)6]3 - + 3Ќ+ = —r(ќЌ)3 + 3Ќ2O
или при растворении в растворах кислот цинкатов, метаалюминатов и т.д.:
Na2ZnO2 + 2HC1 = Zn(OH)2 â + 2NaCl
ZnO22 - + 2Ќ+ = Zn(OH)2
NaAlO2 + HC1 + H2O = Al(OH)3 â + NaCl
AlO2 - + H+ + H2O = Al(OH)3
¬о всех этих реакци€х следует брать строго рассчитанное количество кислоты, поскольку в избытке кислоты образующиес€ амфотерные гидроксиды раствор€ютс€.
5. √енетическа€ св€зь между классами неорганических соединений.
ћногочисленные способы получени€ солей из веществ, принадлежащих к разным классам, свидетельствует о том, что между этими классами неорганических веществ существует тесна€ взаимосв€зь.
—в€зь между классами неорганических соединений, которые основаны на получении веществ одного класса из веществ другого класса, называетс€ генетической.
√енетическа€ св€зь между важнейшими классами неорганических веществ проиллюстрировано на следующей схеме:
ћеталл
|

 Ќеметалл Ќеметалл
|

 +O2
+O2 
 +O2
+O2
| ќсновный |     оксид оксид
|
| ислотный | оксид |

 +H2O +H2O
+H2O +H2O
ќснование

|

| ислота |
| соль |
—ход€щиес€ стрелки в этой схеме показывают взаимодействие тех веществ, от которых они направлены, с образованием солей.
1. Me + неће
2. ће + O2
3. ћеќ + Ќ2O
4. неће + ќ2
5. нећеќ + Ќ2O
6. ћеќ + нећеќ
7. ћеќЌ + нећеќ
8. Ќјu + ћеќ
9. ћеќЌ + Ќјu
10. ће + Ќјu
¬заимосв€зь классов неорганических соединений
| Ќеметалл | ислотный оксид | ислота | —оль | ¬ода | |
| ћеталл | —оль1 | —оль2 + водород | 1 —оль3 + металл | ќснование4 или основной оксид и водород | |
| ќсновный оксид | - | 1 —оль | —оль + вода | - | ќснование5 |
| ќснование | - | 1 —оль + вода6 | —оль + вода | —оль + основание7 | - |
| —оль | - | - | —оль + кислота8 | —оль + соль | ѕродукты |
| гидролиза | |||||
| ¬ода | - | ислота* | - | ѕродукты гидролиза | - |
|
|
|
1. ≈сли неметалл - кислород, то образуетс€ оксид.
2. ≈сли метал в р€ду напр€жений стоит до водорода и если кислота сильна€ (кроме азотной и концентрированной серной).
3. »сходный металл должен обладать большей восстановительной активностью, чем металл, вход€щий в состав соли.
4. ћеталлы, расположенные в р€ду напр€жений от лити€ до натри€ включительно, образуют основани€; а от магни€ до никел€ - оксиды. ќстальные c водой не реагирует.
5. »сходные оксиды должны быть оксидами металлов главных подгрупп I и II групп (кроме берилли€ и магни€).
6. ќснование должно быть растворимо.
7. »сходные вещества должны быть растворимы, а один из продуктов реакции - нерастворим.
8. »сходна€ кислота должна быть устойчивее, чем кислота, образовавша€с€ соль.
9. SiO2 с водой не реагирует.
¬опросы дл€ самоподготовки:
1. ќсуществить превращени€:
 ZnCl2 à Zn(OH)2 à ZnSO4 à Zn(NO3)2
ZnCl2 à Zn(OH)2 à ZnSO4 à Zn(NO3)2

 á
á
 Zn Zn(NO3)2
Zn Zn(NO3)2
Na2Znќ2
2. ѕредложите способ получени€ оксида меди (II) CuO из сульфата меди (»), воды и металлического натри€.
3. Ќапишите уравнени€ реакций, доказывающие амфотерность гидроксида меди (II).
4. ѕредложите способ получени€:
а) тетрагидроксоцинката натри€;
б) метаалюмината натри€.
Ћекци€ є 9.
“ема: омплексные соединени€.
ѕлан:
1. —троение комплексных соединений. “еори€ ¬ернера.
2. ѕрирода химической св€зи в комплексных ионах.
3. лассификаци€ и номенклатура комплексных соединений
4. ƒиссоциаци€ комплексных соединений
5. ѕрименение комплексных соединений в медицине и химическом анализе.
1. —троение комплексных соединений. “еори€ ¬ернера.
¬ результате взаимодействи€ некоторых простых бинарных соединений друг с другом образуютс€ новые, более сложные соединени€ - комплексные.
4KCN + Fe(CN)2 = K4[Fe(CN)6];
омплексные соли при диссоциации образуют сложные комплексные ионы, которые способны существовать в растворе:
K4[Fe(CN)6] → 4K+ +[Fe(CN)6]4-
¬ водном растворе K4[Fe(CN)6] имеютс€ катионы + и анионы [Fe(CN)6]4 -, но нельз€ обнаружить ионы Fe2+ и CN -. »з этого следует, что они соединились и образовали сложный ион [Fe(CN)6]4 -, который называетс€ комплексным. —остав комплексного соединени€ изображают химической формулой, в которой комплексный ион заключают в квадратные скобки. ќбразование двойных солей не ведет к существенным изменени€м свойств исходных соединений, из которых они образовались.
ѕерва€ теори€ строени€ комплексных соединений была разработана швейцарским химиком ј.¬ернером в 1893г. ќсобый тип валентных сил, на котором основано образование комплексных соединений, был назван ¬ернером координационной св€зью, а теори€ получила название координационной.
—огласно теории ¬ернера, центральное место в комплексном соединении занимает комплексообразователь, или центральный атом. омплексообразовател€ми чаще всего €вл€ютс€ катионы металлов. Ќаибольшую склонность к комплексообразованию про€вл€ют ионы d -элементов: Aq+,Cu2+, Co3+, Fe3+, Fe2+ и др. ¬округ центрального атома наход€тс€ (или координируютс€) противоположно зар€женные ионы или нейтральные молекулы, которые называютс€ лигандами или аддендами. омплексообразователь и лиганды образуют внутреннюю координационную сферу комплексного соединени€, котора€ заключаетс€ в квадратные скобки. „исло лигандов, которое св€зываетс€ с комплексообразователем, называетс€ координационным числом. „аще всего они принимают значени€ 4, 6 и завис€т от степени окислени€ центрального атома и природы лиганда.
|
|
|
“аблица 1 оординационные числа дл€ ионов-комплексообразова гелей
| оординационное число | »оны - комплексообразователи |
| Cu+, Aq+, Au+ Cu2+, Co2+, Au2+, Zn2+, Pb2+, Pt 2, Al3+ Fe2+, Fe3+, Co3+, Ni2+, Al3+, Pt4+, Hg4+, Cr3+ |
¬ качестве лигандов выступают нейтральные молекулы (Ќ2O, NH3CO), ионы (NO2-. —l -, ¬r -, ќЌ - и др.). аждый лиганд содержит определенное число атомов (донорных атомов), которые участвуют в непосредственном св€зывании с центральным атомом и образуют определенное число координационных св€зей (дентатность). ≈сли лиганд образует одну св€зь с центральным атомом, то это монодентатный лиганд. монодентатным лигандам относ€тс€ —l -, J -, Br -, CN -, ќЌ - и др. ¬ случае монодентатных лигандов координационное число равно числу лигандов.
Ѕидентатный лиганд содержит два атома, которые образуют св€зь с центральным атомом. Ќапример, этилендиамин (en) H2N - —Ќ2 - —Ќ2 - NH2 €вл€етс€ бидентатным лигандом, в молекуле которого каждый из двух атомов азота образует одну св€зь с центральным атомом. ¬ комплексном ионе [Cu(en)3]2+ координационное число меди равно 6, то есть в случае би - и полидентатных лигандов координационное число равно числу лигандов умноженному на дентатность полидентатным лигандам относ€тс€ аминокислоты, белки пептиды, порфирины, которые называютс€ еще биолигандами.
«ар€д комплексного иона равен алгебраической сумме зар€дов иона-комплексообразовател€ и лигандов:
 Aq+ + 2 CN - [Aq(CN)2] - ; (+1 - 2= -1)
Aq+ + 2 CN - [Aq(CN)2] - ; (+1 - 2= -1)
 Pt4+ + 6C1 [PtCL6]2 -. (+4- 6= -2)
Pt4+ + 6C1 [PtCL6]2 -. (+4- 6= -2)
¬ход€щие в состав комплексного иона электронейтральные молекулы не вли€ют на величину его зар€да. ≈сли лигандами €вл€ютс€ только электронейтральные молекулы, то зар€д комплексного иона равен зар€ду комплексообразовател€. Ќапример, в комплексных ионах [Aq(NH3)2]+ и [Cu(NH3)4]2+ зар€д ионов равен зар€ду комплексообразовател€.
»оны, не вошедшие во внутреннюю координационную сферу. ќбразуют внешнюю координационную сферу. »онами внешней сферы часто €вл€ютс€ ионы щелочных и щелочноземельных металлов, ионы аммони€ и анионы.
2. ѕрирода химической св€зи в комплексных ионах
ƒл€ объ€снени€ образовани€, строени€ и свойств комплексных соединений примен€ютс€
–азличные теории, но наиболее нагл€дным и доступным €вл€етс€ метод валентных св€зей (ћ¬—)
—огласно ћ¬—, в процессе образовани€ комплексного иона устанавливаетс€ прочна€ св€зь между комплекснообразователем и лигандами. ћеханизм образовани€ комплексного иона [Zn(NH3)4]2+.
јтом азота в молекуле аммиака имеет неподеленную пару электронов, а
ион Zn2+ имеет четыре свободные орбитали. ѕоэтому при образовании
комплексного иона [Zn(NH3)4]2+ роль донора играют четыре молекулы аммиака,
а роль акцептора - ион цинка Zn2+, который принимает четыре пары электронов
от четырех молекул аммиака. ¬ образовании св€зи участвуют одна 4s- орбиталь
и три 4р-орбитали иона цинка. омплексный ион [Zn(NH3)4]2+ имеет
геометрическую конфигурацию (тетраэдр), что означает осуществление sp -
гибридизации. ќбразование комплексного иона [Zn(NH3)4]2+ может быть
показано и следующим образом:







