ХОД УРОКА
I. Организационный этап
II. Проверка домашнего задания, актуализация опорных знаний
Задание.
Напишите электронные формулы атомов кислорода и серы. Расставьте степени окисления у соединений: H2S, SO2, SO3, H2SO4.
(O – 1s22s22p4, S – 1s22s22p63s23p43d0; H+12S-2, S+4O-22, S+6O-23, H+12S+6O-24)
Беседа.
1) Что такое аллотропия? Окислитель? Восстановитель? Окисление? Восстановление? (Аллотропия — существование одного и того же химического элемента в виде двух и более простых веществ, различных по строению и свойствам.
Окислитель – вещество, атомы которого отдают во время прохождения окислительно-восстановительной реакции электроны. Окисление – процесс отдачи атомом химического элемента электронов.
Восстановитель – вещество, атомы которого присоединяют во время прохождения окислительно-восстановительной реакции электроны. Восстановление – процесс принятия атомом химического элемента электронов.)
2) Какие аллотропные модификации кислорода вы знаете? (Кислород О2 и озон О3)
3) Каково местоположение элементов кислорода и серы в Периодической системе? (О – 6 группа, 2 период; S – 6 группа, 3 период)
4) Какой из элементов – кислород или сера – проявляет более неметаллические свойства? (Ввиду большей электроотрицательности, а значит, большей способности к присоединению электронов, кислород является бóльшим неметаллом, чем сера)
III. Изучение нового материала
1. Химические свойства кислорода
Рассказ учителя
Кислород, как мы только что повторили, является одним из самых активных неметаллов, потому может реагировать с многими металлами и неметаллами, за исключением фтора, хлора, инертных газов и благородных металлов. Но магний – один из самых активных металлов, потом с ним реакция окисления происходит очень быстро. Итак, первое, что следует записать про химические свойства (ХС) кислорода – взаимодействие с металлами.
2Mg0 + O20 = 2Mg+2O-2
Оксид магния
(магний – восстановитель, кислород – окислитель)
Демонстрация
Щипцами берется стружка металлического магния и вносится в пламя горелки.
Рассказ учителя
Следующий пункт ХС кислорода – взаимодействие с неметаллами.
4P0 + 5O20 = 2P2+5O5-2
Оксид фосфора(V)
(фосфор – восстановитель, кислород – окислитель)
При действии кислорода при высокой температуре некоторые вещества горят. Это вы можете наблюдать каждый день, когда зажигаете дома конфорку на плите. Знает ли кто-нибудь, какой газ у нас в газопроводах? Правильно, метан. Его формула – CH4. Итак, следующий пункт химических свойств кислорода – горение сложных веществ.
СН4 + 2О2 = СО2 + 2Н2О
Беседа
Итак, чем характеризуется горение? (выделением большого количества света и тепла).
Рассказ учителя
Горение – процесс быстрого окисления веществ, сопровождающийся выделением большого количества энергии в виде, преимущественно, света и тепла.
Как мы видим, во всех рассмотренных нами реакциях кислород выступает окислителем.
2. Применение кислорода.
Рассказ учителя
Теперь давайте рассмотрим применение кислорода. Первое, что можно назвать в данной подтеме – это получение высоких температур, которое мы уже рассмотрели. Следующий пункт, который необходимо записать – применение в медицине. В различных ситуациях, когда человеку не хватает кислорода для дыхания, ему дают дышать газовой смесью с повышенным содержанием кислорода.
3. Химические свойства серы.
Рассказ учителя
Сера, как вы помните, желтый порошок, а кислород – бесцветный газ. Несмотря на различные физические свойства, они во многом похожи по химическим, хотя сера менее активный неметалл и может быть как восстановителем, так и окислителем. Соединения, в которых сера проявляет степень окисления -2, называются сульфидами. Например:
C0 + 2S0 = C+4S2-2
Сульфид углерода
(углерод – восстановитель, сера – окислитель)
При реакции водорода с серой может образовываться сероводород:
Н2 + S = H2S
Раствор сероводорода в воде называется сульфидной кислотой.
Поскольку сера – неметалл, то она хорошо реагирует с различными металлами.
Zn0 + S0 = Zn+2S-2
Сульфид цинка
(цинк – восстановитель, сера – окислитель)
2Al0 + 3S0 = Al2+3S3-2
Сульфид алюминия
(алюминий – восстановитель, сера – окислитель)
Hg0 + S0 = Hg+2S-2
Сульфид ртути
(ртуть – восстановитель, сера – окислитель)
Реакция серы со ртутью происходит даже при комнатной температуре, чем пользуются, чтобы собрать разлившуюся ртуть. Как видим, при реакции этих веществ образуется сульфид ртути, который образует нерастворимую пленку на шариках жидкой ртути, препятствуя их растеканию.
Но сера может быть и восстановителем. Например, в реакции с кислородом:
S0 + O20 = S+4O2-2
Оксид серы (IV)
(сера – восстановитель, кислород– окислитель)
Демонстрация
(Под тягой!) В ложечку для сжигания помещается некоторое количество кристаллической серы. Ложечка вносится в пламя газовой горелки, сера поджигается наклоном ложечки и горит ярким голубоватым пламенем.
4. Оксид серы (IV)
Рассказ учителя
Как мы видим по уравнению реакции, в ходе горения серы образуется оксид серы (IV), или сернистый газ. Необходимо записать, что SO2 - бесцветный газ с резким запахом, ядовит, негорючий, растворяется в воде. Его запах мы слышим, когда зажигаем спичку, поскольку в ней содержится сера.
Оксид серы (IV) содержится в вулканических газах; поскольку он является кислотным оксидом, то с водой образует кислоту. Сернистый газ – отход предприятий, они выкидывают его в атмосферу, где он превращается в кислоту и выпадает с дождем на землю – наблюдаются так называемые кислотные дожди.
Получение оксида серы (IV):
Оксид серы может образовываться в ходе сжигания серы и некоторых ее соединений
S + O2 = SO2
2H2S + 3O2 = 2H2O + 2SO2,
при окислении серной кислотой веществ
Cu + 2H2SO4(к) = CuSO4 + SO2 + 2H2O,
при образовании и разложении кислоты H2SO3 (вспомните Н2СО3)
Na2SO3 + 2HCl = 2NaCl + H2O + SO2,
при обжиге пирита
4FeS2 + 11O2 = 2Fe2O3 + 8SO2.
Химические свойства сернистого газа можно разделить на три группы:
1) Реакции без смены степени окисления
Сернистый газ – кислотный оксид, реагирует с основными оксидами и основаниями:
CaO + SO2 = CaSO3
2KOH + SO2 = K2SO3
2) Реакции с повышением степени окисления
2S+4O2 + O2 = 2S+6O3 (реакция происходит при повышенных давлении и температуре с участием катализатора)
3) Реакции с понижением степени окисления
S+4O2 + 2H2S = 3S0 + 2H2O
Беседа
В каких из перечисленных выше реакциях химических свойств сернистый газ выступает восстановителем? (в реакции повышения степени окисления)
Рассказ учителя
Оксид серы (IV) применяется:
· Для получения серной кислоты
· Как отбеливатель бумаги, соломы, шерсти
· Как дезинфицирующее средство
IV. Закрепление и обобщение знаний
Задание
Цинк массой 13 г сплавили с избытком серы. Полученное твердое вещество обработали избытком серной кислоты. Определите объем выделившегося газа (н.у.).
Решение:
Zn + S = ZnS
ZnS + H2SO4 = ZnSO4 + H2S↑
По уравнениям реакций n(Zn) = n(ZnS) = n(H2S).
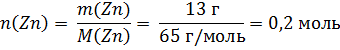
V(H2S) = Vm ∙ n(H2S) = Vm ∙ n(Zn) = 22,4 л/моль ∙ 0,2 моль = 4,48 л
V. Домашнее задание
1. Изучить пар. 3,4.
2. Выписать применение серы, разобрать оксид серы (VI) подобно оксиду серы (IV).
3. Подготовиться к химическому диктанту.
4. Выполнить задания 20-23 (с. 24), на более высокую оценку задания 24-28.






