| Класс веществ
| Реактив
| Признак реакции
| Объяснение
| Уравнение реакции
|
| Алканы
| Реакция горения
| Бесцветное (голубое) пламя
| Алканы химически неактивны.
Обычно определяют путем исключения.
| CH4 + 2O2 → 2H2O + CO2 + Q
|
| Алкены
| Бромная вода Вr2
| Обесцвечивание.
| Происходит присоединение брома по двойной связи.
| СН2=СН2 + Вr2 → CH2Br–CH2Br (1,2- дибромэтан)
|
| Раствор перманганата
калия KMnO4 (реакция Вагнера)
| Обесцвечивание раствора, выпадение бурого осадка МnО2
| Происходит разрыв π- связи и образуется диол.
| СН2=СН2 + [О] + Н2O ––KMnO4® HO - CH2 - CH2 - OH
этилен этиленгликоль
3СН2=СН2 + 2KMnO4 + 4Н2O ® 3HO - CH2 - CH2 – OH + 2 KOH + MnO2↓
|
| Реакция горения
| Коптящее (светящееся) пламя желтого цвета
| Возрастает доля углерода
| С2Н4 + ЗО2 → 2СO2 + 2Н2О + Q
|
| Алкины
| Бромная вода Вr2
| Обесцвечивание.
| Происходит присоединение брома по месту разрыва тройной связи.
| HCєCH + Br2® CHBr = CHBr (1,2- дибромэтен) ––+ Br2® CHBr2–CHBr2(1,1,2,2- тетрабромэтан)
|
| Раствор перманганата
калия KMnO4 (реакция Вагнера)
| Обесцвечивание раствора
|
|
|
| Реакция горения
| Сильно коптящее пламя
| Высока доля углерода
| 2С2Н2 + 5O2 → 4СO2 + 2Н2O + Q
|
| Ацетилен (алкины: тройная связь на конце цепи)
| Аммиачный раствор оксида серебра Ag2O
| Выпадение белого осадка
| Образуется ацетиленид серебра
|
СН≡СН + Ag2O ––амм.р-р t® AgCєCAg↓ + H2O
ацетиленид серебра
|
| Спирты первичные и вторичные
| Реакция горения
| Бесцветное (голубое) пламя.
|
| 2C2H5OH + 2O2 → 4CO2 + 6H2O + Q
|
| Оксид меди (II) CuO при нагревании
| Окраска меняется с черной (CuO) на красную (Cu), Запах альдегида
| Восстановление красной окраски у прокаленной горячей медной проволоки, окисление спиртов до альдегидов.
| C2H5OH + CuO ––t°® CН3СОН+ Cu + H2O
этанол уксусный альдегид
|
| K2Cr2O7, H2SO4
| Оранжевая окраска меняется на зеленую
| Окисление спиртов до альдегидов
| 3С2Н5ОН + K2Cr2O7 + 4Н2SO4 → 3CH3COH + K2SO4 + Сr2(SO4)3 + 7H2O
этанол дихромат калия этаналь сульфат хрома (III)
оранж. зеленый
|
| Многоатомные спирты (глицерин, этиленгликоль).
| Свежеприготовленный гидроксид меди (II) Cu(OH)2 в щелочной среде.
| Осадок Cu(OH)2 растворяется, раствор приобретает ярко синюю окраску (васильковую).
| Образуется комплексное соединение глицерата меди (II).
|   CH2 – OH CH2 – O HO – CH2 CH2 – OH CH2 – O HO – CH2
  | | Cu |
2 CH– OH + Cu(OH)2 →CH - OH O – CH + 2H2О
| | |
CH2 – OH CH2 – OH HO – CH2
глицерин глицерат меди (II) | | Cu |
2 CH– OH + Cu(OH)2 →CH - OH O – CH + 2H2О
| | |
CH2 – OH CH2 – OH HO – CH2
глицерин глицерат меди (II)
|
| Арены (бензол)
| Реакция горения
| Сильно коптящее пламя
| Высока доля углерода
| 2С6H6 + 15O2 → 12CO2 + 6H2O
|
| Арены (гомологи бензола: толуол)
| Раствор перманганата
калия KMnO4
| Обесцвечивание раствора
| Толуол окисляется в бензойную кислоту
| 5 С6Н5СН3 + 6 KMnO4 + 9 H2SO4 →
→ 5 С6Н5СООН + 2 K2SO4 + 6 MnSO4 + 14 H2O
бензойная кислота
|
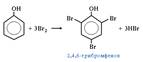
| Фенол
| Бромная вода Вr2
| Выпадение белого осадка
| Образуется нерастворимый 2, 4, 6 – трибромфенол
| 
|
| Раствор хлорида
Железа (III) FeCl3
| Жидкость окрашивается в фиолетовый цвет.
| Образуется фенолят железа (III).
| С6Н5ОН + FeCl3  фиолетовая (аметистовая) окраска фиолетовая (аметистовая) окраска
|
| Альдегиды
| Свежеприготовленный гидроксид меди (II) Cu(OH)2 в щелочной среде и нагревание
| Осадок изменяет цвет на жёлтый, затем на кирпично-красный.
| Гидроксид меди (II) восстанавливается до гидроксида меди (I) (жёлтый). Затем гидроксид меди (I) распадается на воду и оксид меди (I).
Альдегид окисляется до карбоновой кислоты.
| СН3-COН + 2Сu(OH)2 ––t® СН3COOH + 2CuOH↓ + H2O
этаналь уксусная кислота гидроксид меди (I)
желтый
2CuOH –– t® Cu2O↓ + H2O
оксид меди (I) кирпично-красный
суммарное уравнение:
СН3-COН + 2Сu(OH)2 ––t® СН3COOH + Cu2O↓ + 2H2O
|
|
Реактив Толленса - гидроксид
диаминсеребра [Ag(NH3)2] OH (аммиачный раствор оксида серебра Ag2O) и
нагревание.
|
Образование слоя серебра на стенках пробирки «серебряное зеркало» или мелкодисперсного серебра (если пробирка грязная или если жидкость закипела).
|
Восстановление серебра из оксида, окисление карбонильной группы до карбоксильной.
| СН3-COН + 2[Ag(NH3)2]OH → СН3COONH4 + 2Ag↓ +3NH3 + H2O
этаналь аммонийная соль уксусной кислоты
СН3-COН + Ag2O ––амм.р-р t® СН3COOH + 2Ag↓
этаналь уксусная кислота
НCOН + Ag2O ––амм.р-р t® НCOOH + 2Ag↓
метаналь муравьиная кислота
НCOОН + Ag2O ––амм.р-р t® CO2 + Н2O + 2Ag↓
|
| Предельные одноосновные карбоновые кислоты
| Лакмус.
| Лакмус – красный.
| В растворе кислоты есть ионы Н+.
|
|
| Муравьиная кислота
Аммиачный раствор оксида серебра Ag2O и нагревание.
| Реакция «серебряного зеркала».
| Восстановление серебра из оксида, окисление карбонильной группы до карбоксильной.
| НCOОН + Ag2O ––амм.р-р t® CO2 + Н2O + 2Ag↓
|
| Высшие карбоновые кислоты
| Отношение к индикаторам
| Не изменяют окраску.
|
|
|
| Реакция нейтрализации (щелочи)
| Появление хлопьев мыла.
| Образование мыла.
| С17Н35СООН + NaOH → С17Н35СООNa + H2O
|
| Олеиновая кислота
Бромная вода Вr2
| Обесцвечивание.
| Происходит присоединение брома по двойной связи.
| С17Н33СООН + Br2 → С17Н35СООН
олеиновая кислота стеариновая кислота
|
| Амины
| Лакмус, фенолфталеин.
| Лакмус – синий.
| Щелочная среда.
|
|
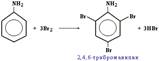
| Анилин
| Бромная вода Вr2
| Выпадение белого осадка
| Образуется нерастворимый 2, 4, 6 – триброманилин.
| 
|
| Хлорная известь СaClOCl
| Фиолетовое окрашивание.
| Происходит окисление анилина.
|
|
| Глюкоза
| Свежеприготовленный гидроксид меди (II) Cu(OH)2 - реакция на годроксильные
группы (как многоатомный спирт).
| Осадок Cu(OH)2 растворяется, раствор приобретает ярко синюю окраску (васильковую).
| Образуется комплексное соединение глюконата меди (II).
|
|
| Свежеприготовленный гидроксид меди (II) Cu(OH)2 и нагревание – реакция на альдегидную группу.
| Осадок изменяет цвет на жёлтый, затем на кирпично-красный.
| Гидроксид меди (II) восстанавливается до гидроксида меди (I) (жёлтый), который распадается на воду и оксид меди (I). Альдегид окисляется до карбоновой кислоты.
| HOСН2-(СНOH)4-СОН + 2Сu(OH)2 ––t® HOСН2-(СНOH)4-СOOH + Cu2O↓ + 2H2O
глюкоза глюконовая кислота
|
| Аммиачный раствор оксида серебра Ag2O и нагревание.
| Реакция «серебряного зеркала»
| Восстановление серебра из оксида, окисление карбонильной группы до карбоксильной.
| HOСН2-(СНOH)4-СОН+Ag2O ––амм.р-р t® HOСН2-(СНOH)4-СOOH + 2Ag↓
глюкоза глюконовая кислота
|
| Крахмал
| I2 раствор
| При обычных условиях появляется синее окрашивание, которое исчезает при нагревании. При охлаждении окраска появляется вновь.
| Образуется комплексное соединение иода с амилозой - линейной формой молекулы крахмала.
|
|
| Белки
| Ксантопротеиновая реакция(наличие бензольных колец)
Конц. HNO3, нагревание.
| Желтое окрашивание при нагревании оранжевое. Белок сворачивается.
| Разрушаются дисульфидные мостики и происходит нитрование бензольных колец в молекуле белка.
|
|
| Биуретовая реакция
Cu(OH)2 в щелочной среде
| Фиолетовое окрашивание
| Образуется комплексное соединение.
|
|
| Жиры
| Реакция горения
| Сильно коптящее пламя
| Высока доля углерода
|
|
| Реакция омыления.
| Появление хлопьев мыла.
| Образование мыла.
|
|
| Мыла (стеарат натрия)
| Отношение к индикаторам (гидролиз)
| Фенолфталеин - малиновый, лакмус - синий
| Щелочная среда.
|
|

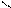 CH2 – OH CH2 – O HO – CH2
CH2 – OH CH2 – O HO – CH2

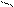 | | Cu |
2 CH– OH + Cu(OH)2 →CH - OH O – CH + 2H2О
| | |
CH2 – OH CH2 – OH HO – CH2
глицерин глицерат меди (II)
| | Cu |
2 CH– OH + Cu(OH)2 →CH - OH O – CH + 2H2О
| | |
CH2 – OH CH2 – OH HO – CH2
глицерин глицерат меди (II)

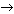 фиолетовая (аметистовая) окраска
фиолетовая (аметистовая) окраска







