–ј—Ќќя–— »… »Ќ—“»“”“ ∆≈Ћ≈«Ќќƒќ–ќ∆Ќќ√ќ
“–јЌ—ѕќ–“ј -
филиал государственного образовательного учреждени€ высшего профессионального образовани€
Ђ»ркутский государственный университет путей сообщени€ї
в г. расно€рске
’»ћ»„≈— »≈ —¬ќ…—“¬ј ћ≈“јЋЋќ¬
ќ––ќ«»я ћ≈“јЋЋќ¬ » «јў»“ј ќ“ ќ––ќ«»»
Ё ¬»¬јЋ≈Ќ“ » ћќЋя–Ќјя ћј——ј Ё ¬»¬јЋ≈Ќ“ј
ћетодические указани€ к лабораторным работам
дл€ студентов первого курса всех специальностей
расно€рск 2007
”ƒ 54
ЅЅ 24.1
’имические свойства металлов. оррози€ металлов и защита от коррозии. Ёквивалент и мол€рна€ масса эквивалентов: ћетодические указани€ к лабораторным работам дл€ студентов первого курса всех специальностей Ц расно€рск: р»∆“ »р√”ѕ—, 2007, 41с.
—оставитель: Ћюдмила јлександровна –убчевска€
–ецензенты: √рачева ≈.¬.., к.т.н., доцент кафедры
общематематических и естественнонаучных
дисциплин р»∆“ »р√”ѕ—.
“юменева √.“., к.т.н., доцент кафедры химии
»нститута архитектуры и строительства
Ђ—ибирского федерального университетаї
ѕечатаетс€ по решению методического совета расно€рского института железнодорожного транспорта
© расно€рский институт железнодорожного транспорта - филиал »ркутского государственного университета путей сообщени€ в г. расно€рске, 2007
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј 1
’»ћ»„≈— »≈ —¬ќ…—“¬ј ћ≈“јЋЋќ¬
÷ель работы: ознакомитьс€ с наиболее характерными дл€ всех металлов химическими свойствами.
“≈ќ–≈“»„≈— ќ≈ ќЅќ—Ќќ¬јЌ»≈
»з 116 известных к насто€щему времени элементов к металлам относ€тс€ более 85. ћеталлы отличаютс€ от неметаллов физическими и химическими свойствами. ƒл€ всех металлов (кроме ртути) характерно твердое агрегатное состо€ние, металлический блеск Ц результат отражени€ световых лучей, электро- и теплопроводность, пластичность. ѕеречисленные физические свойства металлов обусловлены их внутренним строением Ц металлическим типом св€зи в кристаллической решетке. Ќа различии других физических свойств металлов основана их классификаци€ в технике. “ак, по величине плотности металлы дел€тс€ на легкие, плотность которых меньше 5 г/см3, и т€желые Ц ≠≠ с плотностью более 5 г/см3. ¬ зависимости от температуры плавлени€ металлы дел€тс€ на тугоплавкие Ц с температурой плавлени€ выше 15000— и легкоплавкие Ц с температурой плавлени€ ниже 10000—. ¬ технике прин€то деление металлов на черные и цветные. ÷ветные металлы также дел€тс€ на несколько групп. “ака€ классификаци€ (табл. 1) не €вл€етс€ строгой и совершенной, но она находит широкое использование на практике.
“аблица 1
“ехническа€ классификаци€ металлов
| „ерные | ÷ветные | ||||||||
| легкие | драгоценные | т€желые | –едкие | ||||||
| рассе€нные | тугоплавкие | легкие | редкоземельные | радиоактивные | жидкие | ||||
| Fe, Mn, Cr и их сплавы | Li, Na, K, Be, Mg, Ca, Ba, Al | Pt, Ir, Os, Pd, Ru, Rh, Au, Ag | Cu, Zn, Sn, Pb, Bi | Ga, In, Tl, Re | Ti, Zr, V, Nb, Ta, W, Mo, Co, Ni | Rb, Cs, Sr | Sc, Y, La и лантаноиды | Po, Ra, U, Ac и актиноиды | Hg |
¬ химических реакци€х металлы выступают как восстановители
|
|
|
ће Ц ne- = Mеn+ процесс окислени€,
восстановитель
где ће Ц атом металла, e- Ц электрон, Mеn+ Ц положительно зар€женный ион металла с зар€дом n+.
Ёто объ€сн€етс€ тем, что атомы металлов имеют небольшое число электронов на внешнем энергетическом уровне (главным образом 1, 2 и 3). »сключение составл€ют германий (Ge), олово (Sn), свинец (Pb), на внешнем слое которых 4 электрона. ¬ св€зи с этим у металлов низка€ величина потенциала ионизации. ѕотенциал ионизации это количество энергии, необходимое дл€ отрыва электрона от атома. „ем он меньше, тем легче металлы отдают электроны и тем большей восстановительной активностью обладают. Ќаименьшие значени€ ионизационного потенциала Ц у щелочных металлов, которые и €вл€ютс€ самыми энергичными восстановител€ми.
¬заимодействие металлов с кислородом. ислородом окисл€ютс€ почти все металлы, особенно при нагревании. ѕри этом образуютс€ оксиды
2Mg + O2 = 2MgO
ўелочные металлы при горении на воздухе образуют пероксиды и надпероксиды: (Na2O2, KO2, RbO2). ќднако многие металлы (Cr, Al, Be, Mg, Ti и др.) в компактном состо€нии при обычных температурах устойчивы к действию кислорода, так как покрываютс€ тончайшей оксидной пленкой, котора€ предохран€ет их от дальнейшего окислени€. «олото (Au), платина (Pt) и отчасти серебро (Ag) не реагируют с кислородом.
¬ водных растворах химическую активность металла определ€ют на основании р€да напр€жений или р€да стандартных электродных потенциалов металлов (табл.2).
¬ р€ду напр€жений металлы расположены в пор€дке увеличени€ алгебраической величины их стандартных потенциалов, следовательно, убывани€ восстановительной активности атомов металлов и возрастани€ окислительной активности ионов металлов.
ѕоэтому каждый металл восстанавливает все металлы, сто€щие за ним, из растворов их солей.
“аблица 2
—тандартные потенциалы металлических электродов (“ = 298 0 )
| Ёлектрод | Ёлектродна€ реакци€ | ≈0, ¬ |
| Li+/Li | Li + + е - = Li | - 3,045 |
| Rb+/Rb | Rb+ + e - = Rb | - 2,295 |
| K+/K | K+ + e - = K | - 2,924 |
| —s+/—s | Cs+ + e - = Cs | -2,923 |
| ¬а2+/¬а | ¬а2+ +2e - = Ba | -2,906 |
| —а2+/Cа | —а2+ +2e - = Ca | -2,866 |
| Na+/Na | Na+ + e - = Na | -2,714 |
| ћg2+/Mg | Mg2+ + 2e - = Mg | -2,363 |
| Be2+/Be | Be2+ + 2e - = Be | -1,847 |
| Al3+/Al | Al3+ + 3e - = Al | -1,662 |
| Ti2+/Ti | Ti2+ + 2e - = Ti | -1,628 |
| Mn2+/Mn | Mn2+ + 2e - = Mn | -1,180 |
| Zn2+/Zn | Zn2+ + 2e - = Zn | -0,763 |
| Cr3+/Cr | Cr3+ + 3e - = Cr | -0,744 |
| Fe2+/Fe | Fe2+ + 2e - = Fe | -0,440 |
| —d2+/Cd | Cd2+ + 2e - = Cd | -0,403 |
| —о2+/Co | Co2+ + 2e - = Co | -0,277 |
| Ni2+/Ni | Ni2+ + 2e - = Ni | -0,250 |
| Sn2+/Sn | Sn2+ + 2e - = Sn | -0,136 |
| Pb2+/Pb | Pb2+ + 2e - = Pb | -0,126 |
| 2Ќ+/H2 | 2H+ + 2e - = H2 | |
| Cu2+/Cu | Cu2+ + 2e - = Cu | +0,337 |
| Ag+/Ag | Ag+ + e - = Ag | +0,799 |
| Hg2+/Hg | Hg2+ + 2e - = Hg | +0,854 |
| –d2+/Pd | Pd2+ + 2e - = Pd | +0,987 |
| Pt2+/Pt | Pt2+ + 2e - = Pt | +1,190 |
| Au3+/Au | Au3+ + 3e - = Au | +1,498 |
Ќапример, цинк будет восстанавливать из растворов солей только катионы металлов, сто€щих в р€ду напр€жений после него:
|
|
|
Zn + NiSO4 = Ni + ZnSO4
Zn Ц 2e- → Zn2+ окисление
Ni2+ + 2e- → Ni восстановление,
а реакци€ Zn + MgSO4 → невозможна.
¬ р€ду стандартных электродных потенциалов металлов условно можно выделить:
- активные металлы (Li, K, Rb, Cs, Ba, Ca, Na, Mg, Be, Al);
- металлы средней активности (Ti, Mn, Zn, Fe, Cd, Co, Ni, Sn, Pb);
- малоактивные металлы (Cu, Ag, Hg, Pd, Pt, Au), сто€щие после водорода.
¬заимодействие металлов с водой. ќкислителем в воде формально €вл€етс€ ион водорода. “ак как концентраци€ ионов H+ в воде равна 10-7 моль/л, то вычисл€€ по формуле Ќернста потенциал водородного электрода получаем:
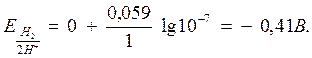
ѕоэтому водород из воды могут вытесн€ть металлы, имеющие потенциал меньший, чем Ц 0,41 ¬, т.е. сто€щие впереди кадми€
(см. табл. 2). ѕрактически же взаимодействуют с водой лишь щелочные и щелочноземельные металлы, причем магний при нагревании:
Ca + H2O = Ca(OH)2 + H2.
ќбъ€сн€етс€ это тем, что необходимым условием реакции между металлом и водой €вл€етс€ удаление с его поверхности оксидной пленки, например, по реакции
CaO + H2O = Ca(OH)2.
ѕри этом образуетс€ растворимый в воде гидроксид. ≈сли поверхностна€ пленка металла не может быть удалена водой вследствие нерастворимости соответствующего гидроксида (Zn(OH)2, Fe(OH)2 и др.), то реакци€ не пойдет. “акие металлы (Zn, Fe) могут реагировать с парами воды при нагревании, так как при высокой температуре поверхностна€ пленка разрушаетс€:
Fe + H2O (пар) ←→ H2 + FeO.
¬заимодействие металлов с хлороводородной (сол€ной) кислотой. ќкислителем в сол€ной кислоте, так же как и в воде, €вл€етс€ ион водорода. —тандартный электродный потенциал водородного электрода приравнен к нулю. ѕоэтому все активные металлы и металлы средней активности реагируют с кислотой с выделением водорода
Fe + 2HCl = FeCl2 + H2
| HCl (окислитель Ќ+) | ||
| јктивные Li Ц Al | —редней активности Ti Ц H2 | ћалоактивные H2 Ц Au |
| реагируют | реагируют, | не реагируют |
| реагирует и пассивируетс€ –b |
Ќо при взаимодействии свинца с сол€ной кислотой образующийс€ в начале реакции хлорид свинца (ѕ) трудно растворим в воде и, остава€сь на поверхности металла, защищает его от доступа новых порций кислоты:
–b + 2HCl = PbCl2↓ + H2≠.
¬ результате реакци€ останавливаетс€.
¬заимодействие с разбавленной серной кислотой.
–азбавленна€ серна€ кислота, так же как и сол€на€, окисл€ет, своим ионом водорода. ќна взаимодействует только с теми металлами, электродные потенциалы которых ниже, чем у водорода, например,
Fe + H2S04 (разб.) = FeS04 + Ќ2,
а реакци€ свинца с разбавленной серной кислотой быстро прекращаетс€, так как образующа€с€ соль PbS04 нерастворима и создает на поверхности металла защитную пленку.
¬заимодействие с концентрированной серной кислотой.
¬ концентрированной серной кислоте в роли окислител€ выступает сера в степени окислени€ +6, вход€ща€ в состав сульфат-иона SO2-4. ¬ св€зи с этим концентрированной кислотой окисл€ютс€ все металлы, стандартный электродный потенциал которых меньше, чем электродный потенциал окислител€. ћаксимальное значение электродного потенциала в электродных процессах с участием сульфат-иона в качестве окислител€ равно 0,36 ¬. ѕоэтому с концентрированной серной кислотой реагируют и некоторые малоактивные металлы:
| Ќ2Sќ4 (конц.) (окислитель S+6) | ||||
| јктивные Li Ц Al | —редней активности Ti Ц H2 | ћалоактивные H2 Ц Au | ||
| реагируют, | реагирует: Cu реагируют при нагревании Hg, Ag | не реагируют: | ||
| реагируют, пассивируетс€ | реагируют и пассивируютс€: | јu, Ru, ќs, | ||
| Al | Fe, —r | Rh, Ir, Pt | ||
| ислота восстанавливаетс€, в основном, до следующих продуктов: | ||||
| H2S (S-2) | S (S0) или Sќ2 (S+4) | Sќ2 (S+4) | 1, | Ч |
¬ зависимости от активности металла, температуры, концентрации кислоты продуктами ее восстановлени€ могут быть H2S, S и Sќ2. —праведливо правило, что чем активней металл, тем глубже степень восстановлени€ кислоты.
|
|
|
¬заимодейству€ с активными металлами (Li Ц Mg в р€ду напр€жений), кислота восстанавливаетс€ до сероводорода (H2S)
4 Mg + 5H2S04 (конц.) = 4MgS04 + H2S + 4Ќ2ќ.
¬ реакци€х с малоактивными металлами (Cu,Ag) образуетс€ всегда газ Sќ2, причем серебро раствор€етс€ в кип€щей кислоте
Cu + 2Ќ2Sќ4 (конц.) = CuS04 + SO2 + 2H2O;
t0 C
2Ag + 2Ќ2Sќ4 (конц.) = Ag2S04↓+ SO2 + 2H2O.
ћеталлы средней активности (Zn, Sn, Pb и др.) реагируют с концентрированной серной кислотой с выделением S или Sќ2
3Zn + 4H2S04 (конц.) = 3ZnS04 + S + 4Ќ2ќ;
Sn + 4Ќ2Sќ4 (конц.) = Sn(S04)2 + 2SO2 + H2O;


Pb + 3Ќ2Sќ4 (конц.) = Pb (ЌS04)2 + SO2 + 2H2O.
ѕриведенные реакции показывают, что олово окисл€етс€ до четырехвалентного состо€ни€ с образованием сульфата олова (IV), а свинец окисл€етс€ до двухвалентного состо€ни€ с образованием растворимого гидросульфата свинца.
“акие металлы как Al, Fe, —r при обычных услови€х серной кислотой, концентраци€ которой близка к 100%, пассивируютс€. ѕассивацией называетс€ €вление образовани€ защитной пленки на поверхности металла при действии сильных окислителей. ¬ большинстве случа€х пассиваци€ играет положительную роль, защища€ металл от разрушени€. «а счет пассивации серную кислоту перевоз€т в стальных цистернах.
¬заимодействие с разбавленной азотной кислотой.
¬ азотной кислоте в качестве окислител€ выступает азот в степени окислени€ +5. «начение электродного потенциала дл€ нитрат-иона Nќ-3 разбавленной кислоты как окислител€ равно 0,96 ¬, т.е. оно больше по сравнению с сульфат-ионом SO2-4. Ёто указывает на то, что азотна€ кислота Ц более сильный окислитель, чем серна€. ƒействительно, она окисл€ет даже серебро. ¬осстанавливаетс€ азотна€ кислота тем глубже, чем активнее металл и чем меньше ее концентраци€:
| ЌNќз (разб.) (окислитель N+5) | |||
| јктивные Li - Al | —редней активности Ti - H2 | ћалоактивные H2 - Au | |
| реагируют | реагируют, | реагируют: Cu, Hg, Ag | не реагируют: јu,Ru, Os, Rh, Ir, Pt |
| пассивируютс€ Ti, Cr | |||
| ислота восстанавливаетс€, в основном, до следующих продуктов: | |||
| NЌ4Nќз (N-3) | N2 (N20), N2O (N+1), NO (N+2) | NO (N+2) | Ч |
»ллюстрацией к таблице могут быть следующие примеры:
8HNO3 (разб.) + 3Cu = 2NO + 3Cu(NO3)2 + 4H2O;
4HNO3 (разб.) + Fe = NO + Fe(NO3)3 + 2H2O;
8HNO3 (разб.) + 3Sn = 2NO + 3Sn(NO3)2 + 4H2O;
10HNO3 (разб.) + 4Zn = N2O + 4Zn(NO3)2 + 5H2O;
10HNO3 (разб.) + 4Ca = NЌ4Nќз + 4Ca(NO3)2 + 3H2O.
»з приведенных примеров видно, что железо окисл€етс€ в разбавленной азотной кислоте до нитрата железа (III), а олово до нитрата олова (II).
“итан и хром пассивируютс€ в разбавленной азотной кислоте.
¬заимодействие с концентрированной азотной кислотой. онцентрированна€ азотна€ кислота, в отличие от разбавленной, обычно восстанавливаетс€ до диоксида азота:
| ЌNќз (конц.) (окислитель N+5) | |||
| јктивные Li - Mg | —редней активности Al, Ti - H2 | ћалоактивные H2 - Au | |
| реагируют | реагируют, реагируют | реагируют: Cu, Hg, Ag | не реагируют: јu, Ru, Os, Rh, Ir, Pt |
| и пассивируютс€ | |||
| Al, Ti, Cr, Fe | |||
| ислота восстанавливаетс€, в основном, до следующих продуктов: | |||
| N2O (N+1) | NO2 (N+4) | NO2 (N+4) | Ч |
ѕримеры:
10HNO3 (конц.) + 4Cа = N2O + 4Cа(NO3)2 + 5H2O;
4HNO3 (конц.) + Zn = 2NO2 + Zn (NO3)2 + 2H2O;
|
|
|
4HNO3 (конц.) + —u = 2NO2 + —u(NO3)2 + 2H2O.
≈сли металл имеет устойчивые высокие степени окислени€, то в результате реакции получаютс€ не нитраты, а оксиды или даже кислоты, например,
Sn + 4ЌNќз (конц.) = Ќ2Snќз + 4Nќ2 + Ќ2ќ.
онцентрированна€ азотна€ кислота (концентраци€ больше 70%) еще в большей степени, по сравнению с серной, обладает пассивирующим действием и не реагирует при обычных услови€х с Al, “i, Fe, —г. ѕоэтому концентрированную азотную кислоту также перевоз€т в стальных или алюминиевых цистернах.
¬заимодействие с Ђцарской водкойї. ќкислительные свойства азотной кислоты усиливаютс€ при добавлении к ней концентрированной сол€ной кислоты. Ќаибольший эффект наблюдаетс€ при соотношении ЌNќз:HCl = 1:3. “ака€ смесь называетс€ Ђцарской водкойї. ѕри действии Ђцарской водкиї раствор€етс€ золото и платина
јu + ЌNќз + 4HCl = H[AuC14] + NO + 2Ќ2ќ.
¬заимодействие с растворами щелочей. ўелочами металлы окисл€тьс€ не могут, так как щелочные металлы €вл€ютс€ одними из наиболее сильных восстановителей, а их ионы Ц одни из наиболее слабых окислителей. ќднако в присутствии щелочей окисл€ющее действие воды может про€витьс€ в большей мере, чем в их отсутствие. ѕри окислении металлов водой образуютс€ гидроксиды и водород. ≈сли оксид и гидроксид относ€тс€ к амфотерным соединени€м, то они будут раствор€тьс€ в щелочном растворе. ¬ результате пассивные в чистой воде металлы могут энергично взаимодействовать с растворами щелочей:
| Ќ2ќ (окислитель H+) + щелочь (например, NaOH) | ||
| јктивные | —редней активности | ћалоактивные |
| реагируют: ¬е, Al | реагируют: Zn, Sn, Pb | не реагируют: |
ѕроцесс растворени€ может быть представлен в виде двух стадий: окислени€ металла водой и растворени€ гидроксида:
Zn + 2ЌќЌ = Zn(OH)2 + Ќ2;
Zn(ќЌ)2 + 2NaOH = Na2[Zn(OH)4].
ќбщее уравнение реакции записываетс€ так:
Zn + 2ЌќЌ + 2NaOH = Nа2[Zn (ќЌ)4] + H2.
ќборудование и реактивы: штатив с пробирками, наждачна€ бумага, спиртовка, вод€на€ бан€, индикатор Ц фенолфталеин, дистиллированна€ вода, растворы HCl, H2SO4, HNO3, NaOH, KI, K3[Fe(CN)6], Fe(SCN)3; чистые металлы Ц медь, магний, цинк, алюминий, олово, свинец, железо; растворы различных солей.
Ё —ѕ≈–»ћ≈Ќ“јЋ№Ќјя „ј—“№






