РЕФЕРАТ
по дисциплине: Неорганическая химия
на тему: Железо, общая характеристика и его свойства
Проверила: Томашева Е.В.
Подготовил: ФЗР, 1 Курс, 1 "а" группа
Кузьмин В.Е.
Гродно 2016
ПЛАН
1. Введение.
2. Нахождение в природе.
3. Получение железа.
4. Физические свойства железа.
5. Химические свойства железа.
6. Применение и биологическая роль железа и его соединений.
Введение
В периодической системе железо находится в четвертом периоде, в побочной подгруппе VIII группы
.
Химический знак – Fe (феррум). Порядковый номер – 26, электронная формула 1s2 2s2 2p6 3d6 4s2.
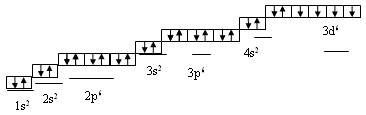
Валентные электроны у атома железа находятся на последнем электронном слое (4s2) и предпоследнем (3d6). В химических реакциях железо может отдавать эти электроны и проявлять степени окисления +2, +3 и, иногда, +6.
Нахождение в природе.
Железо является вторым по распространенности металлом в природе (после алюминия). В свободном состоянии железо встречается только в метеоритах, падающих на землю. Наиболее важные природные соединения:
Fe2O3 *3H2O – бурый железняк;
Fe2O3 – красный железняк;
Fe3O4(FeO *Fe2O3) – магнитный железняк;
FeS2 - железный колчедан (пирит).
Соединения железа входят в состав живых организмов.
Получение железа.
В промышленности железо получают восстановлением его из железных руд углеродом (коксом) и оксидом углерода (II) в доменных печах. Химизм доменного процесса следующий:
C + O2 = CO2,
CO2 + C = 2CO.
3Fe2O3 + CO = 2Fe3O4 + CO2,
Fe3O4 + CO = 3FeO + CO2,
FeO + CO = Fe + CO2.
Физические свойства.
Железо – серебристо серый металл, обладает большой ковкостью, пластичностью и сильными магнитными свойствами. Плотность железа – 7,87 г/см3, температура плавления 1539 oC.
Химические свойства.
В реакциях железо является восстановителем. Однако при обычной температуре оно не взаимодействует даже с самыми активными окислителями (галогенами, кислородом, серой), но при нагревании становится активным и реагирует с ними:
2Fe + 3Cl2 = 2FeCl3 Хлорид железа (III)
3Fe + 2O2 = Fe3O4(FeO *Fe2O3) Оксид железа (II,III)
Fe + S = FeS Сульфид железа (II)
При очень высокой температуре железо реагирует с углеродом, кремнием и фосфором:
3Fe + C = Fe3C Карбид железа (цементит)
3Fe + Si = Fe3Si Силицид железа
3Fe + 2P = Fe3P2 Фосфид железа (II)
Железо реагирует со сложными веществами.
Во влажном воздухе железо быстро окисляется (корродирует):
4Fe + 3O2 + 6H2O = 4Fe(OH)3 (ржавчина)
Железо находится в середине электрохимического ряда напряжений металлов, поэтому является металлом средней активности. Восстановительная способность у железа меньше, чем у щелочных, щелочноземельных металлов и у алюминия. Только при высокой температуре раскаленное железо реагирует с водой:
3Fe + 4H2O = Fe3O4 + 4H2
Железо реагирует с разбавленными серной и соляной кислотами, вытесняя из кислот водород:
Fe + 2HCl = FeCl2 + H2
Fe + H2SO4 = FeSO4 + H2
При обычной температуре железо не взаимодействует с концентрированной серной кислотой, так как пассивируется ею. При нагревании концентрированная H2SO4 окисляет железо до сульфита железа (III):
2Fe + 6H2SO4 = Fe2(SO4)3 + 3SO2 + 6H2O.
Разбавленная азотная кислота окисляет железо до нитрата железа (III):
Fe + 4HNO3 = Fe(NO3)3 + NO + 2H2O.
Концентрированная азотная кислота пассивирует железо.
Из растворов солей железо вытесняет металлы, которые расположены правее его в электрохимическом ряду напряжений:
Fe + CuSO4 = FeSO4 + C
Fe0 + Cu2+ = Fe2+ + Cu0.
Соединения железа (II)
Оксид железа (II) FeO – черное кристаллическое вещество, нерастворимое в воде. Оксид железа (II) получают восстановлением оксида железа(II,III) оксидом углерода (II):
Fe3O4 + CO = 3FeO + CO2.
Оксид железа (II) – основной оксид, легко реагирует с кислотами, при этом образуются соли железа(II):
FeO + 2HCl = FeCl2 + H2O,
FeO + 2H+ = Fe2+ + H2O.
Гидроксид железа (II) Fe(OH)2 – порошок белого цвета, не растворяется в воде. Получают его из солей железа (II) при взаимодействии их со щелочами:
FeSO4 + 2NaOH = Fe(OH)2 + Na2SO4,
Fe2+ + 2OH- = Fe(OH)2.
Гидроксид железа () Fe(OH)2 проявляет свойства основания, легко реагирует с кислотами:
Fe(OH)2 + 2HCl = FeCl2 + 2H2O,
Fe(OH)2 + 2H+ = Fe2+ + 2H2O.
При нагревании гидроксид железа (II) разлагается:
Fe(OH)2 = FeO + H2O.
Соединения со степенью окисления железа +2 проявляют восстановительные свойства, так как Fe2+ легко окисляются до Fe+3:
Fe+2 – 1e = Fe+3
Так, свежеполученный зеленоватый осадок Fe(OH)2 на воздухе очень быстро изменяет окраску – буреет. Изменение окраски объясняется окислением Fe(OH)2 в Fe(OH)3 кислородом воздуха:
4Fe+2(OH)2 + O2 + 2H2O = 4Fe+3(OH)3.
Восстановительные свойства проявляют и соли двухвалентного железа, особенно при действии окислителей в кислотной среде. Например, сульфат железа (II) восстанавливает перманганат калия в сернокислотной среде до сульфата марганца (II):
10Fe+2SO4 + 2KMn+7O4 + 8H2SO4 = 5Fe+32(SO4)3 + 2Mn+2SO4 + K2SO4 + 8H2O.
Качественная реакция на катион железа (II).
Реактивом для определения катиона железа Fe2+ является гексацаноферрат калия (III) (красная кровяная соль) K3[Fe(CN)6]:
3FeSO4 + 2K3[Fe(CN)6] = Fe3[Fe(CN)6]2 + 3K2SO4.
При взаимодействии ионов [Fe(CN)6]3- с катионами железа Fe2+ образуется темно-синий осадок – турнбулева синь:
3Fe2+ +2[Fe(CN)6]3- = Fe3[Fe(CN)6]2
Соединения железа (III)
Оксид железа (III) Fe2O3 – порошок бурого цвета, не растворяется в воде. Оксид железа (III) получают:
А) разложением гидроксида железа (III):
2Fe(OH)3 = Fe2O3 + 3H2O
Б) окислением пирита (FeS2):
4Fe+2S2-1 + 11O20 = 2Fe2+3O3 + 8S+4O2-2.
4e
11e
Fe+2 – 1e Fe+3 2S-1 – 10e 2S+4
4e
O20 + 4e 2O-2
Оксид железа (III) проявляет амфотерные свойства:
А) взаимодействует с твердыми щелочами NaOH и KOH и с карбонатами натрия и калия при высокой температуре:
Fe2O3 + 2NaOH = 2NaFeO2 + H2O,
Fe2O3 + 2OH- = 2FeO2- + H2O,
Fe2O3 + Na2CO3 = 2NaFeO2 + CO2(Феррит натрия)
Гидроксид железа (III) получают из солей железа (III) при взаимодействии их со щелочами:
FeCl3 + 3NaOH = Fe(OH)3 + 3NaCl,
Fe3+ + 3OH- = Fe(OH)3.
Гидроксид железа (III) является более слабым основанием, чем Fe(OH)2, и проявляет амфотерные свойства (с преобладанием основных). При взаимодействии с разбавленными кислотами Fe(OH)3 легко образует соответствующие соли:
Fe(OH)3 + 3HCl = FeCl3 + H2O
2Fe(OH)3 + 3H2SO4 = Fe2(SO4)3 + 6H2O
Fe(OH)3 + 3H+ = Fe3+ + 3H2O
Реакции с концентрированными растворами щелочей протекают лишь при длительном нагревании. При этом получаются устойчивые гидрокомплексы с координационным числом 4 или 6:
Fe(OH)3 + NaOH = Na[Fe(OH)4],
Fe(OH)3 + OH- = [Fe(OH)4]-,
Fe(OH)3 + 3NaOH = Na3[Fe(OH)6],
Fe(OH)3 + 3OH- = [Fe(OH)6]3-.
Соединения со степенью окисления железа +3 проявляют окислительные свойства, так как под действием восстановителей Fe+3 превращается в Fe+2:
Fe+3 + 1e = Fe+2.
Так, например, хлорид железа (III) окисляет йодид калия до свободного йода:
2Fe+3Cl3 + 2KI = 2Fe+2Cl2 + 2KCl + I20
Качественные реакции на катион железа (III)
А) Реактивом для обнаружения катиона Fe3+ является гексацианоферрат калия (II) (желтая кровяная соль) K2[Fe(CN)6].
При взаимодействии ионов [Fe(CN)6]4- с ионами Fe3+ образуется темно-синий осадок – берлинская лазурь:
 4FeCl3 + 3K4[Fe(CN)6] Fe4[Fe(CN)6]3 +12KCl,
4FeCl3 + 3K4[Fe(CN)6] Fe4[Fe(CN)6]3 +12KCl,
4Fe3+ + 3[Fe(CN)6]4- = Fe4[Fe(CN)6]3.
Б) Катионы Fe3+ легко обнаруживаются с помощью роданида аммония (NH4CNS). В результате взаимодействия ионов CNS-1 с катионами железа (III) Fe3+ образуется малодиссоциирующий роданид железа (III) кроваво-красного цвета:
 FeCl3 + 3NH4CNS Fe(CNS)3 + 3NH4Cl,
FeCl3 + 3NH4CNS Fe(CNS)3 + 3NH4Cl,
 Fe3+ + 3CNS1- Fe(CNS)3.
Fe3+ + 3CNS1- Fe(CNS)3.






