ќбратным процессу диссоциации комплекса €вл€етс€ процесс его образовани€.
Co2+ + Cl- ↔ [CoCl]+
[CoCl]+ + Cl- ↔ [CoCl2]0
[CoCl2]0 + Cl- ↔ [CoCl3]-
[CoCl3]-+ Cl- ↔ [CoCl4]2-
–авновесие при комплексообразовании характеризуетс€ с помощью полных (или суммарных) констант образовани€ данного комплекса bi(обр):
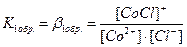



ѕолна€ константа образовани€ комплекса b n (обр) характеризует устойчивость комплексного соединени€ и равна произведению ступенчатых констант:

—тупенчатое комплексообразование можно проиллюстрировать примером взаимодействи€ иона Hg2+ (как комплексообразовател€) с ионами Cl- (как лигандом):
Hg2+ + Cl- ↔ [HgCl]+; K 1(обр) = 1,85 . 105
[HgCl]+ + Cl- ↔ [HgCl2]; K 2(обр) = 3,22 . 107
[HgCl2] + Cl- ↔ [HgCl3]-; K 3(обр) = 14,28
[HgCl3]- + Cl- ↔ [HgCl4]2- ; K 4(обр) = 10,00
Hg2+ + 4Cl- - ↔ [HgCl4]2; K обр. = K 1 обр. ∙ K 2обр. ∙ K 3обр. ∙ K 4обр. = 0,85 ∙ 1015
—тупенчатые и общие константы образовани€ и нестойкости комплексов соотнос€тс€ друг с другом как обратные величины:
β n (обр) = 1 / β n (нест); βn (обр) = 1 / βn (нест),
поэтому дл€ сравнени€ прочности комплексов могут использоватьс€ справочные данные как по значени€м констант образовани€, так и констант нестойкости.
“ак, константа образовани€ (устойчивости) коплексного иона [HgCl4]2- равна частному делени€ единицы на константу нестойкости этого же иона:
K обр. [HgCl4]2- = 1/ K нест. [HgCl4]2- = 1/(1,17 ∙ 10-15) = 0,85 ∙ 1015
„ем больше значение β n (обр), тем более устойчив комплекс данного состава. Ќапример, сравнива€ константы образовани€ катиона диамминсеребра(I) и дицианоаргентат(I)-иона:
(1) Ag+ + 2 NH3 ↔ [Ag(NH3)2]+; βо(обр) = 2,5 Ј 106
(2) Ag+ + 2 CN- ↔ [Ag(CN)2]-; βо(обр) = 1,2 Ј 1021
сделаем очевидный вывод о большей прочности второго комплекса Ц аниона [Ag(CN)2]-, поскольку, суд€ по большему значению константы образовани€, равновесие в реакции (2) сильнее сдвинуто вправо, чем в (1).
»ндекс n у константы образовани€ комплекса равен координационному числу центрального атома в том комплексном соединении, которое оцениваетс€.
“аким образом, величина b n (обр) св€зана со ступенчатыми константами образовани€ Ki (обр) соотношением:
b n (обр) = K 1(обр) ∙ K 2(обр) ∙ K 3(обр) ∙ Е Kn (обр)
¬ растворах — имеет место первична€ и вторична€ диссоциаци€ (в расплавах — происходит их термическа€ диссоциаци€). ѕервична€ диссоциаци€ протекает по типу сильных электролитов - практически необратимо:
K4[Fe(CN)6] → 4K+ + [Fe(CN)6]4-
[Co(NH3)6]Cl3 → [Co(NH3)6]3+ + 3Cl-
ѕервичной диссоциации не подвергаютс€ комплексы без внешней сферы: [Pt(NH3)2Cl2], [Co(NH3)3(NO3)3]. ¬торична€ диссоциаци€ характеризует диссоциацию самого комплекса. ќна протекает в незначительной степени, подчин€етс€ закону действи€ масс. Ётот процесс характеризуют константой диссоциации. “ак как величина этой константы фактически определ€ет прочность комплекса, то ее обычно называют константой нестойкости (K). ¬торична€ диссоциаци€ протекает по типу слабого электролита - обратимо и ступенчато:
|
|
|
[Ag(NH3)2]+ → [Ag(NH3)]+ + NH3
[Ag(NH3)]+ → Ag+ + NH3
¬ цел€х упрощени€ формы записи обычно записывают суммарное уравнение вторичной диссоциации:
[Ag(NH3)2] → Ag+ + 2NH3
¬ыражение константы нестойкости имеет вид:

ќбща€ константа нестойкости комплекса равна произведению констант диссоциации по всем ступен€м:
нест. = 1нест.Ј 2нест.
¬еличину, обратную константе нестойкости, называют константой устойчивости или константой образовани€ комплекса.
«начени€ констант нестойкости, приводимые в справочниках, используют дл€ характеристики устойчивости комплексов, нахождени€ концентраций частиц (комплекса, комплексообразовател€, лигандов) в растворе —, дл€ определени€ направленности химических реакций с участием комплексных соединений и др. Ќапример, равновесие в реакции:
[Ag(NH3)2]Cl + 2KCN = K[Ag(CN)2] + 2KCl + 2NH3
будет смещено вправо, так как Kнест.([Ag(NH3)2]+) = 9,3Ј10-8, а Kнест.([Ag(CN)2]-) = 8,0Ј10-22, т.е. второй комплексный ион значительно прочнее первого:







