»ќЌЌќ≈ ѕ–ќ»«¬≈ƒ≈Ќ»≈ ¬ќƒџ
ќдним из наиболее часто используемых растворителей €вл€етс€ вода, поэтому при различных расчетах используетс€ ионное произведение воды.
¬ода €вл€етс€ очень слабым электролитом, но тем не менее ее молекулы также диссоциируют на ионы. –ассмотрим обратимую реакцию (реакци€ автопротолиза воды):
ЌќЌ ЂЌ+ + ќЌ- или ЌќЌ + ЌќЌ = Ќ3ќ+ + ќЌ-
Ќ3ќ+ - ион гидроксони€.
 или
или  ; д = 1,86∙10-16.
; д = 1,86∙10-16.
Ќайдем ионное произведение воды.
[Ќ+][ќЌ-]= KW = Kд[ЌќЌ]; [HOH] = 1000/18 =55,5 моль/л.
[Ќ+][ќЌ-]= 1,86∙10-16 ∙55,5=10-14.
[Ќ+][ќЌ-]= 10-14
lg[H+] + lg[ќЌ-] = -14
-lg[H+] - lg[ќЌ-] = 14
ќтрицательный логарифм некоторой величины ћ в химии прин€то обозначать рћ, где ћ Ц концентраци€ тех или иных ионов без указани€ их зар€да, нека€ константа и т. д.
рЌ + рќЌ = 14.
¬ чистой воде, не содержащей растворенных веществ, [H+] = [ќЌ-].
[Ќ+] = [ќЌ-] = 10-7; [H+]2 = 10-14; [H+] = 10-7 и рЌ = 7.
рЌ Ц называют Ђводородным показателемї. «на€ рЌ раствора, легко найти его рќЌ и наоборот.
“огда. —ледовательно в водных растворах дл€ нейтральной среды рЌ = 7. “огда шкала рЌ в водных средах имеет вид
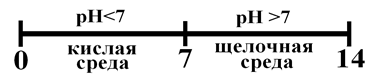
–ис.. Ўкала рЌ в водных растворах.
Ёто не значит, что рЌ кислых растворов не может быть меньше нул€, а щелочных Ц больше 14. ¬озможно и то, и другое. ¬ сильнокислых средах рЌ < 0, в щелочных Ц рЌ > 14.
≈сли растворитель Ц этиловый спирт то
2 —2Ќ5ќЌ Ђ—2Ќ5ќЌ2+ + —2Ќ5ќ-
этаксоний-ион этаксил-ион
д =  .
.
»онное произведение сольвата [—2Ќ5ќЌ2+][—2Ќ5ќ-] = 10-19.
[—2Ќ5ќЌ2+] = [—2Ќ5ќ-] = 
0______ _____9,5____ ______19.
≈сли в качестве растворител€ используетс€ аммиак, то
2 NH3 Ђ .
.
KS = 10-33
0______ _____16,5____ ______33.
10. Ѕуферные растворы. Ѕуферна€ емкость. «адачи по теме.
Ѕ”‘≈–Ќџ≈ –ј—“¬ќ–џ
¬еличина рЌ, характеризующа€ кислотность или щелочность среды, имеет большое значение в аналитической химии. рЌ вли€ет на концентрацию веществ в растворе, на скорость химических реакций, т.е. на точность анализа. ¬ отдельных случа€х введение кислоты или щелочи резко измен€ет значение рЌ, что негативно сказываетс€ на ходе анализа. ѕри введении этих растворов необходимо использовать буферные смеси, которые поддерживают значение рЌ в определенных пределах при введении кислоты или щелочи в раствор.
¬ качестве буферных растворов, примен€емых в анализе, используют::
- смесь уксусной кислоты и ее соли - ацетата натри€ (—Ќ3—ќќЌ и —Ќ3—ќќNa). Ёто ацетатна€ буферна€ смесь;
- смесь водного раствора аммиака и хлорида аммони€ NH4OH и NH4Cl - аммонийна€ буферна€ смесь;
- смесь дигидрофосфата натри€ NaH2PO4 и гидрофосфата натри€ Na2HPO4 - фосфатна€ буферна€ смесь.
—пособность буферных систем поддерживать посто€нство рЌ при добавлении к ним небольших количеств сильной кислоты или сильного основани€ основана на том, что одна составна€ часть буферной системы может взаимодействовать с ионами Ќ+ кислоты, а друга€ Ц с ионами ќЌЦ прибавл€емого основани€. ¬следствие этого буферна€ система может св€зывать как катионы водорода, так и гидроксид-анионы приливаемых кислот и оснований и до определенного предела сохран€ть посто€нство величины рЌ.
|
|
|
Ќапример, если к ацетатной буферной системе приливать сильную кислоту (Ќ—1, H2SO4, HNO3), то пойдет реакци€ между ацетат ионом и ионами водорода кислоты, что можно выразить уравнением:
CH3COONa + HC1 Þ—Ќ3—ќќЌ + NaCl
—Ќ3—ќќЦ + Ќ+ Þ—Ќ3—ќќЌ
—ильна€ кислота (Ќ—1 или H2SO4) замещаетс€ слабой уксусной кислотой, посылающей в раствор ничтожно малое количество ионов водорода.
ѕри добавлении же к ацетатной буферной системе сильной щелочи (NaOH, ќЌ) пойдет реакци€ нейтрализации уксусной кислоты сильным основанием: >
—Ќ3—ќќЌ + NaOH Þ CH3COONa + Ќ2ќ
Ќ+ + ќЌЦ Þ Ќ2ќ
¬ том и другом случае в растворе существенно не измен€ютс€ концентрации ни Ќ+, ни ќЌЦ ионов.
¬ аммиачной буферной системе протекают следующие реакции.
ѕри добавлении сильной кислоты идет нейтрализаци€ раствора аммиака кислотой:
NH4OH + HNO3 Þ NH4NO3 + H2O
ќЌЦ + Ќ+ Þ Ќ2ќ
ѕри добавлении сильного основани€ (NaOH, KOH) идет реакци€ обмена между солью и сильным основанием с образованием слабого основани€ NH4OH:
NH4—1 + NaOH Þ NH4OH + NaC1
NH4+ + ќЌЦ Þ NH4OH
—пособность буферных систем сохран€ть посто€нство рЌ определ€етс€ его буферной емкостью. ќна определ€етс€ числом моль-эквивалентов сильной кислоты или сильного основани€, которое необходимо добавить к 1 л буферного раствора, чтобы изменить его рЌ на единицу.
Ѕуферна€ емкость тем больше, чем выше концентраци€ компонентов буферной смеси. ѕо мере добавлени€ к буферному раствору кислоты или щелочи устойчивость раствора к изменению рЌ постепенно уменьшаетс€ вследствие уменьшени€ концентрации одного из компонентов. „тобы действие буферной системы было достаточно эффективным, т. е. чтобы буферна€ емкость раствора измен€лась не слишком сильно, концентраци€ одного компонента не должна превышать концентрацию другого компонента больше чем в 10 раз.
Ѕуферные системы широко и разносторонне используютс€ в аналитической химии. —остав буферных систем довольно разнообразен, поэтому имеетс€ их широкий выбор.ƒл€ создани€ и поддержани€ величины рЌ к исследуемому раствору добавл€ют ту или иную буферную смесь. “ак, аммиачна€ буферна€ система используетс€ дл€ осаждени€ гидроксида алюмини€, который при применении чистого раствора аммиака частично раствор€етс€ в его избытке.
јцетатный буферный раствор используетс€ при осаждении ионов бари€ дихроматом кали€ в присутствии ионов стронци€ и кальци€.
ћногие аналитические реакции с органическими реактивами, характеризующиес€ образованием окрашенных соединений, провод€тс€ при строго определенной величине рЌ. Ќапример, при обнаружении катиона Ni2+ диметилглиоксимом (реактив „угаева) используетс€ ацетатна€ буферна€ система с рЌ= 5.






