ƒл€ выражени€ кислотности или щелочности раствора используют величину водородного показател€ (рЌ), равную отрицательному дес€тичному логарифму концентрации ионов водорода [H+]:
рЌ = Цlg[H+], где [H+] = —ћ(Ќ+) моль/л.
ѕо характеру среды растворы можно подразделить на: кислые (растворы, в которых [H+] >10-7 моль/л), щелочные (растворы, в которых [H+] < 10-7 моль/л)и нейтральные (растворы, в которых [H+] = 10-7 моль/л).
“аблица 16
¬еличина рЌ раствора, завис€ща€ от характера среды раствора
| ’арактер среды (раствора) | ¬еличина рЌ | –авновесна€ концентраци€, [Ќ+], моль/л |
| кисла€ | < 7 | > 10-7 |
| нейтральна€ | ~ 7 | ~ 10-7 |
| щелочна€ | > 7 | < 10-7 |
–еакцию среды возможно определить при помощи специальных реактивов, или индикаторов, окраска которых мен€етс€ в зависимости от концентрации ионов водорода (таблица 17).
ƒл€ воды и разбавленных водных растворов при “=const:
[Ќ+]∙[ќЌ-] = const = Kв, где Kв Ц ионное произведение воды.
“аблица 17
¬ажнейшие индикаторы
| Ќазвание индикатора | ÷вет индикатора в различных средах | ||
| в кислой | в нейтральной | в щелочной | |
| ћетиловый оранжевый | красный (рЌ < 3,1) | оранжевый (3,1 < рЌ < 4,4) | желтый (рЌ > 4,4) |
| ћетиловый красный | красный (рЌ < 4,2) | оранжевый (4,2 < рЌ < 6,3) | желтый (рЌ > 6,3) |
| ‘енолфталеин | бесцветный (рЌ < 8,0) | бледно-малиновый (8,0 < рЌ < 9,8) | малиновый (рЌ > 9,8) |
| Ћакмус | красный (рЌ < 5,0) | оранжевый (5,0 < рЌ < 8,0) | желтый (рЌ > 8,0) |
ѕри “ = 298 (25∞—) дл€ чистой воды [Ќ+] = [ќЌ-]=10-7 моль/л, поэтому:
KW = Kв = 10-7Ј10-7 = 10-14.
—умма водородного и гидроксильного показателей в водных растворах при 25∞— равна 14. —ледует учитывать, что каковой бы ни была величина реакции раствора (рЌ), произведение концентраций [Ќ+]Ј[ќЌ-] остаЄтс€ неизменным. ¬еличина ионного произведени€ воды может изменитьс€ только при изменении температуры, но не при увеличении [Ќ+] или [ќЌ-].
√идролиз
–еакци€ взаимодействи€ между растворителем и растворЄнным веществом носит название сольволиза, а дл€ водных растворов Ц гидролиза.
¬ общем случае гидролиз Ц разложение веществ водой. ¬ода Ц одно из активных веществ, взаимодействущих с различными классами соединений: сол€ми, белками, жирами и т.д. ѕри растворении большинства солей в воде происходит разложение их с нарушением равновеси€ диссоциации воды за счЄт св€зывани€ ионов Ќ+ или ќЌЦ воды, т.е. вода способна разлагать растворЄнные вещества, действу€ на них как кислота или основание:
PCl3 + 3H2O = H3PO3 + 3HCl,
Al2S3 + 6H2O ↔ 2Al(OH)3 + 3H2S.
ѕри этом измен€етс€ кислотность растворов по сравнению с кислотностью растворител€. —оль гидролизуетс€.
√идролиз соли Ц обратимое взаимодействие ионов соли с ионами воды, привод€щее к изменению равновеси€ между ионами водорода и гидроксида в растворе. Ќапример, следующие реакции €вл€ютс€ реакци€ми гидролиза солей:
|
|
|
KCN+H2O ↔ HCN+KOH
AlCl3+H2O ↔ Al(OH)Cl2+HCl
√идролиз соли происходит лишь в случае образовани€ малодиссоциируемых соединений (в приведЄнных реакци€х HCN, Al(OH)Cl2).
√идролизу подвергаютс€ соли, образованные:
1) анионом слабой кислоты и катионом сильного основани€;
2) анионом сильной кислоты и катионом слабого основани€;
3) анионом слабой кислоты и катионом слабого основани€.
—оли же, образованные анионом сильной кислоты и катионом сильного основани€, не гидролизуютс€, а процесс нейтрализации в этом случае сводитс€ к следующему:
Ќ++ќЌ- ↔ Ќ2ќ
—ледует учитывать, что обратна€ реакци€ диссоциации воды на ионы протекает в крайне малой степени.
—лабыми кислотами €вл€ютс€: HNO2, H2SO3, H2O2, CH3COOH, H2SiO3, HF, H2CO3, HCN, H2S, H3PO4. —ильными кислотами €вл€ютс€: HNO3, H2SO4, HCl, HBr, HJ, ЌClO4, ЌMnO4. Cильными основани€ми €вл€ютс€ все щелочи (кроме NH4OH), например ќЌ, NaOH, Ba(OH)2, Ca(OH)2.
–ассмотрим подробнее основные три случа€ гидролиза солей:
1. √идролиз соли, образованной анионом слабой кислоты и катионом сильного основани€:
Ќапример
H2CO3 + 2 NaOH = Na2—O3 + 2H2O
слаба€ сильное
кислота основание
H2S + 2 KOH = K2S + 2H2O
слаба€ сильное
кислота основание
¬ водных растворах таких солей с H2O будут взаимодействовать анионы слабой кислоты, которые образуютс€ при диссоциации соли:
Na2—O3 Ѓ 2 Na + + CO3 2−
Ёти анионы будут присоедин€ть к себе ионы Ќ+, отщепившиес€ от молекул H2O, в результате этого образуетс€ слабый электролит H—O3− Ц гидрокарбонат-анион, а в растворе станут накапливатьс€ ионы ќЌ−, которые будут сообщать раствору такой соли щелочную реакцию.
HOH + CO32-  H—O3 − + OH Ц
H—O3 − + OH Ц
¬ результате этого рЌ растворов солей, образованных слабой кислотой и сильным основанием, будет > 7. ј в самом растворе станут присутствовать кисла€ соль и щелочь.
¬ молекул€рном виде уравнение гидролиза записываетс€ следующим образом:
Na2—O3 + ЌќЌ  NaH—O3 + NaOH
NaH—O3 + NaOH
¬заимодействие ионов соли с H2O €вл€етс€ обратимой реакцией и с течением времени в растворе устанавливаетс€ равновесие и при этом гидролизу подвергаетс€ только мала€ часть анионов соли. „ем слабее кислота, образующа€ соль, тем сильнее протекает гидролиз.
≈сли соль образована слабой многоосновной кислотой, то гидролиз может идти в несколько стадий или ступеней. »х число равно величине зар€да анионов слабой кислоты. Ќапример:

|
 HS- + OH-
HS- + OH-
молекул€рное уравнение: Na2S + ЌќЌ  NaЌS + NaќЌ
NaЌS + NaќЌ
 |
|
ЌS- + ЌќЌ  Ќ2S + ќЌ-
Ќ2S + ќЌ-
молекул€рное уравнение: NaЌS + ЌќЌ  Ќ2S + NaќЌ
Ќ2S + NaќЌ
—ледует отметить, что в таких случа€х гидролиз протекает, главным образом, по первой стадии и в очень малой степени. ѕо последующим стади€м он идет гораздо хуже и им можно пренебречь. Ёто св€зано с тем, что продукты, образующиес€ на поздних стади€х гидролиза (Ќ2S), €вл€ютс€ более сильными электролитами, чем продукты, образующиес€ на первой стадии (ЌS-), и лучше диссоциируют на ионы.
2. √идролиз соли, образованной слабым основанием и сильной кислотой:
Ќапример
Cu(OH)2 + H2SO4 = CuSO4 + 2 H2O
слабое сильна€
основание кислота
Zn(OH)2 + 2 HCl = ZnCl2 + 2 H2O
слабое сильна€
|
|
|
основание кислота
¬ водных растворах таких солей с H2O будут взаимодействовать катионы слабого основани€ (т.е. ионы металлов или NH4+), образующиес€ при диссоциации соли:
ZnCl2 → Zn 2+ + 2 Cl¯
ќни будут присоедин€ть к себе гидроксильные ионы, отщепившиес€ от молекул H2O, в результате чего образуетс€ слабый электролит (ZnќЌ+), а в растворе станут накапливатьс€ ионы Ќ+, которые сообщат раствору такой соли кислую реакцию.
Zn 2+ + ЌќЌ  ZnќЌ+ + Ќ+
ZnќЌ+ + Ќ+
¬ результате этого рЌ растворов солей, образованных слабым основанием и сильной кислотой, будет < 7, а в самом растворе станут присутствовать основна€ соль и сильна€ кислота.
¬ молекул€рном виде уравнение гидролиза в этом случае можно записать так:
ZnCl2 + ЌќЌ  ZnќЌCl + ЌCl
ZnќЌCl + ЌCl
„ем более слабым электролитом €вл€етс€ основание, образующее соль, тем сильнее протекает ее гидролиз.
≈сли соль образована многокислотным слабым основанием, то ее гидролиз может протекать в несколько стадий:
 Cu(NO3)2 Ѓ Cu2+ + 2 NO3 Ц
Cu(NO3)2 Ѓ Cu2+ + 2 NO3 Ц
|
 Cu2+ + ЌќЌ
Cu2+ + ЌќЌ  CuOH+ + Ќ+
CuOH+ + Ќ+
молекул€рное уравнение: Cu(NO3)2 + ЌќЌ  CuќЌNO3 + ЌNO3
CuќЌNO3 + ЌNO3

|
CuOH+ + ЌќЌ  Cu(ќЌ)2 + Ќ+
Cu(ќЌ)2 + Ќ+
молекул€рное уравнение: CuќЌNO3 + ЌќЌ  Cu(ќЌ)2 + ЌNO3
Cu(ќЌ)2 + ЌNO3
ќднако и в этом случае гидролиз протекает в малой степени только по первой стадии. ѕо последующим стади€м он будет идти крайне незначительно и им можно пренебречь.
3. √идролиз соли, образованной слабой кислотой и слабым основанием:
Ќапример
—Ќ3—ќќЌ + NЌ4OЌ = —Ќ3—ќќNЌ4 + Ќ2ќ
2 Al(OH)3 + 3 H2—O3 = Al2(—O3)3 + 6 Ќ2ќ
“акие соли лучше всего подвергаютс€ гидролизу, так как в этом случае с Ќ2ќ взаимодействуют и катионы основани€, и анионы кислоты:
—Ќ3—ќќNЌ4 → —Ќ3—ќќ‾ + NЌ4+
—Ќ3—ќќ‾ + ЌќЌ  —Ќ3—ќќЌ + ќЌ‾
—Ќ3—ќќЌ + ќЌ‾
NЌ4+ + ЌќЌ  NЌ4ќЌ + Ќ+
NЌ4ќЌ + Ќ+
ѕричем в растворе одновременно образуютс€ и ионы Ќ+ и ионы ќЌ‾. —реда в растворе такой соли может быть кислой, нейтральной или щелочной.
≈сли кислота и основание, образующие соль, €вл€ютс€ слабыми электролитами примерно одинаковой cилы, то катионы основани€ и анионы кислоты будут в одной и той же мере взаимодействовать с молекулами Ќ2ќ. „исло ионов Ќ+ и ќЌ¯ в растворе будет примерно равным. ќни вступ€т в реакцию между собой (Ќ+ + ќЌ¯ = Ќ2ќ) и среда в растворе такой соли останетс€ нейтральной.
≈сли же какой-то из электролитов (кислота или основание) будет слабее другого, то среда в растворе такой соли будет кислой или щелочной. “ак, если кислота, образующа€ соль, €вл€етс€ более слабым электролитом, чем основание, то анионы кислоты будут в большей степени взаимодействовать с Ќ2ќ, чем катионы основани€. ¬ результате этого ионов ќЌ¯ в растворе станет накапливатьс€ больше, чем ионов Ќ+ и среда в нем будет щелочной. ≈сли же основание, образующее соль, слабее, чем кислота, то среда в растворе такой соли, наоборот, будет кислой.
—оли, образованные и слабой кислотой, и слабым основанием, подвергаютс€ гидролизу в гораздо большей степени, чем все остальные, т.к. образующие ионы Ќ+ и ќЌ¯ св€зываютс€ в Ќ2ќ и равновесие в реакции гидролиза смещаетс€ в правую сторону.
¬ этом случае гидролиз часто протекает до конца не только по первой стадии, но и в значительной степени по последующим стади€м.
ƒл€ целого же р€да таких солей гидролиз €вл€етс€ необратимым и приводит к полному разложению такой соли на соответствующую кислоту и основание:
 —ќ2
—ќ2
 Al2(—O3)3 + 6 Ќ2ќ = 2 Al(OH)3↓ + 3 H2—O3
Al2(—O3)3 + 6 Ќ2ќ = 2 Al(OH)3↓ + 3 H2—O3
Ќ2ќ
Al2S3 + 6 Ќ2ќ = 2 Al(OH)3↓ + 3 H2 S↑
“акие соли в водных растворах вследствие этого существовать не могут. ¬ таблицах растворимости в клеточках, соответствующим данным сол€м, часто ставитс€ прочерк.
4. —оль, образованна€ сильной кислотой и сильным основанием, не гидролизуетс€:
Ќапример
NaOH + HCl = NaCl + H2O
сильное сильна€
основание кислота
2 KOH + H2SO4 = K2SO4 + 2 H2O
сильное сильна€
основание кислота
“акие соли гидролизу не подвергаютс€ и их водные растворы имеют нейтральную среду.
|
|
|
√идролизу подвергаетс€ не вс€ соль, а лишь еЄ часть, т.е. в растворе устанавливаетс€ равновесие между солью и образующими еЄ кислотой и основанием. ѕоэтому часть вещества, подвергающа€с€ гидролизу, характеризуетс€ степенью гидролиза. —тепень гидролиза зависит от константы равновеси€, температуры и концентрации соли, а также от еЄ природы. ѕрирода соли про€вл€етс€ в величине константы гидролиза соли.
онстанта гидролиза соли ( г) характеризует способность данной соли подвергатьс€ гидролизу. „ем больше г, тем в большей степени протекает гидролиз (при посто€нстве “ и концентрации соли).
онстанта гидролиза соли, образованной анионом слабой кислоты и катионом сильного основани€, равна:
г =  ,
,
где а Ц константа диссоциации кислоты. јнализ уравнени€ показывает, что чем слабее кислота, тем в большей степени подвергаютс€ гидролизу еЄ соли.
онстанта гидролиза соли, образованной анионом сильной кислоты и катионом слабого основани€, равна:
г =  ,
,
где b Ц константа диссоциации основани€. јнализ уравнени€ показывает, что чем слабее основание, тем в большей степени подвергаютс€ гидролизу им образованные соли.
онстанта гидролиза соли, образованной анионом слабой кислоты и катионом слабого основани€, равна:
г = 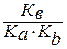
«ависимость степени гидролиза от концентрации соли про€вл€етс€ в том, что с разбавлением раствора степень гидролиза растЄт. ¬ли€ние температуры на степень гидролиза вытекает из рассмотренного ранее принципа Ће Ўателье: степень гидролиза растЄт с повышением температуры. ѕоэтому дл€ ослаблени€ гидролиза растворы необходимо хранить концентрированными и при низких температурах. ƒл€ солей, образованных анионом сильной кислоты и катионом слабого основани€, фактором подавлени€ гидролиза €вл€етс€ подкисление раствора, а в случае соли, образованной анионом слабой кислоты и катионом сильного основани€, таковым фактором €вл€етс€ подщелачивание раствора.
ак было показано выше, процесс гидролиза может протекать ступенчато, например:
 Na2CO3+H2O↔NaHCO3+NaOH 1 ступень, а,2
Na2CO3+H2O↔NaHCO3+NaOH 1 ступень, а,2
CO32- +H2O↔HCO3- +OH-
NaHCO3-+H2O↔H2CO3+OH- 2 ступень, а,1
HCO3- +H2O↔H2CO3+OH-
ак видно из уравнени€ реакции гидролиза, на первой ступени образуетс€ гидрокарбонат-ион, диссоциаци€ которого характеризуетс€ а,2 угольной кислоты, а во второй ступени происходит гидролиз кислой соли с образованием угольной кислоты, диссоциацию которой характеризует а,1 еЄ диссоциации. ѕоэтому можно св€зать константу гидролиза по первой ступени г, 1 со второй константой диссоциации кислоты а, 2, а константу гидролиза по второй ступени г, 2 с первой константой диссоциации кислоты а, 1:
г, 1 = 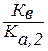 > г, 2 =
> г, 2 = 
√идролиз по первой ступени всегда протекает в большей степени, чем по второй. јналогично протекает гидролиз соли, образованной катионом слабого основани€ многовалентного металла.
ќсобенно глубоко протекает гидролиз соли, образованной анионом слабой кислоты и катионом слабого основани€, т.к. константа гидролиза дл€ этого случа€ обратно пропорциональна произведению констант диссоциации кислоты и основани€, т.е. еЄ значение крайне велико.
—огласно протонной теории кислот и оснований, гидролиз можно представить как частный случай кислотно-основного равновеси€: протон переходит от молекулы воды к данному иону или от данного иона к молекуле воды. ѕримером может служить гидролиз иона аммони€:
NH4++H2O↔H3ќ++NH3






