–ешение типовых задач
ѕример 10.1. ƒл€ данного материала: а) укажите, к каким ви-дам композитов он относитс€; б) перечислите его компоненты; в) дайте физико-химические характеристики армирующей фазы и матрицы; г) объ€сните причины изменени€ свойств при сочетании этих составл€ющих.
ƒан композит Ц углепластик (волокна из углерода, св€зующее Ц эпоксидна€ смола).
– е ш е н и е
“акой композит называетс€ полимерным или, более точно, композитом с полимерной матрицей.
омпоненты: армирующа€ фаза Ц углеродные волокна, св€зую-щее Ц эпоксидна€ смола.
јрмирующа€ фаза Ц на основе углерода. ≈го электронное строение: 6— 1 s 22 s 22 p 2, при возбуждении

” углерода 4 аллотропные модификации: алмаз (sp 3-гибри-дизаци€, тетраэдрическа€ структура); графит (sp 2-гибридизаци€, плоска€ структура); карбин (sp -гибридизаци€, черный порошок); фуллерен (сферическа€ структура, —60).
Ќа основе графита, а в последнее врем€ Ц фуллерена изготавливают углеродные волокна, в том числе и в виде нанотрубок. ¬ысока€ энерги€ св€зи —Ц— углеродных волокон позвол€ет им сохран€ть прочность почти до 2500 .
ћатрица Ц эпоксидна€ смола на основе эпоксигрупп  .
.
¬ведем обозначени€: ћ Ц все другие функциональные группы в смоле; R Ц в отвердителе (диамине). —хема отверждени€:



 CH2ЦCHЦMЦCHЦCH2 NH2 NH2
CH2ЦCHЦMЦCHЦCH2 NH2 NH2
| |
 O O + R R
O O + R R



 CH2ЦCHЦMЦCHЦCH2 | |
CH2ЦCHЦMЦCHЦCH2 | |
NH2 NH2
O O
OH OH
| |
CH2ЦCHЦMЦCHЦCH2
| |
NH NH
| |
 R R
R R
| |
NH NH
| |
ЕЦMЦCHЦCH2 CH2ЦCHЦMЦ Е
| |
OH OH
”лучшение физико-химических и механических характеристик полученного полимерного композита объ€сн€етс€ тем, что он содер-жит волокна высокой прочности; матрица должна в основном пере-распредел€ть напр€жени€ между армирующими волокнами. ¬ключе-ни€ нарушают водородные, ковалентные и электростатические св€зи между молекулами матрицы, обеспечива€ ориентацию ее свойств в том же направлении, что и у армирующей фазы.
ѕример 10.2. ƒл€ заданных компонентов определите их термо-динамическую совместимость в случае получени€ композиционного материала.
«аданы компоненты: Nb3B4; Cr5B3.
– е ш е н и е
ѕо табл. ѕ.11 находим химические потенциалы заданных веществ, э¬: 1) Nb3B4 0,64; 2) Cr5B3 0,59.
¬еличина химического потенциала определ€етс€ формулой
μ i = 
где: Δ G Ц изменение энергии √иббса; ni, nj Ц концентрации данного компонента; – Ц давление; “ Ц температура. »ндекс j показывает, что посто€нны все концентрации, за исключением ni.
—огласно теории твердофазных реакций, химические превращени€ протекают до выравнивани€ химических потенциалов. Ќаходим разность химических потенциалов заданных веществ:
μ1 Ц μ2 = 0,64 Ц 0,59 = 0,05 э¬.
ѕоскольку данна€ величина меньше 10 % от минимального значени€ химического потенциала (μ2), уровень возможного взаимодействи€ нельз€ считать интенсивным, следовательно, компоненты термодинамически совместимы.
|
|
|
ѕример 10.3. ƒл€ композита с расплавленной матрицей (Ge с растворенным в нем кислородом) и армирующей фазой (в виде смеси порошков оксидов ZrO2 и —аќ) рассчитайте:
а) содержание кислорода в расплаве, пользу€сь методом Ёƒ—, если дано: “ = 1233 ; Δ G  = 149,64 кƒж/моль; ≈
= 149,64 кƒж/моль; ≈  = 0,925 ¬;
= 0,925 ¬;
б) величину Δ G  при температурах, : 1233, 1240, 1250.
при температурах, : 1233, 1240, 1250.
– е ш е н и е
а) —оставл€ем электрическую цепь с платиновыми электродами и компонентами композита.



 (Ц) Pt раствор ќ ZrO2+CaO воздух Pt (+)
(Ц) Pt раствор ќ ZrO2+CaO воздух Pt (+)
в Ge (ж) (ќ2)
–еакции на электродах:
јнод, окисление атод (восстановление)
(левый электрод) (правый электрод)
ќ2Ц = ќGe (ж) + 2 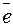 ; ½ ќ2 (г) + 2
; ½ ќ2 (г) + 2 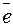 = ќ2Ц;
= ќ2Ц;
—уммарна€ реакци€: ½ ќ2 (г) = ќGe (ж).
ѕо формуле “омсона
Δ G 1233 = Ц zF ≈  = Ц 2Ј96500Ј0,925Ј10Ц3= 178,525 кƒж/моль.
= Ц 2Ј96500Ј0,925Ј10Ц3= 178,525 кƒж/моль.
ѕо уравнению изотермы ¬ант-√оффа
Δ G 1233 = Δ G  + RT ln
+ RT ln  ,
,
где: с 0 Ц концентраци€ кислорода в расплавленном германии, масс. %; – 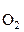 Ц парциальное давление кислорода в атмосфере (0,2 атм. или 0,2026 ћѕа).
Ц парциальное давление кислорода в атмосфере (0,2 атм. или 0,2026 ћѕа).
¬ качестве стандартного состо€ни€ выбраны значени€ 1 масс. % и 1 атм. (0,1013 ћѕа), соответственно.
ln  =
=  =
=  = Ц2,818;
= Ц2,818;
 = 0,0597; с 0 = 0,0597Ј0,21/2 = 0,027 масс. %.
= 0,0597; с 0 = 0,0597Ј0,21/2 = 0,027 масс. %.
ќтсюда Ц содержание кислорода в расплавленном германии при 1233 составл€ет 0,027 масс. %.
б) ƒл€ расчета величины Δ G  используем формулу “омсона
используем формулу “омсона
Δ G  = Ц zFE
= Ц zFE  .
.
»з табл. ѕ.14 находим значени€ E  при заданных температурах
при заданных температурах
“ = 1233 , ≈  = 0,775 ¬; “ = 1240, ≈
= 0,775 ¬; “ = 1240, ≈  = 0,774 ¬;
= 0,774 ¬;
“ = 1250 , ≈  = 0,773 ¬.
= 0,773 ¬.
 = Ц2Ј96500Ј0,775Ј10Ц3 = Ц149,6 кƒж/моль.
= Ц2Ј96500Ј0,775Ј10Ц3 = Ц149,6 кƒж/моль.
јналогично, дл€ других температур получаем:
 = Ц149,4 кƒж/моль;
= Ц149,4 кƒж/моль;  = Ц149,2 кƒж/моль.
= Ц149,2 кƒж/моль.
— ростом температуры отмечаетс€ тенденци€ к снижению Δ G  по абсолютному значению, т.е. глубина протекающих реакций уменьшаетс€, это, веро€тно, св€зано с изменени€ми свойств расплавленного германи€.
по абсолютному значению, т.е. глубина протекающих реакций уменьшаетс€, это, веро€тно, св€зано с изменени€ми свойств расплавленного германи€.
ѕример 10.4. –ассчитайте константу равновеси€ дл€:
а) твердофазной химической реакции
TiO2 (т) + C (т) = TiC (т) + 2CO (г)
при температуре 1700 ;
б) реакции в растворе при химическом восстановлении покрытий на армирующую фазу; восстанавливающийс€ ион Ni2+, восстановитель N2H4, активности компонентов, моль/л: Ni2+ 0,1; N2H4 1,0.
– е ш е н и е
»з табл. ѕ 1 выписываем уравнение дл€ расчета Δ G 
Δ G  = 221 Ц 0,136 “, кƒж/моль (—ќ (г)).
= 221 Ц 0,136 “, кƒж/моль (—ќ (г)).
Δ G 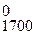 = 221 Ц 0,136Ј1700 = Ц10,2 кƒж/моль (—ќ (г));
= 221 Ц 0,136Ј1700 = Ц10,2 кƒж/моль (—ќ (г));
Δ G 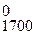 = Ц RT ln K p; K p =
= Ц RT ln K p; K p =  .
.
ln K p = Ц  = Ц
= Ц  = 0,723.
= 0,723.
K p = 2,061.
ѕолученное значение K p указывает на протекание процесса с небольшой интенсивностью, это указывает на устойчивость композита на основе TiO2 (т) и — (т). ¬ то же врем€, когда по этой реакции получают порошки TiC (т) дл€ композитов, следует использовать более высокие температуры, чем 1700 дл€ интенсификации процесса.
б) ѕользу€сь табл. ѕ.1, ѕ.15 составл€ем сокращенное ионное и полное молекул€рное уравнени€ реакций
N2H4 + 4OH  =
= N2 + 4H2O + 4 =
= N2 + 4H2O + 4 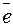
| восстановитель | 
| |
Ni2+ + 2  = Ni0 = Ni0
| окислитель |  = Ц0,25 ¬ = Ц0,25 ¬
|
|
|
|
4 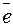

2NiSO4 + N2H4 + 4KOH = 2Ni + N2 + 4H2O + 2K2SO4;


 4
4 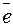
2Ni2+ + N2H4 + 4OH  = 2Ni0 + N2 + 4H2O.
= 2Ni0 + N2 + 4H2O.
Δ G  = Ц zF (
= Ц zF ( +
+  Ц
Ц  ) = Ц RT ln K p;
) = Ц RT ln K p;
ln K p =  = 137,173;
= 137,173;
K p = 3,745Ј1059,
т.е. реакци€ практически необратима.
ѕример 10.5. Ќапишите схему реакции полимеризации изобу-тилена и определите степень полимеризации, если полученный полимер имеет среднюю молекул€рную массу M n = 61600 г/моль.
– е ш е н и е






