1. јктуальность темы:
†††††† ѕоддержание кислотно-основного и электролитного равновеси€ в ткан€х, биологических жидкост€х крайне важно дл€ нормального функционировани€ организма. »зменение электролитного состава приводит к изменению реакции среды биологических жидкостей. ¬о-первых, ионы Ќ+ оказывают каталитическое действие на многие биохимические превращени€. ¬о-вторых, ферменты и гормоны про€вл€ют биологическую активность только в строго определенном интервале значений рЌ (например, фермент пепсин, участвующий в расщеплении пищи в желудке, активен только при рЌ = 1,5). ¬-третьих, даже небольшие изменени€ концентрации ионов Ќ+ ощутимо вли€ют на величину осмотического давлени€ в этих жидкост€х. ѕоддержание посто€нного рЌ в крови и тканевых жидкост€х регулируетс€ с помощью нескольких буферных систем. ќдним из способов диагностики заболеваний €вл€етс€ определение рЌ желудочного сока, крови, мочи.
2. ÷ель обучени€:
† «нать основные положени€ теории электролитической диссоциации; пон€тие о сильных и слабых электролитах; пон€тие о буферных растворах; механизм действи€ буферных растворов; буферные системы организма. ”меть решать расчетные задачи по вычислению концентрации ионов водорода и рЌ растворов сильных и слабых электролитов; решать расчетные задачи на вычисление рЌ буферных растворов; буферной Ємкости.
3. ¬опросы дл€ обсуждени€:
1. ƒиссоциаци€ воды. »онное произведение воды.
2. ¬одородный показатель. Ўкала рЌ. рЌ биологических жидкостей. јцидоз, алкалоз.
3. “еори€ кислот и оснований јррениуса, Ѕренстеда-Ћоури, Ћьюиса.
4. “ипы протолитических реакций: нейтрализации, гидролиза, ионизации.
5. √идролиз солей, типы гидролиза. —тепень и константа гидролиза. ‘акторы, вли€ющие на степень гидролиза.
6. –оль гидролиза в биохимических процессах.
7. Ѕуферные растворы. лассификаци€ и химический состав.
8. ћеханизм буферного действи€ (гидрокарбонатного, фосфатного, ацетатного, аммиачного).
9. –асчЄт pH буферных растворов. ”равнени€ √ендерсонаЦ’ассельбаха дл€ кислотного и основного буферных растворов.
10. Ѕуферна€ Ємкость. ќпределение. ‘ормулы дл€ расчЄта. ‘акторы, вли€ющие на буферную Ємкость.
11. Ѕуферные системы крови. ѕон€тие о кислотно-основном равновесии.
4. —итуационные задачи:
| є задачи | “екст задани€: |
| 1. | –ассчитать [H+] и рЌ раствора гидроксида натри€, —=0,1 моль/л, α=100% |
| 2. | –ассчитать [H+] и рЌ раствора уксусной кислоты, —=0,01 моль/л, α=0,042 |
| 3. | —тепень диссоциации слабой одноосновной кислоты в 0,2 ћ растворе равна 0,03. ¬ычислить значение [H+], [ќH-] и рќЌ дл€ этого раствора. |
| 4. | ¬ычислить pH буферного раствора, приготовленного из 200 мл раствора CH3COOH (с=0,1 моль/л) и 100 мл CH3COONa (c=0,2 моль/л). ак изменитс€ pH этого раствора при добавлении к нему 10 мл раствора NaOH (с=0,1моль/л) и при разбавлении в 15 раз? |
| 5. | –ассчитать pH буферного раствора, приготовленного из 200 мл раствора NH4Cl (c=0,1 моль/л) и 150 мл NH3ХH2O (c=0,25 моль/л) |
| 6. | 100 мл крови добавили 25 мл раствора HCl (c=0,05моль/л), при этом значение pH изменилось от 7,36 до 6,9. –ассчитать буферную Ємкость крови по кислоте. |
| 4. | ¬ычислить pKa молочной кислоты, если pH раствора в 1 литре которого содержитс€ 0,01 моль молочной кислоты и 0,0139 моль лактат-иона (анион молочной кислоты) равен 4,0. |
| є задачи | Ёталоны ответов: |
| 1. | NaOH → Na+ ††+ OH- [OH-] = C ∙ α = 0.1моль-ион/л [Ќ+] = 10-14 = 10-13††††††††††††††† †††††††††††††††††††††††††††рЌ= -lg10-13 = 13 ††††††† 10-1 |
| 2. | CH3COOH → H+ + CH3COO- [H+] = C ∙ α = 0.01 ∙ 0.042 = 4.2 ∙ 10-4 ††††††††††††††††††pH = -lg 4.2 ∙ 10-4 = 4 Ц lg4.2 = 3.38 |
| 3. | HA → H+ + A- [H+] = C ∙ α; [H+] = 0.2 ∙ 0.03 = 6 ∙ 10-3 [OH-] = 1∙10-14 = 1.7∙10-12 ††††††††††††††††††††††††††††††††††pOH = -lg 1.7∙10-12 = 12-lg1.7 = 11.78 †††††† 6∙10-3 |
| 4. | ј) 
 ; ;  =4.74
б) ѕри добавлении раствора NaOH: =4.74
б) ѕри добавлении раствора NaOH: 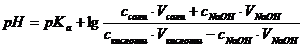
  »зменение pH составл€ет 4.78-4.74=0.04
¬) ѕри разбавлении в 15 раз:
»зменение pH составл€ет 4.78-4.74=0.04
¬) ѕри разбавлении в 15 раз:  ѕри разбавлении буферного раствора pH не изменетс€.
ѕри разбавлении буферного раствора pH не изменетс€.
|
| 5. |  ††††††††††††††††† ††††††††††††††††† 

|
| 6. | 
|
| 7. | ¬ соответствии с уравнением √ендерсона-’ассельбальха:
 
|
|
|
|
1. ¬ыполнить тестовые задани€ (письменно):
| 1. | ѕо протолитической теории јррениуса основани€ - это электролиты, диссоциирующие в растворе с образованием ионов: | |
| A | HCOOЦ | |
| B | ќЌ- | |
| C | —ќ32- | |
| D | Ќ+ | |
| 2. | ѕо протолитической теории јррениуса кислоты - это электролиты, диссоциирующие в растворе с образованием ионов: | |
| † | A | Ќ+ |
| B | + | |
| C | Na+ | |
| D | ќЌ- | |
| 3. | ¬ыделите электролиты: | |
| A | сера, сахар | |
| B | бензол, этилен | |
| C | ацетат Na, циановодородна€ кислота | |
| D | вода, глюкоза | |
| 4. | »онна€ сила плазмы крови человека равна 0,15 ћ; заменителем плазмы крови €вл€етс€: | |
| A | 0,7 м раствор глюкозы | |
| B | 0,15 м раствор карбоната натри€ | |
| C | 0,9% раствор хлорида натри€ | |
| D | 9% раствор хлорида натри€ | |
| 5. | ¬одородный показатель рЌ характеризует: | |
| A | кислотность | |
| B | ионную силу | |
| C | образование осадка | |
| D | в€зкость | |
| 6. | ≈сли рЌ раствора равен 8, то концентраци€ ионов ќЌЦ равна: | |
| A | lg 10-8 | |
| B | 10-6 | |
| C | 10-8 | |
| D | 10-2 | |
| 7. | рЌ 0,0001 н раствора ќЌ равен: | |
| A | 12 | |
| B | 10 | |
| C | 11 | |
| D | 4 | |
| 8. | ѕри приеме внутрь раствора гидрокарбоната натри€ рЌ желудочного сока: | |
| A | уменьшаетс€ | |
| B | увеличиваетс€ | |
| C | не измен€етс€ | |
| 9. | рЌ раствора при равных концентраци€х больше у: | |
| A | HCN | |
| B | NaOH | |
| C | NH4OH | |
| D | Ќ—l | |
| 10. | ѕроцесс гидролиза Ц это: | |
| A | растворение соли в воде | |
| B | распад вещества на ионы | |
| C | взаимодействие ионов соли с составными част€ми воды | |
| 11. | —ледствием гидролиза €вл€етс€: | |
| A | разбавление раствора | |
| B | изменение активной концентрации ионов соли | |
| C | образование слабого электролита (электролитов) | |
| D | накопление ионов Ќ+ или ќЌЦ в растворе | |
| 12. | √идролизу соли, образованной слабой кислотой и сильным основанием, соответствует уравнение: | |
| A | —Ќ3—ќќЦ + Ќ2ќ = —Ќ3—ќќЌ + ќЌЦ | |
| B | NH4+ + H2O = NH4OH + H+ | |
| C | NH4+ + CH3COOЦ + H2O = NH4OH + CH3COOH | |
| D | Al3+ + H2O = AlOH2+ + H+ | |
| 13. | √идролизу соли, образованной сильной кислотой и слабым основанием, соответствует уравнению: | |
| A | HCOOЦ + HOH = OHЦ + HCOOH | |
| B | NH4+ + HOH = H+ + NH4OH | |
| C | Al3+ + CH3COOЦ + HOH = AlOH2+ + CH3COOH | |
| D | NH4+ + CNЦ + HOH = NH4OH + HCN | |
| 14. | √идролиз ј“‘ в организме Ц источник: | |
| A | ионов водорода | |
| B | воды | |
| C | белков | |
| D | энергии | |
| 15. | Ѕуферные растворы Ц это система: | |
| A | поддерживающа€ посто€нство рЌ при добавлении небольших количеств кислот, щелочей, при разбавлении | |
| B | измен€юща€ рЌ при добавлении сильных кислот, щелочей | |
| C | измен€юща€ рЌ при разбавлении | |
| D | не измен€юща€ рЌ при добавлении больших количеств сильных кислот, щелочей | |
| 16. | ¬ыбрать из предлагаемых веществ возможные компоненты дл€ приготовлени€ буферных растворов: | |
| A | NaCl, HHb, NaHb, HCl | |
| B | NaHCO3, CO2∙H2O, NaH2PO4, Na2HPO4 | |
| C | NaHCO3, CO2∙H2O, Na2SO4, H2SO4 | |
| D | —Ќ3—ќќNa, —Ќ3—ќќЌ, NH4OH, NH4Cl | |
| 17. | ќт каких факторов зависит pH буферного раствора? | |
| A | ќт концентрации компонентов | |
| B | ќт соотношени€ концентрации компонентов | |
| C | ќт константы диссоциации слабого электролита | |
| D | ќт константы гидролиза соли | |
| 18. | рЌ крови в норме: | |
| A | 1. 6,80-6,90 | |
| B | 2. 7,90-8,02 | |
| C | 7,36-7,42 | |
| D | 7,42-7,50 | |
| 19. | ¬ плазме крови (рЌ=7,36-7,40) присутствуют буферные растворы: | |
| A | белковый | |
| B | аммиачный | |
| C | фосфатный | |
| D | бикарбонатный (гидрокарбонатный) | |
| 20. | ¬ эритроцитах (рЌ=7,25) наход€тс€ следующие буферные системы: | |
| A | гемоглобинова€ | |
| B | аммиачна€ | |
| C | бикарбонатна€ | |
| D | фосфатна€ |
6. «адачи дл€ самосто€тельного решени€ (выполнить письменно):
|
|
|
1. †»зобразить схемы гидролиза (в молекул€рном, ионном и сокращенном ионном виде) следующих солей, указать характер среды в растворе:
а) Na2S
б) †NH4Cl
†
в) NO3
2. –ассчитать [H+] и рЌ раствора H2SO4, C(H2SO4) = 0,01 моль/л, α = 100%
3. ќпределить pH раствора, в 1л которого содержитс€ 0,1 г NaOH, α=100%.
4. –ассчитать рЌ желудочного сока, если массова€ дол€ сол€ной кислоты в желудочном соке
равна 0,45%.
5. –ассчитать pH гидрокарбонатного буферного раствора, состо€щего из 15 мл раствора
NaH—O3, — = 0,1 моль/л и 50 мл раствора H2—O3, — = 0,02 моль/л. (pKa = 6,35).
8. –ассчитать рЌ аммиачного буферного раствора, приготовленного при смешивании 200 м놆†
раствора NЌ 4CI, — (NH4CI) = 0,1 моль/л и 100 мл раствора NЌ4OH, — (NH4ќЌ) = 0,2 моль/л
(pKb(NH4ќЌ) = 4,75).
9. 25 мл ацетатного буфера с рЌ 4,3 прибавили 5,0 мл 0,1M HCl, что привело к снижению рЌ до 3,7. –ассчитать буферную емкость по кислоте.
|
|
|
7. ¬спомогательные материалы по самоподготовке:
ѕриложение 1
“аблица трехзначных дес€тичных логарифмов






