1. Ёлектронный баланс - метод нахождени€ коэффициентов в уравнени€х окислительно-восстановительных реакций, в котором рассматриваетс€ обмен электронами между атомами элементов, измен€ющих свою степень окислени€. „исло электронов, отданное восстановителем равно числу электронов, получаемых окислителем.
2. Ёлектронно-ионный баланс (метод полуреакций) метод нахождени€ коэффициентов, в котором рассматриваетс€ обмен электронами между ионами в растворе с учетом характера среды:
| 2Cl1- Ц 2ē Ѓ | Cl20 | 5 | |
| MnO41- + 8H+ | + 5ē Ѓ | Mn2+ + 4H2O | 2 |
| 7+ | 2+ |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
10Cl- + 2MnO41- + 16H+ Ѓ 5Cl20≠ + 2Mn2+ + 8H2O
(дл€ уравнивани€ ионной полуреакции используют H+, OH- или воду)
“ипичные реакции окислени€-восстановлени€.
1.–еакции с участием перманганата кали€ в качестве окислител€.
ѕри взаимодействии перманганата кали€ с восстановителем образуютс€ различные продукты восстановлени€ в зависимости от pH среды.
–еакции в кислой среде.
5K2S+4O3 + 2KMn+7O4 + 3H2SO4 Ѓ 6K2S+6O4 + 2Mn+2SO4 + 3H2O
| MnO4- + 8H+ + 5ē Ѓ Mn2+ + 4H2O | 2 |
| SO32- + H2O Ц 2ē Ѓ SO42- + 2H+ | 5 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
2MnO4- + 16H+ + 5SO32- + 5H2O Ѓ 2Mn2+ + 8H2O + 5SO42- + 10H+
или 2MnO4- + 6H+ + 5SO32- Ѓ 2Mn2+ + 3H2O + 5SO42-
–еакции в нейтральной среде
3K2S+4O3 + 2KMn+7O4 + H2O Ѓ 3K2S+6O4 +2Mn+4O2¯ + 2KOH
| MnO41- + 2H2O + 3ē Ѓ MnO2 + 4OH- | 2 | |
| SO32- + 2OH- - 2ē Ѓ SO42- + H2O | 3 | |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
2MnO4- + 4H2O + 3SO32- + 6OH- Ѓ 2MnO2 + 8OH- + 3SO42- + 3H2O
или 2MnO4- + H2O + 3SO32- Ѓ 2MnO2 + 2OH- + 3SO42-
–еакции в щелочной среде.
K2S+4O3 + 2KMn+7O4 + 2KOH Ѓ K2S+6O4 +2K2Mn+6O4 + H2O
| SO32- + 2OH- - 2ē Ѓ SO42- + H2O | 1 |
| MnO41- + ē Ѓ MnO42- | 2 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
SO32- + 2OH- + 2MnO4- Ѓ SO42- + H2O + 2MnO42-
2.–еакции с дихроматом кали€ в качестве окислител€
—тепень окислени€ хрома понижаетс€ с +6 до +3.
1.
K2Cr2+6O7 + 3H2S-2 + 4H2SO4 Ѓ K2SO4 + Cr2+3(SO4)3 + 3S0¯ + 7H2O
| Cr2O72- + 14H+ + 6ē Ѓ 2Cr3+ + 7H2O | 1 | |
| H2S0 - 2ē Ѓ S0 + 2H+ | 3 | |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
Cr2O72- + 8H+ + 3H2S Ѓ 2Cr3+ + 7H2O + 3S0
2.
K2Cr2+6O7 + 6Fe+2SO4 + 7H2SO4 Ѓ 3Fe2+3(SO4)3 + K2SO4 + Cr2+3(SO4)3 + 7H2O
| Cr2O72- + 14H+ + 6ē Ѓ 2Cr3+ + 7H2O | 1 |
| Fe2+ - ē Ѓ Fe3+ | 6 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
6Fe2+ + Cr2O72- + 14H+ Ѓ 2Cr3+ + 6Fe3+ + 7H2O
3.
K2Cr2+6O7 + 14HCl-1 Ѓ 3Cl20≠ + 2KCl + 2Cr+3Cl3 + 7H2O
| Cr2O72- + 14H+ + 6ē Ѓ 2Cr3+ + 7H2O | 1 |
| 2Cl1- - 2ē Ѓ Cl20 | 3 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
Cr2O72- + 6Cl- + 14H+ Ѓ 2Cr3+ + 3Cl20 + 7H2O
3.ѕероксид водорода в окислительно-восстановительных реакци€х
ќбычно пероксид водорода используют как окислитель:
|
|
|
H2O2 + 2HI-1 Ѓ I20 + 2H2O
| 2I- - 2ē Ѓ I20 | 1 |
| H2O2 + 2H+ + 2ē Ѓ 2H2O | 1 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
2I- + H2O2 + 2H+ Ѓ I2 + 2H2O
ѕри действии сильных окислителей пероксид водорода может окисл€тьс€, образу€ кислород и воду.
5H2O2 + 2KMn+7O4 + 3H2SO4 Ѓ 5O20≠ + K2SO4 + 2Mn2+SO4 + 8H2O
| MnO4- + 8H+ + 5ē Ѓ Mn2+ + 4H2O | 2 |
| H2O2 - 2ē Ѓ O2 + 2H+ | 5 |
ЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦЦ
2MnO4- + 5H2O2 + 16H+ Ѓ 2Mn2+ + 8H2O + 5O2 + 10H+
или 2MnO4- + 5H2O2 + 6H+ Ѓ 2Mn2+ + 8H2O + 5O2
Ћабораторна€ работа.
Ђ ачественные раекции на анионы 1-3 группї
≤ группа анионов.
1. ƒействие группового реактива на анионы ≤ группы BaCl2.
¬ п€ть пробирок налить по 2-3 капли растворов солей анионов: S042-, S2032-, C032- и прибавить в каждую пробирку во 2 капли раствора BaCl2. полученным осадкам прибавить при перемешивании по 3-4 капли 2ћ хлороводородной кислоты и сделать вывод о растворимости осадков.
–≈ј ÷»» —”Ћ№‘ј“-»ќЌј SO42-
1. ƒействие ионов - —а2 +.
—а2+ + S042- †††††††††† ††††—аS04†††††††††
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
2 -3 капл€м раствора соли Na2Sќ4 прибавить 3 капли раствора соли —а2+. ¬ыпавший осадок поместить на предметное стекло и рассмотреть кристаллы.††††
–≈ј ÷»» —”Ћ№‘»“ - »ќЌќ¬ SO32-.
1. ƒействие окислителей (KMnO4).
5K2SO3 + 2KMnO4 + 3H2SO4 Ѓ 6K2SO4 + 2MnSO4 + 3H2O
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»» †††††††
2-3 капл€м раствора перманганата кали€ прибавить 3-4 капли 1ћ раствора серной кислоты и по капл€м добавить раствор Na2SO3 до обесвечивани€ реакционной смеси.
–≈ј ÷»» “»ќ—”Ћ№‘ј“ Ц »ќЌј S2O32-
1. –еакци€ с HCl.
S2O32− + 2H+ → S + SO2 + H2O
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
†††††† 2- 3 капл€м раствора тиосульфата натри€ прибавить 2 капли 2 ћ раствора хлороводородной кислоты. Ќаписать, что наблюдаетс€ и уравнение реакции.
2.ƒействие окислителей
2S2O32− + I2 → S4O62− + 2I−
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
¬ одну пробирку к 2-3 капл€м раствора I2 †прибавить 2-3 капли раствора тиосульфата натри€, в другую пробирку также к 2-3 капл€м раствора перманганата кали€ прибавить 2 капли 1 ћ раствора серной кислоты и 3-4 капли раствора тиосульфата натри€. ќпишите наблюдени€ и составьте уравнени€ реакций.
3. –еакци€ с AgNO3.
Ag+ + 2S2O32− → [Ag(S2O3)2]3−
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
3 капл€м раствора нитрата серебра прибавить 2 кали раствора тиосульфата натри€ до образовани€ осадка, а затем добавить избыток того же тиосульфата до растворени€ осадка.
†††††††††††
–≈ј ÷»» ј–ЅќЌј“ Ц »ќЌј CO32 --.
1, –еакции с кислотами.
Na2C03+2HCl=2NaCl+H20+CO2
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
¬ пробирку с газоотводной трубки поместить 10 капель раствора Na2C03 и равный объем 2ћ раствора HCl. ¬ыделившейс€ газ пропустить через газоотводную трубку в другую пробирку с известковой водой. „то наблюдаетс€?
2.–еакци€ с AgN03.
AgN03 + Na2C03 = 2NaN03 + Ag2C03
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
2-3 капл€м раствора карбоната натри€ прибавить 2 капли раствора нитрата серебра. ѕолученный осадок исследвать на растворимость в азотной кислоте.
–≈ј ÷»» ‘ќ—‘ј“ Ц »ќЌџ P043-.
1. –еакци€ с магнезильной смесью. (MgCl2 + NH40 H + NH4 Cl). –еакци€ фармокопейна€.
–еакци€ протекает по уравнению:
†††††††††††††††††† Na2HP04 + MgCl2 + NH40H ↔ MgNH4P04 ↓ + 2 NaCl + H20
|
|
|
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
2 -3 капл€м раствора MgCl2 прибавить 2-3 капли раствора NH40H, образовавшийс€ осадок Mg(0H)2 растворить в NH4Cl прибавл€€ его по капл€м. ѕолученную смесь нагреть на вод€ной бане и прибавить раствор Na2HPO4 до получени€ осадка. „то наблюдаетс€?
2. ƒействие молибденовой жидкости (смесь (NH4)2 Mo04 c HN03)
ѕри реакции данного реактива с фосфат Ц ионами образуетс€ желтый кристаллический осадок фосформолибдата аммони€.
Na3P04 + 3 NH4Cl +12(NH4)2Mo04 + 24HN03 → (NH4)3H4[P(Mo207)6↓ +
†24 NH4N03 + + 3 NACl + 10 H2O
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
5 капл€м подогретой молибденовой жидкости постепенно при перемешивании добавить 2 капли раствора фосфата и гидрофосфата натри€.
≤≤ группа анионов.
1.ƒействие группового реактива на анионы II группы AgNO3
Ќитрат серебра осаждает анионы II группы в виде галогенидов серебра AgCl Ц белого цвета, AgBr Ц желтовато Ц белого цвета, AgI Ц желтого цвета. ќсадки не раствора€ютс€ в азотной кислоте. ’орошо раствор€етс€ в концентрированном растворе аммиака AgCl с образованием белого комплекса диамина серебра [Ag(NH3)2]Cl. AgBr с трудом раствор€етс€ в аммиаке, а AgI вообще не раствор€етс€ в аммиаке.
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
¬ три пробирки поместить по 3-4 капли растворов NaCl, KBr и 1, прибавить по 3 капли раствора нитрата серебра. ¬ыпавшие осадки испытать на растворимость в азотной кислоте и в концентрированном аммиаке. Ќаписать уравнени€ реакций.
–≈ј ÷»» ’Ћќ–»ƒ-»ќЌј.††††††
1 ƒействие окислителей.
†††††† —ильные окислители ( ћп04, 2—г207, —103, ћп02) в кислой среде окисл€ют ионы —1- до свободного хлора —12:†††††††
†††††††††††††††††† 2 KCI + MnO2 + 2 H2SO4 → —l2 ↑ + K2S04 +MnS04 + 2 H2O
¬ыделение хлора можно обнаружить по запаху или по посинению иодкрахмальной бумажки.
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
5 капл€м раствора —l прибавить 3 капли конц. серной кислоты, немного твердого ћп02 и нагреть под т€гой. ќпределить C12 помощью иодкрахмальной бумажки.
††††††
–≈ј ÷»» Ѕ–ќћ»ƒ-»ќЌј.
I. ƒействие окислителей.
ќкислители Ц ћп04,ћп02, —12 и др.в кислой среде способны окисл€ть бромид- ионы до свободного брома, который хорошо раствор€етс€ в хлороформе, окрашива€ его в оранжевый цвет. –еакци€ фармакопейна€.
—12 + 2 ¬г- → 2 —1- + ¬г2
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
5 капл€м раствора бромида натри€ прибавить 1мл хлороформа,
2 капли 2ћ серной кислоты и затем по капл€м, при встр€хивании,2-3 капли хлорной воды. Ќаблюдаетс€ окрашивание сло€ хлороформа.
†††††† ¬ыделение свободного брома можно также обнаружить по реакции с флюоресцеином, который, взаимодейству€ с парами брома, превращаетс€ в тетра Ц бромфлюоресцеин (эозин) красного цвета.
–≈ј ÷»» »ќƒ»ƒ - »ќЌј.
1.ƒействие окислителей. –еакци€ фармакопейна€.
ќкислители легче, окисл€ют иодид-ионы до свободного иода, чем хлорид и бромид- ионы, что облегчает определение иодид - иона в присутствии других галогенидов. „аще всего, в этом случае дл€ окислени€ используют хлорную воду. ¬ первую очередь окисл€ютс€ I- - ионы и свободный I2 окрашивает слой органического растворител€ (бензол, толуол) в малиновый цвет.††††††††
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
3 капл€м раствора иодида кали€ прибавить 4 капли 2ћ раствора серной кислоты, 6 капель органического растворител€ и по капл€м при интенсивном встр€хивании хлорную воду. Ќаблюдать окрашивание раствора†
Ќаписать уравнени€ реакции
2. –еакции с сол€ми свинца Ќитрат или ацетат свинца (II) из растворов иодида осаждает желтый иодид свинца, который хорошо раствор€етс€ в гор€чей воде. ѕри охлаждении полученного раствора –bl2 выдел€етс€ в виде золотистых чешуек.
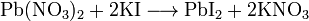
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
5 капл€м раствора иодита кали€ прибавить столько же раствора соли свинца(II). образующемус€ желтому осадку прибавить около 2 мл воды,3 капли уксусной кислоты и нагреть смесь до полного растворени€ осадка. ѕолученный раствор охладить и наблюдать выпадение золотистых чешуек иодида свинца. Ќаписатъ уравнение реакции.
|
|
|
Ў группа анионов.
¬ отличии от анионов 1 и II группы, анионы Ў группы с катионами серебра и бари€ образуют растворимые в воде соли. √руппового реактива дл€ анионов третьей группы нет.
–≈ј ÷»» Ќ»“–ј“ - »ќЌќ¬.
1. ƒействие дифениламина (C6Ќ5)2NH. –еакци€ фармокопейна€.
†††††† ƒифениламин в кислой среде окисл€етс€ N03- ионами до хиноидного соединени€ синего цвета. Ќитрит - ионы NO-2, ионы хромата Ц—r042- †и некоторые другие окислители также дают синее окрашивание, что надо иметь в виду при разделении смесей анионов.

¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
3-4 капл€м дифениламина в концентрированной серной кислоте прибавить 1-2 капли раствора нитрата.
–≈ј ÷»» ј÷≈“ј“ - »ќЌќ¬ (—Ќ3—ќќ-)
1.ƒействие сильных кислот.
†††††† ѕри действии сильных кислот на соли уксусной кислоты выдел€етс€ свободна€ уксусна€ кислота, обладающа€ характерным запахом.
¬џѕќЋЌ≈Ќ»≈ –≈ј ÷»»
¬ пробирку поместить 5 капель раствора ацетата натри€ и столько же капель 1 ћ серной кислоты, перемешать и слегка подогреть. ќбразование уксусной кислота проверить по запаху.






