ѕри решении задач этого раздела см. табл. 9 и прилож. 2.
»онно-молекул€рные, или ионные, уравнени€ реакций обмена отражают состо€ние электролита в растворе. ¬ этих уравнени€х сильные растворимые электролиты, поскольку они полностью диссоциированы, записывают в виде ионов, а слабые электролиты, малорастворимые и газообразные вещества, записывают в молекул€рной форме.
¬ ионно-молекул€рном уравнении одинаковые ионы из обеих его частей исключаютс€. ѕри составлении ионно-молекул€рных уравнений следует помнить, что сумма электрических зар€дов в левой части уравнени€ должна быть равна сумме электрических зар€дов в правой части уравнени€.
ѕример 1. Ќапишите ионно-молекул€рные уравнени€ реакций взаимодействи€ между водными растворами следующих веществ:
а) Ќ—1 и NaOH; б) –b(NO3)2 и Na2S; в) NaCIO и ЌNќ3; г) 2—ќз и H2Sќ4; д) —Ќз—ќќЌ и NaOH.
–ешение. «апишем уравнени€ взаимодействи€ указанных веществ в молекул€рном виде
а) Ќ—1 + NaOH = NaCI + H2O;
б) Pb(Nќ3)2 + Na2S = PbS + 2 NaNќ3;
в) NaCIO + HNќ3 = NaNќ3 +HC1ќ;
г) 2—ќз + Ќ2S04 = 2Sќ4 + —ќ2 + Ќ2ќ;
д) —Ќ3—ќќЌ + NaOH = CH3COONa + Ќ2ќ.
ќтметим, что взаимодействие этих веществ возможно, ибо в результате происходит св€зывание ионов с образованием слабых электролитов (Ќ2ќ, HC10), осадка (PbS), газа (—ќ2).
¬ реакции (д) два слабых электролита, но так как реакции идут в сторону большего св€зывани€ ионов и вода Ч более слабый электролит, чем уксусна€ кислота, то равновесие реакции смещено в сторону образовани€ воды. »сключив одинаковые ионы из обеих частей равенства:
а) Na+ и —l-; б) Na+ и NO 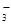 ; в) Na+ и NO
; в) Na+ и NO 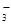 3; г) + и SO
3; г) + и SO 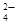 ; д) Na+,
; д) Na+,
получим ионно-молекул€рные уравнени€ этих реакций:
а) H+ + ќЌ  = Ќ2ќ;
= Ќ2ќ;
в) Pb2+ + S 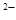 = PbS;
= PbS;
б) —1ќ  + H+ = HC1ќ;
+ H+ = HC1ќ;
г) —ќ 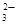 +2 H+ = Cќ2+H2ќ;
+2 H+ = Cќ2+H2ќ;
д) —Ќз—ќќЌ + ќЌ  = —Ќз—ќќ
= —Ќз—ќќ  + Ќ2ќ.
+ Ќ2ќ.
ѕример 2. —оставьте молекул€рные уравнени€ реакций, которым соответствуют следующие ионно-молекул€рные уравнени€:
а) SO 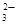 +2 H+ = S02 + H2ќ;
+2 H+ = S02 + H2ќ;
б) Pb2+ + Crќ 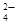 = PbCr04;
= PbCr04;
в) Ќ—ќ 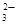 + ќЌ
+ ќЌ  = —ќ
= —ќ 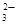 + Ќ2ќ;
+ Ќ2ќ;
г) ZnOH+ + H+ = Zn2+ + H2O.
–ешение. ¬ левой части данных ионно-молекул€рных уравнений указаны свободные ионы, которые образуютс€ при диссоциации растворимых сильных электролитов. Cледовательно, при составлении молекул€рных уравнений следует исходить из соответствующих растворимых сильных электролитов. Ќапример:
а) Na2Sќ3 + 2 Ќ—1 = 2 NaCl + SO2 + Ќ2ќ;
б) Pb(NO3)2 + K2CrO4 = PbCrO4 + 2 KNO3;
в) Ќ—ќ3 + ќЌ = 2—ќ3 + H2O;
г) ZnOHCl + Ќ—1 = ZnCl2 + H2O.
ќЌ“–ќЋ№Ќџ≈ ¬ќѕ–ќ—џ
161. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: а) NаЌ—ќз и NaOH;
б) K2SiO3 и Ќ—1; в) BaCl2 и Na2SO4.
162. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) K2S и Ќ—1;
б) FeSќ4 и (NH4)2S; в) —г(ќЌ)3 и ќЌ.
163. —оставьте по три молекул€рных уравнени€ реакций, которые выражаютс€ ионно-молекул€рными уравнени€ми:
а) Mg2+ + CO 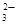 = MgCќ3; б) H+ + ќЌ
= MgCќ3; б) H+ + ќЌ  = H2O.
= H2O.
164. акие из веществ - ј1(ќЌ)3; H2Sќ4; ¬а(ќЌ)2 - взаимодействуют с гидроксидом кали€? ¬ыразите эти реакции молекул€рными и ионно-молекул€рными уравнени€ми.
|
|
|
165. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: а) Ќ—ќ3 и H2Sќ4;
б) Zn(OH)2 и NaOH; в) —а—l2 и AgNќ3.
166. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) CuSќ4 и H2S;
б) ¬а—ќ3 и HNO3; в) Fе—13 и ќЌ.
167. —оставьте по три молекул€рных уравнени€ реакций, которые выражаютс€ следующими ионно-молекул€рными уравнени€ми:
а) —и2+ + S 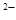 = CuS;
= CuS;
6) SiO 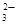 + 2 H+ = H2SiO3.
+ 2 H+ = H2SiO3.
168. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) Sn(OH)2 и Ќ—1;
б) BeSќ4 и ќЌ; в) NH4Cl и ¬а(ќЌ)2.
169. акие из веществ Ч Ќ—ќ3, —Ќ3—ќќЌ, NiS04, Na2S Ч взаимодействуют с раствором серной кислоты? Ќапишите молекул€рные и ионно-молекул€рные уравнени€ этих реакций.
170. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: а) AgNќ3 и 2—rO4;
б) Pb(Nќ3)2 и KI; в) CdSќ4 и Na2S.
171. —оставьте молекул€рные уравнени€ реакций, которые выражаютс€ ионно-молекул€рными уравнени€ми:
а) —а—ќ3 + 2 Ќ+ = —а2+ + H2O + CO2;
б) ј1(ќЌ)3 + ќЌ  = ј1O
= ј1O 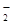 + 2 Ќ2ќ;
+ 2 Ќ2ќ;
в) –№2+ + 2 I  = PbI2
= PbI2
172. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: а) ¬е(ќЌ)2 и NaOH; б) —и(ќЌ)2; и HNќ3; в) ZnOHNќ3 и HNќ3.
173. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) Na3Pќ4 и CaCl2;
б) 2—ќ3 и ¬а—l2; в) Zn(OH)2 и ќЌ.
174. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций, которые выражаютс€ ионно-молекул€рными уравнени€ми:
Fе(ќЌ)3 + « H+ = Fe3+ + « Ќ2ќ;
Cd2+ + 2 OЌ  = Cd(OH)2;
= Cd(OH)2;
H+ + NO 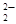 = HNO2.
= HNO2.
175. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) CdS и Ќ—1;
б) —г(ќЌ)3 и NaOH; в) Ba(OH)2 и CoCl2.
176. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций, которые выражаютс€ ионно-молекул€рными уравнени€ми:
а) Zn2+ + H2S = ZnS + 2 H+;
б) Ќ—ќ 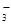 + Ќ+ = H2O + —ќ2;
+ Ќ+ = H2O + —ќ2;
в) Ag+ + —1  = AgCl.
= AgCl.
177. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: a) H2S04 и ¬а(ќЌ)2;
б) Fе—13 и NH40H; в) CH3COONa и Ќ—1.
178. —оставьте молекул€рные и ионно-молекул€рные уравнени€ реакций взаимодействи€ в растворах между: а) Fе—13 и ќЌ;
6) NiSO4 и (NH4)2S; в) MgCO3 и HNO3.
179. —оставьте молекул€рные уравнени€ реакций, которые выражаютс€ ионно-молекул€рными уравнени€ми:
а) ¬е(ќЌ)2 + 2 OЌ  = ¬еќ
= ¬еќ 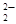 + 2 Ќ2O
+ 2 Ќ2O
б) —Ќз—ќќ  + Ќ+ = —Ќз—ќќЌ
+ Ќ+ = —Ќз—ќќЌ
в)Ba2+ + SO 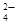 = BaSO4
= BaSO4
180. акие из веществ Ч NaCl, NiS04, ¬е(ќЌ)2, Ќ—ќз Ч взаимодействуют с раствором гидроксида натри€. «апишите молекул€рные и ионно-молекул€рные уравнени€ этих реакций.
10. »ќЌЌќ≈ –ј¬Ќќ¬≈—»≈ ¬ќƒџ.
¬ќƒќ–ќƒЌџ… ѕќ ј«ј“≈Ћ№ рЌ
¬ода €вл€етс€ очень слабым электролитом и лишь в незначительной степени диссоциирует на ионы водорода и гидроксила:
 Ќ2ќ Ќ+ + ќЌ
Ќ2ќ Ќ+ + ќЌ 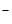 .
.
Ётому процессу соответствует константа диссоциации:
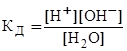 . (1)
. (1)
онстанта диссоциации воды очень мала ( ƒ.(Ќ 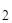 ќ) = 1,86ּ10
ќ) = 1,86ּ10 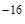 ), поэтому равновесна€ концентраци€ недиссоциированных молекул воды практически равна общей концентрации воды:
), поэтому равновесна€ концентраци€ недиссоциированных молекул воды практически равна общей концентрации воды:
[H2O] = —ћ = 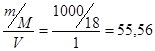 моль/л.
моль/л.
¬ разбавленных водных растворах концентраци€ воды мен€етс€ незначительно, так что ее можно считать посто€нной. “огда из формулы (1) получим
|
|
|
[H+]ּ[OH 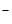 ] = ƒ.ּ[H2O] = 1,86ּ10
] = ƒ.ּ[H2O] = 1,86ּ10 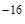 ּ55,56 = 10
ּ55,56 = 10 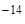 .
.
“аким образом, произведение концентраций ионов водорода и гидроксила представл€ет собой посто€нную величину (при данной температуре) и называетс€ ионным произведением воды Ќ 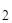 O. ѕри стандартной температуре “ = 298 (250—)
O. ѕри стандартной температуре “ = 298 (250—)
Ќ 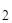 O = [H+]ּ[OH
O = [H+]ּ[OH 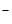 ] = 10
] = 10 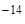 . (2)
. (2)
 |

 ¬ нейтральных растворах [H+] =ּ[OH
¬ нейтральных растворах [H+] =ּ[OH 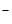 ] = Ö 10
] = Ö 10 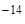 = 10
= 10 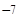 моль/л.
моль/л.
ƒл€ характеристики кислотности (щелочности) среды используетс€ водородный показатель рЌ Ц дес€тичный логарифм концентрации ионов водорода, вз€тый с обратным знаком:
рЌ = - lg [H+].
¬ нейтральных растворах рЌ = 7. ¬ кислых растворах [H+] > 10 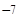 , следовательно, рЌ < 7; в щелочных растворах [H+] < 10
, следовательно, рЌ < 7; в щелочных растворах [H+] < 10 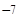 , поэтому рЌ > 7.
, поэтому рЌ > 7.
ѕо аналогии с водородным показателем рЌ введен показатель рќЌ.
рќЌ = - lg [OH 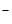 ].
].
Ћогарифмиру€ соотношение (2), получим рЌ + рќЌ = 14.
ѕример 1. ќпределить концентрацию ионов водорода в растворе, если рЌ = 4,60.
–ешение. ѕо условию задачи рЌ = 4,60 = -lg [H+].
ќтсюда [H+] = 10-4,60 = 2,5.10-5 моль/л.
ѕример 2. ќпределть концентрацию ионов [OH 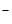 ] в растворе, если рЌ = 10,80.
] в растворе, если рЌ = 10,80.
–ешение. »з соотношени€ рЌ + рќЌ = 14 находим
ќЌ = 14 Ц 10,80 = 3,20.
ќтсюда Цlg [OH 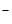 ] = 3,20. —ледовательно [OH
] = 3,20. —ледовательно [OH 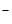 ] = 10-3,20 = 6,31.10-4.
] = 10-3,20 = 6,31.10-4.
ѕример 3. ¬ычислить рЌ 0,01 ћ раствора гидроксида натри€.
–ешение. √идроксид натри€ €вл€етс€ сильным основанием, диссоциирует на ионы практически полностью
NaOH = Na+ + OH  .
.
»з уравнени€ диссоциации следует, что из одного мол€ гидроксида натри€ образуетс€ один моль ионов OH 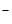 ,следовательно в 0,01 ћ растворе [OH
,следовательно в 0,01 ћ растворе [OH 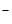 ] = 0,01. Ќайдем рќЌ = - lg [OH
] = 0,01. Ќайдем рќЌ = - lg [OH 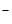 ] = -lg 0,01 = 2.
] = -lg 0,01 = 2.
ќтсюда рЌ = 14 Ц рќЌ = 12.
ќЌ“–ќЋ№Ќџ≈ ¬ќѕ–ќ—џ
181. ќпределите рЌ раствора, в 0,5 л которого содержитс€ 0,05 г NaOH.
ќтвет: 11;40.
182. ¬о сколько раз концентраци€ ионов водорода в крови (рЌ = 7,36) меньше, чем в желудочном соке (рЌ = 1,00)?
ќтвет: ¬ 2,3∙10 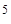 раз.
раз.
183. ќпределите [H+] и [OH 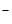 ] в растворе, рЌ которого равен 6,2.
] в растворе, рЌ которого равен 6,2.
ќтвет: 6,3∙10 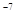 моль/л; 1,6∙10
моль/л; 1,6∙10 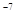 моль/л.
моль/л.
184. –ассчитайте рЌ раствора, полученного смешением 25 мл 0,5 ћ раствора HCl, 10мл 0,5 ћ раствора NaOH и 15 мл воды.
ќтвет: 0,82.
185. ак изменитс€ рЌ 0,2 ћ раствора HCl, если его вдвое разбавить водой?
ќтвет: ”величитс€ на 0,3.
186. ¬ычислите рЌ 0,5%-ного раствора азотной кислоты.
ќтвет: 1,10.
187. 500 мл воды прибавили 20 мл 0,1 н раствора Ќ—1. Ќайдите рЌ полученного раствора.
ќтвет: 2,4.
188. ¬ычислите рЌ 1,5%-ного раствора ќЌ.
ќтвет: 13,43.
189. 25 мл 10%-ного раствора HCl (плотность 1,05) разбавили водой до 500 мл. ¬ычислите рЌ полученного раствора.
ќтвет: 0,84.
190. 250 мл нейтрального раствора прибавили 50 мл 0,5 н раствора ќЌ. ¬ычислите рЌ полученного раствора.
ќтвет: 12,9.
191. 10,0 мл 20%-ного раствора ќЌ (плотность 1,18) разбавили водой до 250 мл. ¬ычислите рЌ полученного раствора.
ќтвет: 13,23.
192. ¬ мерную колбу на 250 мл налили 10,0 мл 24%-ного раствора HCl (плотность 1,12) и довели раствор водой до метки. »з полученного раствора 5,0 мл перенесли в мерную колбу на 100 мл и разбавили водой до метки. Ќайдите рЌ полученного раствора.
ќтвет: 1,83.
193. 25 мл 0,2 н раствора HCl прибавили 25 мл 0,1 н раствора NaOH.¬ычислите рЌ полученного раствора.
ќтвет: 1,3.
194. 20,0 мл 12%-ного раствора NaOH (плотность 1,14) разбавили водой до 500 мл. 50,0 мл полученного раствора перенесли в другую колбу и разбавили водой до 1000 мл. Ќайдите рЌ последнего раствора.
ќтвет: 11,85.
195. 100 мл 0,1 н раствора HNO3 прибавили 2 мл 6%-ного раствора NaOH (плотность 1,0). Ќайдите рЌ полученного раствора.
ќтвет: 1,2
196. Ќайдите рЌ раствора, в 100 мл которого содержитс€ 0,12 мг NaOH.
ќтвет: 9,48.
197. 100 мл 0,1 н раствора NaOH прибавили 5 мл 4%-ного раствора HCl (плотность 1,0). Ќайдите рЌ исходного и полученного раствора.
ќтвет: 13; 12,6.
|
|
|
198. 100 мл 0,2 н раствора HCl прибавили 5 мл 3%-ного раствора ќЌ. Ќайдите рЌ исходного и полученного раствора.
ќтвет: 0,7; 0,78.
199. акую массу NaOH следует растворить в 400 мл воды, чтобы получить раствор, рЌ которого равен 12?
200. ¬ычислите мол€рную концентрацию раствора гидроксида бари€, если известно, что рЌ данного раствора равен 11.
√»ƒ–ќЋ»« —ќЋ≈…
√идролизом называетс€ обменное взаимодействие ионов растворенной соли с водой, сопровождающеес€ обычно изменением рЌ среды. ѕричина гидролиза заключаетс€ в св€зывании ионов соли ионами воды в слабодиссоциирующие продукты (молекулы слабых кислот или оснований, анионы кислых или катионы основных солей).
ѕример 1. —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза солей: а) KCN; б) Na2CO3; в) ZnSO4. ќпределите реакцию среды растворов этих солей.
–ешение. а) ÷ианид кали€ KCN Ч соль слабой кислоты HCN и сильного основани€KOH. ѕри растворении в воде молекулы KCN полностью диссоциируют на катионы + и анионы CN  . атионы + не могут св€зывать ионы ќЌ
. атионы + не могут св€зывать ионы ќЌ  воды, так как ќЌ Ц сильный электролит. јнионы же CN
воды, так как ќЌ Ц сильный электролит. јнионы же CN  св€зывают ионы Ќ+ воды, образу€ молекулы слабого электролита HCN. —оль гидролизуетс€ по аниону. »онно-молекул€рное уравнение гидролиза
св€зывают ионы Ќ+ воды, образу€ молекулы слабого электролита HCN. —оль гидролизуетс€ по аниону. »онно-молекул€рное уравнение гидролиза
 CN
CN  + Ќ2ќ HCN + ќЌ
+ Ќ2ќ HCN + ќЌ 
или в молекул€рной форме
 KCN + H2O HCN + KOH.
KCN + H2O HCN + KOH.
¬ результате гидролиза в растворе по€вл€етс€ некоторый избыток ионов ќЌ  , поэтому раствор KCN имеет щелочную реакцию (рЌ > 7).
, поэтому раствор KCN имеет щелочную реакцию (рЌ > 7).
б) арбонат натри€ Na2CO3 Ч соль слабой многоосновной кислоты и сильного основани€. ¬ этом случае анионы соли —ќ 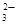 , св€зыва€ ионы воды Ќ+, образуют анионы кислой соли Ќ—ќ
, св€зыва€ ионы воды Ќ+, образуют анионы кислой соли Ќ—ќ 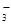 , а не молекулы Ќ2—ќ3, так как ионы Ќ—ќ
, а не молекулы Ќ2—ќ3, так как ионы Ќ—ќ 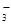 диссоциируют гораздо труднее, чем молекулы Ќ2—ќ3. ¬ обычных услови€х гидроли идет по первой ступени. —оль гидролизуетс€ по аниону. »онно-молекул€рное уравнение гидролиза
диссоциируют гораздо труднее, чем молекулы Ќ2—ќ3. ¬ обычных услови€х гидроли идет по первой ступени. —оль гидролизуетс€ по аниону. »онно-молекул€рное уравнение гидролиза
 —ќ
—ќ 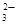 + Ќ2ќ Ќ—ќ
+ Ќ2ќ Ќ—ќ 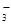 + ќЌ
+ ќЌ 
или в молекул€рной форме
 Na2CO3 + H2O NaHCO3 + NaOH.
Na2CO3 + H2O NaHCO3 + NaOH.
¬ растворе по€вл€етс€ избыток ионов ќЌ  , поэтому раствор Na2CO3 имеет щелочную реакцию (рЌ>7).
, поэтому раствор Na2CO3 имеет щелочную реакцию (рЌ>7).
в) —ульфат цинка ZnSO4 Ч соль слабого многокислотного основани€ Zn(OH)2 и сильной кислоты H2SO4. ¬ этом случае катионы Zn2+ св€зывают гидроксильные ионы воды, образу€ катионы основной соли ZnOH+. ќбразование молекул Zn(OH)2 не происходит, так как ионы ZnOH+ диссоциируют гораздо труднее, чем молекулы Zn(OH)2. ¬ обычных услови€х гидролиз идет по первой ступени. —оль гидролизуетс€ по катиону. »онно-молекул€рное уравнение гидролиза
 Zn2+ + H2O ZnOH+ + H+
Zn2+ + H2O ZnOH+ + H+
или в молекул€рной форме
 2 ZnSO4 + 2H2O (ZnOH)2SO4 + H2SO4.
2 ZnSO4 + 2H2O (ZnOH)2SO4 + H2SO4.
¬ растворе по€вл€етс€ избыток ионов водорода, поэтому раствор ZnSO4 имеет кислую реакцию (рЌ < 7).
ѕример 2. акие продукты образуютс€ при смешивании растворов ј1(Nќ3)3 и 2—ќ3? —оставьте ионно-молекул€рное и молекул€рное уравнение реакции.
–ешение. —оль ј1(Nќ3)3 гидролизуетс€ по катиону, а 2—ќ3 Ч по аниону:
 Al3+ + Ќ2ќ јlOЌ2+ + Ќ+;
Al3+ + Ќ2ќ јlOЌ2+ + Ќ+;
 CO
CO 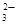 +Ќ20 Ќ—ќ
+Ќ20 Ќ—ќ 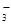 + ќЌ
+ ќЌ  .
.
≈сли растворы этих солей наход€тс€ в одном сосуде, то идет взаимное усиление гидролиза каждой из них, так как ионы H+ и ќЌ  образуют молекулу слабого электролита Ќ20. ѕри этом гидролитическое равновесие сдвигаетс€ вправо и гидролиз каждой из вз€тых солей идет до конца с образованием ј1(ќЌ)3 и CO2 (Ќ2—ќз).
образуют молекулу слабого электролита Ќ20. ѕри этом гидролитическое равновесие сдвигаетс€ вправо и гидролиз каждой из вз€тых солей идет до конца с образованием ј1(ќЌ)3 и CO2 (Ќ2—ќз).
»онно-молекул€рное уравнение
2 ј13+ + 3 CO 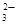 + « Ќ2ќ = 2 ј1(ќЌ)3 + « —ќ2
+ « Ќ2ќ = 2 ј1(ќЌ)3 + « —ќ2
молекул€рное уравнение
2 A1(N03)3 + « 2—ќ3 + « Ќ2ќ = 2 ј1(ќЌ)3 + « —ќ2 + 6 KN03.
ќЌ“–ќЋ№Ќџ≈ ¬ќѕ–ќ—џ
201. —оставьте ионно-молекул€рное и молекул€рное уравнени€ гидролиза, происход€щего при смешивании растворов K2S и —г—13. ажда€ из вз€тых солей гидролизуетс€ необратимо до конца с образованием соответствующих основани€ и кислоты.
202. раствору Fе—13 добавили следующие вещества: а) Ќ—1; б) ќЌ; в) ZnCl2;r) Na2CO3. ¬ каких случа€х гидролиз хлорида железа (III) усилитс€? ѕочему? —оставьте ионно-молекул€рные уравнени€ гидролиза соответствующих солей.
|
|
|
203. акие из солей Ч јl2(SO4)3, K2S, Pb(NO3)2, KC1 Ч подвергаютс€ гидролизу? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза соответствующих солей. акое значение (7 < рЌ < 7) имеют растворы этих солей?
204. ѕри смешивании Fе—13 и Nа2—ќ3 кажда€ из вз€тых солей гидролизуетс€ необратимо до конца с образованием соответствующих основани€ и кислоты. ¬ыразите этот совместный гидролиз ионно-молекул€рным и молекул€рным уравнени€ми.
205. раствору Na2CO3 добавили следующие вещества: а) Ќ—1;
б) NaOH; в) Cu(Nќ3)2; г) K2S. ¬ каких случа€х гидролиз карбоната натри€ усилитс€? ѕочему? —оставьте ионно-молекул€рные уравнени€ гидролиза соответствующих солей.
206. акое значение рЌ (7< рЌ < 7) имеют растворы солей Na2S, ј1—l3, NiSO4? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза этих солей.
207. —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза солей –b(Nќ3)2, Na2CO3, Fe2(SO4)3. акое значение рЌ
(7 < рЌ < 7) имеют растворы этих солей?
208. —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза солей —Ќ3—ќќ , ZnSO4, ј1(Nќ3)3. акое значение рЌ
(7 < рЌ < 7) имеют растворы этих солей?
209. акое значение рЌ (7< рЌ < 7) имеют растворы солей 3–O4, K2S, CuSO4? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза этих солей.
210. —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза солей CuCl2, —s2—ќ3, —г(Nќ3)3. акое значение рЌ (7 < рЌ < 7) имеют растворы этих солей?
211. акие из солей Ч RbCl, —г2(SO4)3, Ni(NO3)2, Na2SO3 Ч подвергаютс€ гидролизу? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза соответствующих солей. акое значение рЌ
(7< рЌ < 7) имеют растворы этих солей?
212. раствору Al2(SO4)3 добавили следующие вещества: а) H2SO4; б) ќЌ; в) Na2SO3; г) ZnSO4. ¬ каких случа€х гидролиз сульфата алюмини€ усилитс€? ѕочему? —оставьте ионно-молекул€рные уравнени€ гидролиза соответствующих солей.
213. ака€ из двух солей при равных услови€х в большей степени подвергаетс€ гидролизу: Na2CO3 или Nа2SO3; Fе—13 или FeCl2? ѕочему? —оставьте ионно-молекул€рные уравнени€ гидролиза этих солей.
214. ѕри смешивании растворов ј12(SO4)3 и Na2CO3 кажда€ из вз€тых солей гидролизуетс€ необратимо до конца с образованием соответствующих основани€ и кислоты. —оставьте ионно-молекул€рные и молекул€рное уравнени€ происход€щего совместного гидролиза.
215. акие из солей Ч NaBr, Na2S, 2—ќ3, CoCl2 Ч подвергаютс€ гидролизу? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза соответствующих солей. ќпределите рЌ (7 < рЌ < 7).
216. ака€ из двух солей при равных услови€х в большей степени подвергаетс€ гидролизу: NaCN или NaCIO; MgCl2 или ZnCl2? ѕочему? —оставьте ионно-молекул€рные и молекул€рные уравнени€ гидролиза соответствующих солей.






