Ёлектролитическа€ диссоциаци€ Ц это распад вещества в растворе на сольватированные (в случае водной среды на гидратированные) ионы под действием молекул растворител€.
—тепень диссоциации a есть отношение числа молекул, подвергшихс€ в растворе распаду на ионы, к общему числу растворенных молекул. ѕо степени диссоциации в растворах электролиты подраздел€ютс€ на сильные и слабые.
ƒл€ сильных электролитов a =1 (диссоциаци€ полна€) и не зависит от концентрации раствора. ним относ€тс€ водные растворы подавл€ющего числа солей, гидроксидов щелочных и щелочноземельных металлов, р€да кислот (HCl, HBr, HI, HClO4, HNO3, H2SO4, H2SeO4 и другие).
ƒл€ слабых электролитов a <1 и уменьшаетс€ с ростом концентрации раствора. слабым электролитам относ€тс€ водные растворы аммиака, некоторых органических азотсодержащих соединений (гидразин NH2NH2, гидроксиламин NH2OH, и др.), таких кислот, как HNO2, H3BO3, H2CO3, CH3COOH, H2SO3, HOCl, HCN, водный раствор H2S и другие).
¬ растворах слабых электролитов устанавливаетс€ диссоционное равновесие, характеризующеес€ константой диссоциации. ћногоосновные слабые кислоты и многокислотные слабые основани€ диссоциируют ступенчато, в их растворах устанавливаетс€ сложное равновесие между недиссоциированными молекулами и ионами различного знака.
—в€зь между константой диссоциации слабого электролита и его степенью диссоциации выражаетс€ законом разбавлени€ ќствальда.
ѕусть электролит диссоциирует на два иона Ц положительный катион и отрицательный анион:
ј ⇆ + + јЦ.
ƒл€ такого электролита закон разбавлени€ ќствальда записываетс€ в виде
д=  ,(2.5.1)
,(2.5.1)
где д Ц константа диссоциации электролита;
— Ц мол€рна€ концентраци€;
α Ц степень диссоциации.
≈сли дл€ слабого электролита выполн€етс€ условие α <<1, то справедливо приближенное равенство
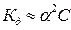 (2.5.2)
(2.5.2)
ѕоскольку растворы электролитов значительно отличаютс€ по свойствам от идеальных растворов, то при расчетах следует пользоватьс€ не концентраци€ми, а активност€ми ионов, в соответствии с которыми ионы про€вл€ют свое действие. јктивность а определ€етс€ выражением:
a=f × C,
где f Ц коэффициент активности ионов, завис€щий от концентрации и состава раствора, зар€да, природы иона, температуры и других факторов;
— Ц концентраци€ ионов.
јктивности ионов св€заны с их мол€рными концентраци€ми аналогичными соотношени€ми:
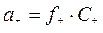
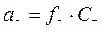 , (2.5.3)
, (2.5.3)
где f Ц мол€рный коэффициент активности, формально учитывающий все отклонени€ реальных растворов от идеальных.
—уществует р€д экспериментальных и теоретических способов определени€ коэффициентов активности.
¬ разбавленных растворах (— не превышает 0,5 моль/л) можно приближенно считать, что коэффициент активности зависит только от зар€да иона и ионной силы раствора, котора€ вычисл€етс€ по формуле:
 , (2.5.4)
, (2.5.4)
где —i Ц концентрации ионов;
zi Ц зар€ды всех наход€щихс€ в растворе ионов.
¬ последней формуле под —i строго следует понимать мол€льность ионов, однако в разбавленных растворах, дл€ которых справедлива эта формула, мол€льность по величине мало отличаетс€ от мол€рности.
|
|
|
¬ разбавленных растворах коэффициент активности f мало зависит от природы растворенных веществ. ƒл€ таких растворов при 298 средний коэффициент активности ионов электролита можно рассчитать по формуле ƒеба€-√юккел€ в первом приближении:
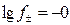 ,
,  . (2.5.5)
. (2.5.5)
ƒл€ приближенного нахождени€ коэффициентов активности также можно воспользоватьс€ справочными таблицами. ¬ таблице 2.5.1 приведена зависимость коэффициентов активности ионов Ќ+ и ќЌ - от ионной силы раствора.
“аблица 2.5.1 Ц «ависимость активности ионов от ионной силы раствора
| »оны | оэффициенты активности при заданной ионной силе раствора | |||||||
| 0,001 | 0,002 | 0,005 | 0,01 | 0,02 | 0,05 | 0,1 | 0,2 | |
| Ќ+ | 0,975 | 0,967 | 0,950 | 0,923 | 0,914 | 0,880 | 0,860 | 0,830 |
| ќЌ - | 0,975 | 0,964 | 0,946 | 0,926 | 0,900 | 0,855 | 0,810 | 0,760 |
¬ сильно разбавленных растворах коэффициент активности приближаетс€ к единице, поэтому дл€ не очень точных расчетов различием между активностью и концентрацией можно пренебречь.
ислотность или щелочность раствора прин€то характеризовать водородным показателем среды рЌ.
¬одородный показатель среды рЌ водного раствора равен вз€тому с обратным знаком дес€тичному логарифму активности ионов водорода в этом растворе:
 (2.5.6)
(2.5.6)
—реда водного раствора электролита может быть кислой, нейтральной или щелочной. Ќосител€ми кислотных свойств €вл€ютс€ ионы Ќ+, за основные свойства водных растворов отвечают ионы ќЌ -.
„иста€ вода €вл€етс€ слабым электролитом. ≈е электролитическа€ диссоциаци€ протекает в соответствии с уравнением:
Ќ2ќ D Ќ+ + ќЌ -,
при этом образуетс€ равное количество катионов водорода и гидроксид-анионов.
онстанта диссоциации воды выражаетс€ следующим образом:
 . (2.5.7)
. (2.5.7)
ѕри 220— она равна 1,8Ј10-16. јктивность чистой воды при той же температуре равна примерно 55,56 моль/л. “огда из выражени€ (2.5.7) следует:
 (2.5.8)
(2.5.8)
¬еличина  носит название ионного произведени€ воды. ѕри 220— W=10-14.
носит название ионного произведени€ воды. ѕри 220— W=10-14.
¬ чистой воде и в нейтральных растворах 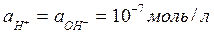 (рЌ=7).
(рЌ=7).
¬ кислых растворах 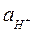 >10-7 моль/л (рЌ<7).
>10-7 моль/л (рЌ<7).
¬ щелочных растворах 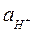 <10-7 моль/л (рЌ>7).
<10-7 моль/л (рЌ>7).
Ќар€ду с водородным показателем рЌ в р€де случаев используют показатель рќЌ, равный вз€тому с обратным знаком дес€тичному логарифму активности гидроксид-ионов.
 (2.5.9)
(2.5.9)
ѕутем логарифмировани€ и умножени€ на -1 выражени€ (2.5.8) можно прийти к следующему соотношению:
рЌ+рќЌ= 14 (2.5.10)
ѕри проведении расчетов рЌ нужно учитывать, что сильные кислоты и основани€ диссоциируют практически полностью, степень диссоциации α =1. ƒл€ слабых кислот и оснований диссоциаци€ частична€. ƒл€ них выполн€етс€ закон разбавлени€ ќствальда.
—уществует больша€ группа малорастворимых солей, гидроксидов, кислот, которые не могут, строго говор€, быть отнесены ни к сильным, ни к слабым электролитам. »х нельз€ считать слабыми электролитами, так как водные растворы из-за малой растворимости веществ очень разбавлены, и вещества в них подвергаютс€ полному распаду на ионы. Ќо и к сильным электролитам указанные соединени€ также нельз€ отнести, поскольку их растворы содержат ничтожно малое количество ионов. “акие вещества прин€то называть малорастворимыми электролитами.
¬ насыщенном растворе малорастворимого электролита устанавливаетс€ гетерогенное равновесие между осадком и наход€щимис€ в растворе ионами:
|
|
|
јmBn 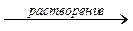 mAn++nBm-.
mAn++nBm-.
осадок  насыщенный раствор
насыщенный раствор
¬ насыщенном растворе скорости процессов растворени€ и кристаллизации одинаковы, а концентрации ионов над твердой фазой €вл€ютс€ равновесными при данной температуре.
онстанта равновеси€ данного гетерогенного процесса определ€етс€ только произведением активностей ионов в растворе и не зависит от активности твердого компонента. ќна получила название произведение растворимости ѕ–.

 (2.5.11)
(2.5.11)
“аким образом, произведение активностей ионов в насыщенном растворе малорастворимого электролита при заданной температуре есть величина посто€нна€.
≈сли электролит имеет очень низкую растворимость, то в его растворе концентрации ионов ничтожны. ¬ этом случае межионным взаимодействием можно пренебречь и считать концентрации ионов равными их активност€м. “огда произведение растворимости можно выразить через равновесные мол€рные концентрации ионов электролита:
 . (2.5.12)
. (2.5.12)
ѕроизведение растворимости, как люба€ константа равновеси€, зависит от природы электролита и от температуры, но не зависит от концентрации ионов в растворе.
ѕри увеличении концентрации одного из ионов в насыщенном растворе малорастворимого электролита, например, в результате введени€ другого электролита, содержащего тот же ион, произведение концентраций ионов становитс€ больше величины произведени€ растворимости. ѕри этом равновесие между твердой фазой и раствором смещаетс€ в сторону образовани€ осадка. ќсадок будет образовыватьс€ до тех пор, пока не установитс€ новое равновесие, при котором снова выполн€етс€ условие (2.5.12), но уже при других соотношени€х концентраций ионов. ѕри увеличении концентрации одного из ионов в насыщенном растворе над твердой фазой концентраци€ другого иона уменьшаетс€ так, чтобы произведение растворимости осталось величиной посто€нной при неизменных услови€х.
»так, условием выпадени€ осадка €вл€етс€:
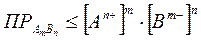 . (2.5.13)
. (2.5.13)
≈сли в насыщенном растворе малорастворимого электролита уменьшить концентрацию какого-либо его иона, то ѕ– станет больше произведени€ концентраций ионов. –авновесие сместитс€ в сторону растворени€ осадка. –астворение будет продолжатьс€ до тех пор, пока снова не станет выполн€тьс€ условие (2.5.12).
»так, условием растворени€ осадка €вл€етс€:
 . (2.5.14)
. (2.5.14)
ѕример 1
¬ычислите рЌ 0,01 ћ раствора HNO3.
–ешение
јзотна€ кислота €вл€етс€ сильным электролитом, диссоциирует полностью. онцентраци€ катионов водорода будет равна концентрации кислоты. јктивность ионов водорода €вл€етс€ функцией концентрации ионов водорода.

оэффициенты активности ионов дл€ растворов с различной ионной силой приведены в таблице 2.5.1.
»онную силу раствора можно рассчитать по уравнению:

»з таблицы 2.5.1 дл€ I =0,01 находим значение коэффициента активности fH+ =0.923.
ƒалее рассчитываем активность иона водорода и рЌ раствора:
ан+ = 0,92Ј0,01 = 9,2Ј10-3моль/л;
рЌ = -lg9,2Ј10-3 = 2,04.
ѕримечание
¬ случае расчета рЌ раствора щелочи целесообразно сначала рассчитать гидроксильный показатель среды рќЌ, а затем найти водородный показатель среды по разности рЌ = 14 - рќЌ.
ѕример 2
¬ычислите рЌ 0,01 ћ раствора аммиака.
–ешение
ѕри расчете водородного показател€ среды водных растворов слабых кислот и оснований следует учитывать обратимость процесса их электролитической диссоциации (α <1).
NH3ЈH2O ⇆ NH4+ +OHЦ
онцентраци€ ионов H+ в растворах слабых кислот и концентраци€ ионов ќЌЦ в растворах слабых оснований численно равны концентрации продиссоциировавших молекул электролита, а не исходной концентрации раствора, как в случае сильных кислот и оснований.
онцентрацию продиссоциировавших на ионы молекул электролита определ€ют, исход€ из степени диссоциации:
—=α—о,
где —о - исходна€ концентраци€ молекул, моль/л.
—тепень диссоциации рассчитывают на основании закона разбавлени€ ќствальда, при этом константы диссоциации слабых электролитов берут из справочной литературы.
|
|
|
¬ случа€х, когда расчет по упрощенному уравнению (2.5.2) дает результат α> 0,1, вычислени€ повтор€ют по более точной формуле (2.5.1).
ѕри вычислении водородного или гидроксильного показател€ среды раствора слабой кислоты или слабого основани€ можно приближенно считать, что вследствие малых ионных сил растворов, активности ионов равны их мол€рным концентраци€м.
—тепень диссоциации NH3ЈЌ2ќ в 0,01 ћ растворе равна:
 =
= 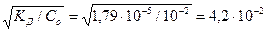

Ќаходим концентрацию молекул NH3ЈЌ2ќ, распавшихс€ на ионы, и равную ей концентрацию ионов ќЌ -:
—=αЈ—о = 4,2 10-2 Ј10-2 = 4,2Ј10-4 моль/л,
CќЌ- = = 4,2Ј10-4 моль/л.
ќпредел€ем рќЌ и рЌ 0,01 ћ раствора аммиака:
рќЌ = -lg —ќЌ- = -lg 4,2Ј10-4 = 3,33
рЌ = 14 - 3,33 = 10,67.
ѕример 3
онстанта диссоциации HCN равна 6,2·10Ц10. ¬ычислите массу цианид-анионов, содержащихс€ в 0,1 л раствора кислоты с концентрацией 0,1 моль/л.
–ешение
—тепень диссоциации HCN можно вычислить на основании закона разбавлени€ ќствальда. ћожно считать, что если отношение константы диссоциации слабого электролита к его концентрации больше 100, то можно пользоватьс€ упрощенной формулой
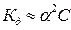 .
.



 .
.
Ќайдем массу цианид-анионов:

ѕример 4
акова ионна€ сила 0,01 н. раствора Na2SO4 с учетом полной диссоциации соли?
–ешение
Ёквивалентом сульфата натри€ €вл€етс€ условна€ частица 1/2 Na2SO4. ѕоэтому мол€рна€ концентраци€ раствора сульфата натри€ будет в два раза меньше его нормальной концентрации, то есть равна 0,005 моль/л.
“ак как одна частица Na2SO4 распадаетс€ на два катиона натри€ и один сульфат-анион, то
C (Na +)=2 C (Na2SO4)=0,01 моль/л;
C (SO42- -)= C (Na2SO4)=0,005 моль/л.
–ассчитаем ионную силу раствора.

ќтвет: ионна€ сила раствора равна 0,015.
ѕример 5
акова растворимость Ag2CrO4, выраженна€ в г/л, если величина произведени€ растворимости дл€ этой соли составл€ет 1,3∙10-12?
–ешение
Ag2CrO4 ⇆ 2Ag+ + CrO42Ц
ѕ–= [ Ag+ ]2 · [ CrO42Ч ]
ѕусть мол€рна€ концентраци€ Ag2CrO4 в насыщенном растворе равна x, тогда [ CrO42Ч ]= x; [ Ag+ ]=2 x.
1,3∙10--12 = (2 x)2· x
1,3∙10Ч12 = 4 x 3
C (Ag2CrO4)= x = 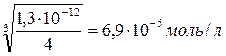
ƒл€ перевода этой величины в г/л, необходимо умножить ее на мол€рную массу Ag2CrO4:
6,9·10--5 моль/л ·332 г/моль =0,023 г/л
ќтвет: растворимость Ag2CrO4 равна 0,023 г/л.
ѕример 6
ќбразуетс€ ли осадок гидроксида железа (III), если к 1 л 0,006 ћ раствора хлорида железа (III) прибавить 0,125 л 0,0001 ћ раствора гидроксида кали€? ѕроизведение растворимости гидроксида железа (III) равно 3,8×10-38. —тепень электролитической диссоциации исходных веществ прин€ть равной единице.
–ешение
¬ насыщенном растворе гидроксида железа (III) устанавливаетс€ равновесие
Fe(OH)3 ⇆ Fe3++3OH-,
дл€ которого произведение растворимости, в силу малой концентрации растворов, может быть выражено через равновесные мол€рные концентрации:
ѕ–=[Fe3+][OH-]3.
ћол€рна€ концентраци€ катионов железа равна мол€рной концентрации хлорида железа (III), а концентраци€ ионов ќЌ- равна концентрации гидроксида кали€. –ассчитаем концентрации ионов, принима€ во внимание, что после сливани€ растворов гидроксида железа (III) и гидроксида кали€ объем раствора стал 1 л + 0,125 л = 1,125 л.
 ,
,
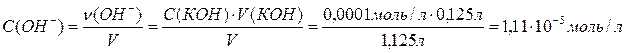
[Fe3+][OH-]3= 0,0053×(1,11×10-5)3 = 7,2×10-18.
“ак как ѕ–<[Fe3+][OH-]3, то в соответствии с (2.5.13) можно сделать вывод, что в заданных услови€ будет выпадать осадок гидроксида железа(III).
Ѕуферные растворы
Ѕуферными называют растворы, рЌ которых не измен€етс€ при разбавлении или добавлении небольших количеств сильной кислоты или сильного основани€.
|
|
|
Ѕуферный раствор состоит из растворенных в воде слабой кислоты и ее соли, например, —Ќ3—ќќЌ и —Ќ3——ќNa, или из слабого основани€ и его соли, например, NH4OH и NH4Cl.
»з слабых многоосновных кислот и их кислых солей также можно приготовить буферные системы, например, Ќ3–ќ4 Ц NaH2PO4 или
NaH2–ќ4 Ц Na2HPO4.
«начени€ рЌ буферного раствора, состо€щего из слабой кислоты и ее соли, рассчитывают по формуле (2.6.1):
рЌ= р Ќј - lg  , (2.6.1)
, (2.6.1)
где —Ќј и —¬ј Ц мол€рные концентрации кислоты и соли соответственно в буферном растворе, моль/л;
Ќј Ц константа диссоциации кислоты.
»з уравнени€ (2.6.1) следует, что значени€ рЌ буферных растворов зависит только от отношени€ общих концентраций компонентов раствора и не зависит от разбавлени€ (до определенных пределов). ѕри изменении объема раствора концентраци€ каждого компонента измен€етс€ в одинаковое число раз, а отношение концентраций не измен€етс€.
ѕри условии —Ќј= —¬ј отношение 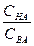 =1 и lg
=1 и lg 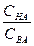 =0, то есть, рЌ= р Ќј.
=0, то есть, рЌ= р Ќј.
ƒл€ буферных растворов, состо€щих из слабого основани€ ¬ќЌ и его соли ¬ј, значение рЌ рассчитывают по формуле (2.6.2):
рЌ=14 - р ¬ќЌ + lg 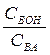 (2.6.2)
(2.6.2)
¬еличину буферного действи€ характеризуют с помощью буферной емкости, равной количеству вещества эквивалентов сильной кислоты или сильного основани€, которое нужно добавить к 1 литру буферного раствора, чтобы изменить рЌна единицу. Ѕуферна€ емкость измер€етс€ в моль экв./л или в ммоль экв./л.
Ѕуферна€ емкость зависит от природы и общих концентраций компонентов буферного раствора, а также от соотношени€ их концентраций. „ем больше концентраци€ компонентов буферного раствора и чем ближе к единице отношение 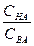 (в случае кислых буферов) и
(в случае кислых буферов) и 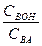 (в случае основных буферов), тем больше буферна€ емкость. Ѕуферна€ емкость максимальна, когда вышеприведенные отношени€ равны единице.
(в случае основных буферов), тем больше буферна€ емкость. Ѕуферна€ емкость максимальна, когда вышеприведенные отношени€ равны единице.
ѕусть Ѕ Ц буферна€ емкость, а и b Ц количество вещества эквивалентов соответственно кислоты (ЌCl) и основани€ (NaOH), тогда справедливы формулы (2.6.3) и (2.6.4):
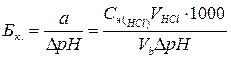 (2.6.3)
(2.6.3)
 , (2.6.4)
, (2.6.4)
где  Ц изменение рЌ при добавлении данного количества кислоты или основани€;
Ц изменение рЌ при добавлении данного количества кислоты или основани€;
Cн(HCl) и Cн(NaOH) Ц нормальные концентрации кислоты и основани€ соответственно, моль экв./ л;
VHCl, VNaOH, Vb Ц объемы кислоты, щелочи и буферного раствора соответственно, л;
1000 Ц коэффициент пересчета дл€ того случа€, когда буферна€ емкость измер€етс€ в ммоль экв./л.
ѕример 1
¬ычислить [Ќ+], [ќЌ-] и рЌ раствора, образовавшегос€ в результате смешивани€ 30 мл 0,1 ћ раствора уксусной кислоты —Ќ3—ќќЌ и 50 мл 0,3 ћ раствора ацетата кали€ —Ќ3—ќќ .
–ешение
ќбъем образовавшегос€ раствора V = 30+50=80 мл.
онцентрации уксусной кислоты и ацетата кали€ после смешивани€:
[ —Ќ3—ќќЌ ] = 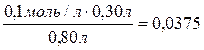 моль/л,
моль/л,
[ —Ќ3—ќќ ] = 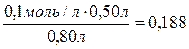 моль/л.
моль/л.
“ак как катионы водорода образуютс€ только за счет диссоциации уксусной кислоты, а ацетат анионы также и за счет диссоциации ацетата кали€, то, принима€ за ’ количество продиссоциировавшей кислоты, получим:
[ —Ќ3—ќќЌ ]ост = (0,0375 Ц ’) моль/л;
[Ќ+]= ’ моль/л;
[ —Ќ3—ќќ -] = (0,188+ ’) моль/л.
¬ыражение дл€ константы диссоциации уксусной кислоты:

“ак как ’ << 0,0375 то его вкладом в сумму и разность предыдущего выражени€ можно пренебречь.
“огда
’ = [H+] = 1,75Ј10-5Ј0,0375: 0,188= 3,5Ј10-6 моль/л;
[OH-] = 10-14: 3,5Ј10-6 =2,9Ј10-9 моль/л;
рЌ= -lg(3,5Ј10-6) = 5,46.
“акой же результат может быть получен, если воспользоватьс€ формулой (2.6.1):
рЌ = р Ќј - lg 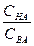 = - lg (1,74Ј10-5) - lg
= - lg (1,74Ј10-5) - lg  = 4,76 Ц (-0,7) = 5,46.
= 4,76 Ц (-0,7) = 5,46.
ѕример 2
ќпределить рЌ раствора, получившегос€ в результате смешени€ равных объемов 0,12 ћ растворов —Ќ3—ќќЌ и —Ќ3—ќќ .
–ешение
ѕосле смешени€ —кис= —соль= 0,12/2=0,06 моль/л
рЌ = р Ќј - lg 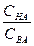 = -lg(1,74Ј10-5 ) - lg
= -lg(1,74Ј10-5 ) - lg  = 4,76 Ц lg1 = 4,76 - 0 = 4,76.
= 4,76 Ц lg1 = 4,76 - 0 = 4,76.
ѕример 3
—колько миллилитров 0,25ћ раствора ацетата кали€ —Ќ3—ќќ следует прибавить к 50 мл 1,0ћ раствора уксусной кислоты —Ќ3—ќќЌ, чтобы получить буферную систему с рЌ=3?
–ешение
¬оспользуемс€ формулой (2.6.1):
рЌ = р Ќј - lg 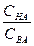 ;
;
lg 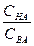 = р Ќј Ц рЌ = 4,76 - 3=1,76;
= р Ќј Ц рЌ = 4,76 - 3=1,76;
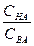 = 101,76 = 57,5
= 101,76 = 57,5
Ёто означает, что после смешени€ в полученном растворе концентраци€ кислоты в 57,5 раза должна быть выше, чем концентраци€ соли. ¬ растворе кислоты содержитс€ 0,05Ј1,0=0,05 моль кислоты. —ледовательно, количество вещества соли в этом растворе должно быть в 57,5 раза меньше, а именно 0,05:57,5=0,00087 моль.
Ќайдем объем 0,25 ћ раствора ацетата кали€:
V =0,00087/ 0,25 = 0,00348 л = 3,48 мл.
ѕример 4
Ѕуферна€ система, состо€ща€ из дигидрофосфата кали€ Ќ2–ќ4 и гидрофосфата кали€ 2Ќ–ќ4 приготовлена при мольном соотношении солей 16:1. ќпределить рЌ данного раствора.
|
|
|
–ешение
¬ данном случае роль слабой кислоты играет анион Ќ2–ќ4Ц ( 2= 6,2Ј10-8 или р = 7,2).
рЌ = р Ќј Ц lg 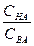 ,
,
рЌ = 7,2 Ц lg16= 6,0
ѕример 5
¬ычислить рЌ буферного раствора, содержащего 0,2 моль NH4Cl и 0,2 моль NH4ќЌ.
–ешение
¬ соответствии с формулой (2.6.2), рЌ =14 - р ¬ќЌ + lg 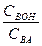 .
.
“ак как р (NH4ќЌ) = 4,755, то рЌ = 14 - 4,755 + lg  = 9,25.
= 9,25.
ѕример 6
100 мл 0,5 ћ раствора Ќ—ќќNa прилили 440 мл 0,1 ћраствора Ќ—ќќЌ. –ассчитать буферную емкость по кислоте и по основанию дл€ образовавшегос€ раствора с рЌ=3,8.
–ешение
–ассчитаем буферную емкость по кислоте, то есть, сколько моль эквивалентов сол€ной кислоты нужно прибавить к 1 л буфера, чтобы понизить его рЌ на единицу.
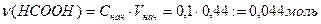
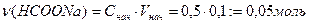
¬ычислим концентрации муравьиной кислоты и формиата натри€ в буферном растворе.
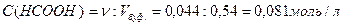

ѕусть х Ц число моль эквивалентов сол€ной кислоты, прибавленных к одномулитру буфера дл€ уменьшени€ рЌ на единицу, то есть буферна€ емкость.
ѕри добавлении сол€ной кислоты протекает реакци€:
HCOONa + HCl = HCOOH + NaCl
“огда концентраци€ муравьиной кислоты возрастет на х моль/л и станет равной (0,0018+ х) моль/л, а концентраци€ формиата натри€ уменьшитс€ на х моль/л и станет равной (0,0014 Ц х) моль/л, при этом рЌ понизитс€ до 2,8.
— учетом уравнени€ (2.6.1), запишем:
 .
.
»з данного выражени€ можно найти буферную емкость, обозначенную переменной х. Ѕуферна€ емкость по кислоте равна 0,075 моль/л.
–ассчитаем буферную емкость по основанию. ѕусть у Ц буферна€ емкость по основанию.
ѕри добавлении основани€ протекает реакци€:
HCOOH + NaOH = HCOONa + H2O
“огда при добавлении гидроксида натри€ концентраци€ муравьиной кислоты уменьшитс€ на у моль/л, а концентраци€ формиата натри€ увеличитс€ на у моль/л, при этом водородный показатель буфера возрастет на единицу и станет равным 4,8.
¬ соответствии с уравнением (2.6.1), запишем:
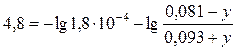 .
.
–еша€ это уравнение относительно у, получаем у= 0,067 моль/л. »так, буферна€ емкость по основанию равна 0,067 моль/л.
√идролиз солей
√идролиз (разложение водой (лат.)) Ц в широком смысле слова Ц это процесс обменного взаимодействи€ между водой и веществом. √идролизу подвергаютс€ соединени€ различных классов, в том числе и соли.
√идролиз соли Ц это химическое взаимодействие ионов соли с молекулами воды, привод€щее к образованию слабого электролита. ¬ результате гидролиза происходит смещение равновеси€ диссоциации воды вследствие образовани€ малодиссоциирующих соединений, малорастворимых или летучих веществ. ¬ соответствии с этим гидролизуютс€ только катионы слабых оснований и анионы слабых кислот, то есть гидролизу подвергаютс€ три типа солей:
¾ соли, образованные слабыми основани€ми и сильными кислотами (гидролиз по катиону), например, NЌ4CI, Cu(N03)2, Fe—13;
¾ соли, образованные сильными основани€ми и слабыми кислотами (гидролиз по аниону), например, —Ќ3—ќќNa, Na2CO3, NaHCO3;
¾ соли, образованные слабыми основани€ми и слабыми кислотами (гидролиз по катиону и аниону), например, CH3COONЌ4, јI2S3, (NH4)2S.
—оли, образованные сильными основани€ми и сильными кислотами (NaCI, KNO3, NaCIO4 и другие), гидролизу не подвергаютс€, так как при растворении их в воде не происходит св€зывани€ ни катионов Ќ+, ни анионов ќЌЦ.
–ассмотрим написание ионно-молекул€рных уравнений гидролиза на примере соли Fе—13. Ёта соль образована слабым основанием Fе(ќЌ)3 и сильной кислотой H—1, следовательно, гидролизу подвергаетс€ только катион соли Fe3+.
¬ соответствии с ионным уравнением
Fe3++HOHD[FeOH]2++H+ (2.7.1)
при гидролизе происходит разрыв св€зи ЌЧќЌ в молекуле воды, и образуетс€ слабый электролит [FeOH]2+, его константа диссоциации K(FeOH2+)=1,4Ј10Ц12. »оны Ќ+ создают кислотную среду.
¬ молекул€рной форме уравнение гидролиза имеет вид
FeCl3+HOHDFeOHCl2+HCl. (2.7.2)
”равнени€ (2.7.1) и (2.7.2) отражают первую ступень гидролиза соли FeCl3. √идролиз солей, образованных слабыми многокислотными основани€ми и сильными кислотами (Cu(NO3)2, ј1—13 и другие), или солей, образованных слабыми многоосновными кислотами и сильными основани€ми (Na2S, 3–O4 и другие), протекает ступенчато, но при обычных услови€х практически ограничиваетс€ первой ступенью.
ѕри нагревании и разбавлении раствора гидролиз усиливаетс€, и станов€тс€ заметными последующие ступени гидролиза.
¬тора€ ступень:
[FeOH]2++ЌќЌD[Fe(ќЌ)2]++Ќ+
FeOHCl2+HOHDFe(OH)2Cl+HCl; (2.7.3)
треть€ ступень:
[Fе(ќЌ)2]++ЌќЌDFе(ќЌ)3+Ќ+
Fе(ќЌ)2—1+ЌќЌDFе(ќЌ)3+Ќ—1. (2.7.4)
ѕреимущественное протекание гидролиза по первой ступени объ€сн€етс€ тем, что, чем слабее образующийс€ электролит, тем сильнее гидролизуетс€ соль. ƒл€ слабых электролитов, диссоциирующих ступенчато, всегда справедливо следующее соотношение констант диссоциации по ступен€м: 1> 2> 3 (данные по константам диссоциации приведены в таблице 2.7.1). ѕоэтому в приведенном примере частица [FeOЌ]2+, диссоциаци€ которой характеризуетс€ константой 3, €вл€етс€ наиболее слабым электролитом из образующихс€ при гидролизе, что и обусловливает преимущественное протекание гидролиза по первой ступени, на которой образуетс€ эта частица.
—ледует подчеркнуть, что ступенчатый гидролиз солей нельз€ изображать суммарным уравнением, например, Fe3++«Ќ2ќ=Fe(ќH)3+3H+, получаемым путем сложени€ уравнений отдельных ступеней гидролиза (при сложении сокращаетс€ главный продукт гидролиза Ц ион [FeќЌ]2+).
≈сли соль образована сильным основанием и слабой кислотой (Na2S, Na2—ќ3, Na2Sќ3, Na—N, NaNO2, CH3COONa и другие), гидролиз происходит по аниону, то есть по остатку слабой кислоты.
–ассмотрим ступенчатый гидролиз Na2S.
ѕерва€ ступень:
S2Ц+ЌќЌDЌSЦ+ќЌЦ (2.7.5)
онстанта диссоциации иона ЌSЦ очень мала, 2(ЌSЦ)=4Ј10Ц14, поэтому равновесие реакции (2.7.5) смещено вправо. ¬ результате в растворе повышаетс€ концентраци€ ќЌЦ, то есть реакци€ среды щелочна€ (рЌ>7).
–еакци€ (2.7.5) в молекул€рной форме имеет следующий вид:
Na2S+HOHDNaHS+NaOH.
¬тора€ ступень:
ЌSЦ+ЌќЌDЌ2S+NaOH (2.7.6)
NaЌS+ЌќЌDЌ2S+NaOH.
онстанта диссоциации 1(Ќ2S)=10-7, поэтому можно заключить, что реакци€ (2.7.6) менее смещена вправо, чем реакци€ (2.7.5), то есть гидролиз в основном протекает по первой ступени, и в растворе Na2S преобладают ионы ЌSЦ, ќЌЦ и Na+ в составе соединений NaHS и NaOH.
≈сли соль образована слабым основанием и слабой кислотой (CЌ3—ќќNH4 и другие), то гидролизу подвергаютс€ катион и анион соли:
—Ќ3—ќќЦ+NH4++ЌќЌD—Ќ3—ќќH+NЌ3ЈЌ2ќ. (2.7.7)
¬ подобном случае реакци€ среды близка к нейтральной и зависит от сравнительной силы образующихс€ кислоты и основани€ (соотношени€ констант диссоциации кислоты и основани€). ≈сли »—.> ќ—., то реакци€ среды слабокисла€, если ќ—.> »—., то среда слабощелочна€, если ќ—.= »—., то среда нейтральна€.
‘ормулы дл€ расчета количественных характеристик гидролиза различных типов солей привод€тс€ в таблице 2.7.2.
“аблица 2.7.1 Ц онстанты электролитической диссоциации некоторых слабых электролитов в водных растворах при 25∞ —
| Ќазвание электролита | ‘ормула | онстанта диссоциации |
| јзотиста€ кислота | HNќ2 | 4,6Ј10Ц 4 |
| —инильна€ кислота | HCN | 7,9Ј10Ц10 |
| —ерниста€ кислота | H2SO3 | K1=1,6Ј10Ц2 2=6,2Ј10Ц8 |
| —ероводородна€ кислота | H2S | K1=1Ј10Ц7 K2=4Ј10Ц14 |
| ”гольна€ кислота | Ќ2—ќ3 | K1= 4,5Ј10Ц7 K2=4,7Ј10Ц11 |
| ”ксусна€ кислота | —Ќ3—ќќЌ | 1,75Ј10Ц5 |
| ‘осфорна€ кислота | Ќ3–ќ4 | 1=7,1Ј10Ц3 2=6,3Ј10Ц8 3=5Ј10Ц13 |
| ’лорноватиста€ кислота | HCIO | 5Ј10Ц8 |
| √идроксид аммони€ | NH4ќH или NЌ3ЈЌ2ќ | 1,75Ј10Ц5 |
| √идроксид алюмини€ | ј1(ќЌ)3 | 3=10Ц9 |
| √идроксид железа (III) | Fе(ќЌ)3 | K1=4Ј10Ц9 2=6Ј10Ц11 3=4Ј10Ц12 |
| √идроксид магни€ | ћg(ќЌ)2 | K2=1,5Ј10Ц3 |
| √идроксид меди (II) | —u(ќЌ)2 | K2=10Ц7 |
| √идроксид кальци€ | —а(ќЌ)2 | K2=2Ј10Ц1 |
| √идроксид цинка | Zn(OH)2 | 1=10Ц5 2=5Ј10Ц7 |
“ак как гидролиз большинства солей €вл€етс€ обратимым процессом, то степень гидролиза можно измен€ть, смеща€ равновесие реакций гидролиза. „тобы усилить гидролиз, необходимо уменьшить концентрацию соли, повысить температуру, подкислить раствор соли, дл€ которого рЌ>7, либо подщелочить раствор соли, дл€ которого рЌ<7.
ѕри сливании растворов солей, одна из которых гидролизуетс€ по катиону, а друга€ Ц по аниону, происходит взаимное усиление гидролиза обеих солей, и гидролиз может протекать необратимо.
“аблица 2.7.2 Ц ’арактеристики растворов гидролизующихс€ солей
| “ип соли | –еакци€ среды в растворе | ѕродукты гидролиза | онстанта гидролиза √ | —тепень гидролиза h | онцентрации [H+] или [OЌЦ] |
| —оль, образованна€ слабым основанием и сильной кислотой (гидролиз по катиону) | кисла€ pЌ< 7 | слабые основани€ или основные соли |

|

|
[H+]= 
|
| Cоль, образованна€ слабой кислотой и сильным основанием (гидролиз по аниону) | щелочна€ pЌ>7 | —лабые кислоты или кислые соли |

|

|
[ќЌЦ]=
=  [H+]=
=
[H+]=
= 
|
| —оль, образованна€ слабым основанием и слабой кислотой (гидролиз по аниону и по катиону). | слабокисла€, слабощелочна€ или нейтральна€ в завилимости от соотношени€ констант диссоциации продуктов гидролиза
ной
pЌ  7 7
| —лабые кислоты и слабые основани€ или кислые соли |
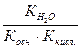
|
ї  ,
более точно: ,
более точно:
 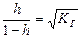
|
[H+]=
= 
|
| исла€ соль, образованна€ слабой двухосновной кислотой и сильным основанием | pЌ>7, если (K√>Kкисл I); рЌ <7, если (K√<Kкисл I) | —лаба€ кислота |

|

|
 [H+]=
= [H+]=
= 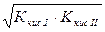
|
“ак, в растворах Fе—13 и Na2S, вз€тых в отдельности, гидролиз протекает практически только по первой ступени, и устанавливаютс€ соответственно равновеси€ (2.7.1) и (2.7.5).
ѕри сливании этих растворов происходит реакци€ нейтрализации:
Ќ+ + ќЌ- = Ќ2ќ,
ѕри этом равновеси€ дл€ реакций (2.7.1) и (2.7.5) смещаютс€ вправо, вплоть до образовани€ конечных продуктов гидролиза обеих солей. »ными словами, протекает необратимый совместный гидролиз этих двух солей:
FeCl3 + 3Na2S + 6H2ќ = 2Fe(OH)3¯ + 3H2S≠ + 6NaCl
Fe3+ + 3S2-+ 6H2ќ = 2Fe(OH)3¯ + 3H2S≠
ѕо аналогичной схеме взаимного усилени€ гидролиза, привод€щего к образованию осадка и газа, взаимодействуют соли, образованные катионами јl3+, Cr3+, Fe3+ c карбонатами и сульфидами щелочных металлов. ѕоэтому эти соли невозможно получить реакци€ми обмена из водных растворов.
ѕример 1
Ќаписать молекул€рные и ионно-молекул€рные уравнени€ гидролиза сульфита кали€ K2SO3.
–ешение
—оль образована катионом сильного основани€ и анионом слабой двухосновной кислоты. √идролиз идет по аниону. ¬ растворе щелочна€ реакци€ среды.
1 ступень: 2Sќ3 + H2O D KHSO3 + ќЌ
SO32-Ц + Ќ2ќ D HSO3Ц+OHЧ
2 ступень: KHSO3 + Ќ2O D Ќ2Sќ3 + ќЌ
HSO3Ц+ Ќ2O D Ќ2Sќ3 + ќЌЦ
ѕример 2
Ќаписать молекул€рные и ионно-молекул€рные уравнени€ гидролиза нитрата свинца (II)
–ешение
—оль образована катионом слабого двухкислотного основани€ и анионом сильной кислоты. √идролиз идет по катиону. –аствор имеет кислую реакцию среды.
1 ступень: Pb(NO3)2 + Ќ2ќ D (PbOH)NO3 + HNO3
–№2+ + Ќ2ќ D –№ќЌЦ + Ќ+
2 ступень: (–№ќЌ)Nќ3 + Ќ2ќ D Pb(OH)2 + ЌNќ3
–bќЌЦ + Ќ2ќ D Pb(OH)2 + Ќ+
ѕример 3
Ќаписать уравнение гидролиза цианида аммони€ NH4CN. акова реакци€ среды в водном растворе этой соли?
–ешение
—оль образована катионом слабого основани€ и анионом слабой кислоты, поэтому гидролиз идет как по катиону, так и по аниону:
NH4CN + Ќ2ќ D NH4OH + HCN
NH4+ + CNЦ + Ќ2ќ D NH4OH + HCN
ƒл€ оценки реакции среды необходимо сравнить константы диссоциации NH4OH и HCN (таблица 2.7.1). “ак как константа диссоциации гидроксида аммони€ больше, чем константа диссоциации синильной кислоты, то гидроксид аммони€ €вл€етс€ более сильным электролитом. ¬ растворе будут преобладать ионы ќЌ Ц, следовательно, реакци€ среды щелочна€.
ѕример 4
”читыва€ только первую ступень гидролиза 0,5 ћ раствора Na2S, рассчитайте константу гидролиза, степень гидролиза и рЌ среды.
–ешение
√идролизу по первой ступени соответствует следующее ионно-молекул€рное уравнение:
S2- + Ќ2ќ D ЌSЦ + OHЦ
онстанта гидролиза Na2S пo первой ступени есть отношение ионного произведени€ воды к константе диссоциации слабой кислоты HS Ц.
онстанта диссоциации гидросульфид-иона представл€ет собой константу диссоциации H2S по второй ступени, ее значение можно вз€ть из таблицы 2.7.1.
√ = 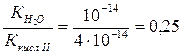
—тепень гидролиза:
h = 
ƒл€ нахождени€ рЌ среды рассчитаем концентрацию водородных ионов:
[H+]= 
pH = - lg[H+] = - lg (2.83Ј10 Ц 14 ) = 13,55 
∆есткость воды
ѕриродна€ вода в своем составе всегда содержит различные примеси: соли и газы, механические примеси, наход€щиес€ во взвешенном состо€нии, эмульсии, гидрозоли и другие образовани€. Ќекоторые соли, присутствующие в воде, вызывают ее жесткость.
∆есткость воды Ц это совокупность свойств воды, обусловленных присутствием в ней катионов Ca2+ и Mg2+, реже Fe2+.
—одержание в воде большого количества примесей растворимых солей кальци€ и магни€ делает ее непригодной дл€ технических целей. ѕовышенна€ жесткость воды приводит к образованию накипи в паровых котлах и бытовой посуде. Ёто ухудшает теплообмен, а, следовательно, приводит к перерасходу топлива, электроэнергии, перегреву металлических поверхностей.
¬ жесткой воде ухудшаетс€ пенообразование и увеличиваетс€ расход мыла при стирке, так как часть содержащихс€ в нем растворимых солей жирных кислот переходит в нерастворимое состо€ние:
2C17H35COONa + CaSO4 = (C17H35COO)2Ca ¯ + Na2SO4.
ѕри этом также ухудшаетс€ качество тканей вследствие осаждени€ на них нерастворимых кальциевых и магниевых солей высших жирных кислот.
¬ воде с повышенной жесткостью плохо развариваютс€ овощи и м€со, так как катионы кальци€ образуют с белками нерастворимые соединени€. Ѕольша€ магниева€ жесткость придает воде горький вкус.
—уммарное содержание Ca2+ и Mg2+ в воде называетс€ общей жесткостью. ∆есткость воды оцениваетс€ по-разному. ¬ нашей стране ее чаще всего выражают количеством вещества эквивалентов кальци€ и магни€ (в ммоль) в одном литре воды.
∆есткость воды хоз€йственно-питьевых водопроводов не должна превышать 7 ммоль экв./л. ѕо величине жесткости воду условно подраздел€ют на м€гкую (до 4 ммоль экв./л), средней жесткости (4 Ц 8 ммоль экв./л), жесткую (8 Ц 12 ммоль экв./л) и очень жесткую (более 12 ммоль экв./л).
ќбща€ жесткость складываетс€ из карбонатной (временной) и некарбонатной (посто€нной). арбонатна€ жесткость обусловлена присутствием в воде гидрокарбонатов кальци€, магни€, а иногда также и гидрокарбоната
железа (II). Ётот вид жесткости можно устранить кип€чением:
Ca(HCO3)2  CaCO3 ¯ + H2O + CO2 ≠
CaCO3 ¯ + H2O + CO2 ≠
Mg(HCO3)2  MgCO3 ¯ + H2O + CO2 ≠;
MgCO3 ¯ + H2O + CO2 ≠;
2MgCO3 + H2O  (MgOH)2CO3 ¯ + CO2 ≠
(MgOH)2CO3 ¯ + CO2 ≠
(MgOH)2CO3+H2O  2Mg(OH)2 ¯ +CO2 ≠
2Mg(OH)2 ¯ +CO2 ≠
Fe(HCO3)2  Fe(OH)2 ¯ + 2CO2 ≠;
Fe(OH)2 ¯ + 2CO2 ≠;
4Fe(OH)2 + O2 + 2 H2O Ѓ 4Fe(OH)3.
ѕри кип€чении воды, растворенные в ней гидрокарбонаты разлагаютс€, и карбонатна€ жесткость сильно снижаетс€, однако полного устранени€ карбонатной жесткости не происходит вследствие того, что карбонаты кальци€ и магни€ несколько растворим в воде.
Ќар€ду с пон€тием карбонатна€ жесткость, используетс€ термин устранима€ жесткость. Ёто та величина, на которую понижаетс€ жесткость при дес€тиминутном кип€чении воды. ∆есткость, оставша€с€ после кип€чени€ воды, называетс€ посто€нной жесткостью.
ƒл€ уменьшени€ карбонатной жесткости примен€ют также метод известковани€:
Ca(HCO3)2 + Ca(OH)2 Ѓ 2CaCO3 ¯ + 2H2O
Mg(HCO3)2 + 2Ca(OH)2 Ѓ Mg(OH)2 ¯ + 2CaCO3 ¯ + 2H2O.
Ќекарбонатна€ жесткость обусловлена присутствием в воде растворимых, устойчивых к нагреванию солей кальци€ и магни€. „аще всего это сульфаты и хлориды.
Ќекарбонатную жесткость можно устранить обработкой воды карбонатом или фосфатом натри€:
MeSO4 + Na2CO3 Ѓ MeCO3 ¯ + Na2SO4.
ƒл€ устранени€ жесткости примен€ют также катиониты. Ёто ионообменные смолы и алюмосиликаты, содержащие в своем составе подвижные катионы, например, Na+, H+, способные обмениватьс€ на катионы среды. ≈сли пропускать воду через слои катионита, то его подвижные катионы будут обмениватьс€ на катионы кальци€ и магни€, при этом катионы жесткости остаютс€ в катионите, а подвижные ионы катионита переход€т в раствор. ѕри помощи
Ќ+ -катионировани€ ум€гчают воду с преобладанием карбонатной жесткости, а при помощи Na+ -катионировани€ Ц с преобладанием некарбонатной жесткости.
ƒл€ ум€гчени€ воды можно также использовать и физические методы: электродиализ, ультразвуковую, магнитную и магнитно-ионизационную обработку воды.
ѕример 1
–ассчитайте общую жесткость воды (ммоль экв./л), если в 0,15 л воды содержитс€ 16,2 мг гидрокарбоната кальци€, 2,92 мг гидрокарбоната магни€, 11,10 мг хлорида кальци€ и 9,50 мг хлорида магни€.
–ешение
¬ыразим общую жесткость воды как сумму миллимол€рных концентраций эквивалентов двухзар€дных катионов металлов (или соответствующих им солей) в воде:
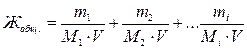 ,
,
где m1, m2, mi Ц массы двухзар€дных катионов металлов, либо массы соответствующих им солей, либо массы веществ, пошедших на устранение жесткости воды, мг;
M1, M2, Mi Ц мол€рные массы эквивалентов двухзар€дных катионов металлов, либо массы соответствующих им солей, либо массы веществ, пошедших на устранение жесткости воды, г/моль;
V Ц объем воды, л.
ћол€рные массы эквивалентов солей, перечисленных в условии задачи, рассчитываютс€ по формуле:
 ,
,
где z Ц эквивалентное число, дл€ солей жесткости оно равно 2;
 Ц мол€рна€ масса соли ’.
Ц мол€рна€ масса соли ’.
| ‘ормула соли | —а(HCќ3)2 | Mg(HCќ3)2 | —аCl2 | MgCl2 |
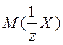 , г/моль , г/моль
| 55,5 |
ќбща€ жесткость данного образца воды равна сумме временной и посто€нной жесткости:
 =
=
=2,56 ммоль экв./л
ѕример 2
ќпределите временную жесткость воды, если на титрование 0,1 л воды, содержащей гидрокарбонат магни€, израсходовано 7,2∙10-3 л 0,14 н. раствора HCl.
–ешение
ѕри титровании воды сол€ной кислотой происходит реакци€:
Mg(HCO3)2 + 2Ќ—1 = MgCl2 + 2Ќ2ќ + 2—ќ2
¬ременную жесткость воды рассчитаем по формуле (8.1):

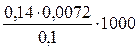 =10,08 ммоль экв./л
=10,08 ммоль экв./л
ѕример 3
ƒл€ устранени€ общей жесткости по известково-содовому методу добавлено 7,4 г —а(ќЌ) 2 и 5,3 г Na2CO3. –ассчитать временную и посто€нную жесткость воды.
–ешение
ƒобавление к воде —а(ќЌ)2 может устранить временную жесткость, а добавление Na2CO3 Ц посто€нную жесткость. ѕри добавлении этих реагентов к воде происход€т следующие реакции:
Ca(HCO3)2 + Ca(OH)2 Ѓ 2CaCO3 ¯ + 2H2O
Mg(HCO3)2 + 2Ca(OH)2 Ѓ Mg(OH)2 ¯ + 2CaCO3 ¯ + 2H2O.
Ca(Nќ3)2 + Na2CO3 = MeCO3 ¯ + 2NaNO3
Mg(Nќ3)2 + Na2CO3 = MeCO3 ¯ + 2NaNO3
¬ременную жесткость воды ∆вр измер€ют количеством вещества эквивалентов гидроксида кальци€, участвующего в реакции, а посто€нную жесткость ∆пост Ц количеством вещества эквивалентов карбоната натри€.
M(1/2—а(ќЌ)2) = 74/2=37 г/моль;
M(1/2Na2CO3) = 106/2=53 г/моль;
 7400/(37∙50) = 4 ммоль экв./л;
7400/(37∙50) = 4 ммоль экв./л;
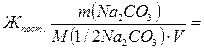 5300/(53∙50) = 2 ммоль экв./л
5300/(53∙50) = 2 ммоль экв./л
ќбща€ жесткость воды равна:
∆общ = ∆вр + ∆пост = 4 + 2 = 6 ммоль экв./л






