22. ”становите соответствие между типом реакции и уравнением, по которому она протекает.
| “»ѕ –≈ј ÷»» | ”–ј¬Ќ≈Ќ»≈ –≈ј ÷»» | ||||
| ј) | соединени€ | 1) | 2—Ќ4 → —2Ќ2 + 3Ќ2 | ||
| Ѕ) | разложени€ | 2) | 2SO2 + O2 = 2SO3 | ||
| ¬) | замещени€ | 3) | —Ќ4 + Cl2 → —Ќ3Cl + HCl | ||
| √) | обмена | 4) | AgNO3 + KCl = AgCl↓ + KNO3 | ||
| ј | Ѕ | ¬ | √ | ||
23. ”становите соответствие между типом реакции и уравнением, по которому она протекает.
| “»ѕ –≈ј ÷»» | ”–ј¬Ќ≈Ќ»≈ –≈ј ÷»» | ||||
| ј) | соединени€, эндотермическа€ | 1) | Cu(OH)2 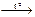 CuO + H2O CuO + H2O
| ||
| Ѕ) | соединени€, экзотермическа€ | 2) | CaO + H2O = Ca(OH)2 | ||
| ¬) | разложени€, эндотермическа€ | 3) | N2 + O2  2NO 2NO
| ||
| √) | обмена, экзотермическа€ | 4) | 2K + 2H2O = 2KOH + H2 | ||
| 5) | HCl + NaOH = NaCl + H2O | ||||
| ј | Ѕ | ¬ | √ | ||
“епловой эффект химической реакции
Ѕазовый уровень сложности
1. ¬ результате реакции, термохимическое уравнение которой
2Mg + O2 = 2MgO + 1200 кƒж,
выделилось 600 кƒж теплоты. ќбъем (н.у.) вступившего при этом в реакцию кислорода равен
| 1) | 44,8 л | 2) | 22,4 л | 3) | 11,2 л | 4) | 112 л |
2. ¬ результате реакции, термохимическое уравнение которой
2—a + O2 = 2CaO + 1270 кƒж,
образовалось 56 г оксида кальци€. оличество выделившейс€ при этом теплоты равно
| 1) | 635 кƒж | 2) | 2540 кƒж | 3) | 317,5 кƒж | 4) | 254 кƒж |
3. ¬ соответствии с термохимическим уравнением реакции
4Al(тв) + 3O2(г) = 2Al2O3(тв) + 3352 кƒж
количество теплоты, выдел€ющеес€ при сгорании 270 г алюмини€, равно
| 1) | 6704 кƒж | 2) | 1340,8 кƒж | 3) | 8380 кƒж | 4) | 838 кƒж |
4. ¬ результате реакции, термохимическое уравнение которой
S(тв) + O2(г) = SO2(г) + 297 кƒж,
выделилось 1188 кƒж теплоты. ќбъем (н.у.) образовавшегос€ при этом сернистого газа равен
| 1) | 44,8 л | 2) | 89,6 л | 3) | 890 л | 4) | 112 л |
5. ¬ результате реакции, термохимическое уравнение которой
2SO2(г) + O2(г) = 2SO3(г) + 198 кƒж,
выделилось 990 кƒж теплоты. ћасса вступившего в реакцию сернистого газа равна
| 1) | 1280 г | 2) | 1600 г | 3) | 320 г | 4) | 640 г |
6. ¬ соответствии с термохимическим уравнением реакции
2—ќ(г) + ќ2(г) = 2—ќ2(г) + 568 кƒж
количество теплоты, выделившейс€ при сгорании 11,2 л (н.у.) оксида углерода(II), равно
| 1) | 113,6 кƒж | 2) | 142 кƒж | 3) | 284 кƒж | 4) | 838 кƒж |
7. ¬ результате реакции, термохимическое уравнение которой
2KClO3(тв) = 2KCl(тв) + 3O2(г) + 90 кƒж,
образовалось 224 л (н.у.) кислорода. оличество теплоты, выделившейс€ при этом, равно
| 1) | 450 кƒж | 2) | 270 кƒж | 3) | 300 кƒж | 4) | 150 кƒж |
8. ¬ результате реакции, термохимическое уравнение которой
N2(г) + 3Ќ2(г) = 2NЌ3(г) + 92 кƒж,
выделилось 46 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию водорода равен
|
|
|
| 1) | 67, 2 л | 2) | 44,8 л | 3) | 11,2 л | 4) | 33,6 л |
9. ¬ результате реакции, термохимическое уравнение которой
CH4(г) + 2O2(г) = CO2(г) + 2H2O(г) + 802 кƒж,
выделилось 160,4 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию кислорода равен
| 1) | 4,48 л | 2) | 13,44 л | 3) | 8,96 л | 4) | 11,2 л |
10. ¬ результате реакции, термохимическое уравнение которой
4NH3(г) + 5O2(г) = 4Nќ(г) + 6H2O(г) + 902 кƒж,
выделилось 451 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию аммиака равен
| 1) | 44,8 л | 2) | 112 л | 3) | 67,2 л | 4) | 22,4 л |
11. ¬ результате реакции, термохимическое уравнение которой
4NH3(г) + 3O2(г) = 2N2(г) + 6H2O(ж) + 1532 кƒж,
образовалось 22,4 л (н.у.) азота. оличество выделившейс€ при этом теплоты равно
| 1) | 1532 кƒж | 2) | 766 кƒж | 3) | 306,4 кƒж | 4) | 3064 кƒж |
12. ¬ результате реакции, термохимическое уравнение которой
2Ќ2(г) + ќ2(г) = 2Ќ2ќ(ж) + 572 кƒж,
образовалось 7,2 г воды. оличество выделившейс€ при этом теплоты равно
| 1) | 114,4 кƒж | 2) | 57,2 кƒж | 3) | 143 кƒж | 4) | 228,8 кƒж |
13. ¬ результате реакции, термохимическое уравнение которой
—(графит) + 2Ќ2(г) = —Ќ4(г) + 74,6 кƒж,
выделилось 373 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию водорода равен
| 1) | 448 л | 2) | 56 л | 3) | 112 л | 4) | 224 л |
14. ¬ результате реакции, термохимическое уравнение которой
CO2(г) + C(графит) = 2CO(г) Ц 173 кƒж,
поглотилось 34,6 кƒж теплоты. ќбъем (н.у.) образовавшегос€ при этом оксида углерода(II) равен
| 1) | 22,4 л | 2) | 4,48 л | 3) | 8,96 л | 4) | 11,2 л |
15. ¬ результате реакции, термохимическое уравнение которой
Ќ2(г) + —l2(г) = 2Ќ—l(г) + 184,6 кƒж,
образовалось 11,2 л (н.у.) хлороводорода. оличество выделившейс€ при этом теплоты равно
| 1) | 18,46 кƒж | 2) | 37,92 кƒж | 3) | 46,15 кƒж | 4) | 92,3 кƒж |
16. ¬ соответствии с термохимическим уравнением реакции
—6Ќ12ќ6(к) + 6ќ2(г) = 6—ќ2(г) + 6Ќ2ќ(ж) + 2800 кƒж
количество теплоты, выдел€ющеес€ при окислении 36 г глюкозы, равно
| 1) | 560 кƒж | 2) | 280 кƒж | 3) | 140 кƒж | 4) | 420 кƒж |
17. ¬ соответствии с термохимическим уравнением реакции
—2Ќ4(г) + 3ќ2(г) = 2—ќ2(г) + 2Ќ2ќ(ж) + 1410 кƒж
количество теплоты, выдел€ющеес€ при образовании 8,8 г углекислого газа, равно
| 1) | 705 кƒж | 2) | 70,5 кƒж | 3) | 282 кƒж | 4) | 141кƒж |
18. ¬ соответствии с термохимическим уравнением реакции
2—2Ќ6(г) + 7ќ2(г) = 4—ќ2(г) + 6Ќ2ќ(ж) + 3120 кƒж
при сгорании 112 л (н.у.) этана количество выделившейс€ теплоты равно
| 1) | 15600 кƒж | 2) | 7800 кƒж | 3) | 3900 кƒж | 4) | 1560 кƒж |
19. ¬ соответствии с термохимическим уравнением реакции
2—2Ќ2 + 5O2 = 4CO2 + 2H2O + 2610 кƒж
при сгорании 26 г ацетилена количество выделившейс€ теплоты равно
| 1) | 1305 кƒж | 2) | 5220 кƒж | 3) | 522 кƒж | 4) | 652,5 кƒж |
20. ¬ результате реакции, термохимическое уравнение которой
—2Ќ5ќЌ + 3ќ2 = 2—ќ2 + 3Ќ2ќ + 1374 кƒж,
выделилось 458 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию кислорода равен
| 1) | 144,4 | 2) | 33,6 л | 3) | 67,2 л | 4) | 22,4 л |
21. ¬ результате реакции, термохимическое уравнение которой
2NO(г) + O2(г) = 2NO2(г) + 114 кƒж,
выделилось 570 кƒж теплоты. ќбъем (н.у.) вступившего в реакцию оксида азота(II) равен
|
|
|
| 1) | 144,4 | 2) | 336 л | 3) | 112 л | 4) | 224 л |
22. ¬ результате реакции, термохимическое уравнение которой
N2(г) + O2(г) = 2NO(г) Ц 180,8 кƒж,
образовалось 56 л (н.у.) оксида азота(II). оличество поглотившейс€ при этом теплоты равно
| 1) | 90,4 кƒж | 2) | 113 кƒж | 3) | 226 кƒж | 4) | 452 кƒж |
23. ¬ результате реакции, термохимическое уравнение которой
2AgNO3(тв) = 2Ag(тв) + 2NO2(г) + O2(г) Ц 317 кƒж,
образовалось 10,8 г серебра. оличество поглотившейс€ при этом теплоты равно
| 1) | 31,7 кƒж | 2) | 15,85 кƒж | 3) | 63,4 кƒж | 4) | 12,68 кƒж |
“ермодинамика химических процессов. «акон √есса. ѕон€тие об энтальпии, энтропии, энергии √иббса*
1*. ќпределите ΔЌо образовани€ этилена по следующим данным:
| —(графит) + ќ2(г) = —ќ2(г), | ΔЌº = - 393,5 кƒж |
| Ќ2 + 1/2 ќ2(г) = Ќ2ќ(г), | ΔЌо = - 241,8 кƒж |
| —2Ќ4(г) + 3ќ2(г) = 2—ќ2(г) + 2Ќ2ќ(г), | ΔЌо= - 1323 кƒж |
2*. ¬ычислите ΔЌо образовани€ карбоната магни€, использу€ следующие данные:
| —(графит) + ќ2(г) = —ќ2(г), | ΔЌº = - 393,5 кƒж |
| 2ћg(тв) + ќ2(г) = 2ћgќ(тв), | ΔЌº = - 1203,6 кƒж |
| ћgќ(тв) + —ќ2(г) = ћg—ќ3(тв), | ΔЌº = - 117,7 кƒж |
3*. –ассчитайте ΔЌº реакции 2–(белый) + 5Cl2(г.) = 2–Cl5(г.), использу€ следующие данные:
| 2–(белый) + 3Cl2(г) = 2–Cl3(г), | ΔЌº = - 558,9 кƒж |
| –Cl3(г) + Cl2(г) = –Cl5(г) | ΔЌº = - 90,5 кƒж |
4*. ќксид серы (VI) трудно получить при непосредственном сгорании серы в кислороде. ќпределите энтальпию образовани€ SO3(г.), зна€, что энтальпи€ образовани€ SO2(г.) составл€ет Ц 297,1 кƒж/моль, а энтальпи€ реакции окислени€ 1 моль SO2(г.) газообразным кислородом до SO3(г.) равна Ц 99,0 кƒж.
5*. ќпределите энтальпию образовани€ глюкозы по следующим данным:
| —(графит) + ќ2(г) = —ќ2(г), | ΔЌº = - 393,5 кƒж |
| Ќ2 + 1/2 ќ2(г) = Ќ2ќ(г), | ΔЌо = - 241,8 кƒж |
| —6Ќ12ќ6(тв) + 6ќ2(г) = 6—ќ2(г) + 6Ќ2ќ(г), | ΔЌо= - 2538,8 кƒж |
6*. ќпределите, какой из сульфидов можно растворить в разбавленной сол€ной кислоте, использу€ следующие данные:
| ZnS(тв) + 2HCl(р-р) = H2S(г) + ZnCl2(р-р), | ΔGº = - 462,6 кƒж |
| PbS(тв)+ 2HCl(р-р) = H2S(г) + PbCl2(р-р), | ΔGº = + 31,0 кƒж |
7*. ќпределите, можно ли синтезировать хлориды фосфора из простых веществ при стандартных услови€х, использу€ следующие данные:
| –(белый) + 3/2Cl2 = –Cl3(г), | ΔGº = - 286,7 кƒж |
| –(белый) + 5/2Cl2 = –Cl5(г), | ΔGº = - 325,1 кƒж |
8*. ќпределите, какой из углеводородов можно получить из простых веществ при стандартных услови€х, использу€ следующие данные:
| —(графит) + Ќ2(г) = —Ќ4(г), | ΔGº = - 50,5 кƒж |
| 2—(графит) + 2Ќ2(г) = —2Ќ4(г), | ΔGº = + 68,1 кƒж |
| 2—(графит) + Ќ2(г) = —2Ќ2(г), | ΔGº = + 209,9 кƒж |
9*. ќпределите, какие из тетрагалогеналканов можно получить из простых веществ при стандартных услови€х, использу€ следующие данные:
| —(графит) + 2F2(г) = —F4(г), | ΔGº = - 636,0 кƒж |
| —(графит) + 2Cl2(г) = —Cl4(г), | ΔGº = - 64,1 кƒж |
| —(графит) + 2Br2(г) = —Br4(г), | ΔGº = + 36,0 кƒж |
| —(графит) + 2I2(г) = —I4(г), | ΔGº = + 124,9 кƒж |
10*. ќпределите, какой из гидроксидов (II) наиболее устойчив к окислению при стандартных услови€х, использу€ следующие данные:
| Fe(OH)2(тв) + 1/4ќ2(г) + 1/2Ќ2ќ(ж) = Fе(ќЌ)3(тв), | ΔGº = - 92,2 кƒж |
| —о(OH)2(тв) + 1/4ќ2(г) + 1/2Ќ2ќ(ж) = —о(ќЌ)3(тв), | ΔGº = - 23,7 кƒж |
| Ni(OH)2(тв) + 1/4ќ2(г) + 1/2Ќ2ќ(ж) = Ni(ќЌ)3(тв), | ΔGº = + 22,9 кƒж |
11*. Ёнтропи€ системы возрастает в процессах
| 1) | —аќ(тв) + SO2(г) = CaSO3(тв) |
| 2) | Fe3O4(тв) + 4Ќ2(г) = 3Fe(тв) + 4Ќ2ќ(ж) |
| 3) | —6Ќ12ќ6(тв) + 6ќ2(г) = 6—ќ2(г) + 6Ќ2ќ(г) |
| 4) | 2H2(г) + ќ2(г) = 2Ќ2ќ(ж) |
| 5) | —а—ќ3(тв) = —аќ(тв) + —ќ2(г) |
| 6) | 2—u(NO3)2(тв) = 2CuO(тв) + 4NO2(г) + ќ2(г) |
|
|
|
12*. Ёнтропи€ системы возрастает в процессах
| 1) | (NH4)2Cr2O7(тв) = Cr2O3(тв) + N2(г) + 4H2O(г) |
| 2) | Fe2O3(тв) + 3Ќ2(г) = 2Fe(тв) + 3Ќ2ќ(ж) |
| 3) | NH4NO2(тв) = N2(г) + 2Ќ2ќ(г) |
| 4) | NH4—l(тв) = NH3(г) + Ќ—l(г) |
| 5) | —аќ(тв) + —ќ2(г) = —а—ќ3(тв) |
| 6) | 2Fe(тв) + 3Cl2(г) = 2FeCl3(тв) |
13*. Ёнтропи€ системы уменьшаетс€ в процессах
| 1) | N2(г) + 3Ќ2(г) = 2NЌ3(г) |
| 2) | 2KClO3(тв) = 2KCl(тв) + 3O2(г) |
| 3) | 4Al(тв) + 3O2(г) = 2Al2O3(тв) |
| 4) | —Ќ4(г) = —(тв) + 2Ќ2(г) |
| 5) | NH4NO3(тв) = N2O(г) + 2Ќ2ќ(г) |
| 6) | Mgќ(тв) + —ќ2(г) = Mg—ќ3(тв) |
14*. Ёнтропи€ системы уменьшаетс€ в процессах
| 1) | 2NЌ3(г) = N2(г) + 3Ќ2(г) |
| 2) | 3—а(тв) + N2(г) = Ca3N2(тв) |
| 3) | 2Al(тв) + 3Cl2(г) = 2AlCl3(тв) |
| 4) | 2KNO3(тв) = 2KNO2(тв) + ќ2(г) |
| 5) | 2KMnO4(тв) = K2MnO4(тв) + MnO2(тв) + O2(г) |
| 6) | CH3NH2(г) + HBr(г) = CH3NH3Br(тв) |






