ƒано:
с 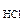 = 0,02 M
с = 0,02 M
с  = 0,05 M
V = 0,05 M
V 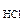 = V = V 
|
| ќбразуетс€ ли осадок-? |
–≈Ў≈Ќ»≈:
ѕри сливании растворов протекает следующа€ реакци€:
Pb(NO3)2 + 2HCl= PbCl2  + 2HNO3
+ 2HNO3
ќсадок PbCl2 образуетс€ только в случае, если
с  Јс
Јс  > ѕ–
> ѕ–  = 2Ј10
= 2Ј10  (табл.)
(табл.)
— учетом увеличени€ объема раствора при сливании в 2 раза:
с  =
=  с
с  Ј
Ј  Јn
Јn  =
=  = 2,5Ј10-2 моль/л
= 2,5Ј10-2 моль/л
с  =
=  с
с 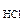 Ј
Ј  Јn
Јn  =
= 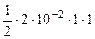 = 1Ј10-2 моль/л
= 1Ј10-2 моль/л
с  Јс
Јс  = 2,5Ј10
= 2,5Ј10 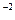 Ј(1Ј10
Ј(1Ј10 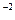 )2 = 2,5Ј10
)2 = 2,5Ј10 
“ак как 2,5Ј10  < 2Ј10-5 (ѕ–
< 2Ј10-5 (ѕ–  ), то осадок PbCl2 не образуетс€.
), то осадок PbCl2 не образуетс€.
ќтвет: осадок PbCl2 не образуетс€.
3. —оставить схему коррозионного √Ё, возникающего при контакте олов€нной пластинки, площадью 35 см2 с медной в растворе хлороводородной кислоты. Ќаписать уравнени€ электродных процессов и суммарной реакции процесса коррозии.
а) ¬ычислить объемный и весовой показатели коррозии, если за 70 минут в процессе коррозии выделилось 1,5 см3 газа (н.у.).
б) ¬ычислить весовой и глубинный показатели коррозии, если за 80 минут потер€ массы корродируемого металла составила 5,6 ∙10-3 г. ѕлотность металла 7,3 г/см3.
–≈Ў≈Ќ»≈:
ѕо таблице находим значени€ стандартных электродных потенциалов олова (II) и меди (II):
 = - 0,14 ¬,
= - 0,14 ¬,  = 0,34 ¬.
= 0,34 ¬.
“ак как  <
<  , то анодом в коррозионном √Ё будет олово, катодом Ц медь.
, то анодом в коррозионном √Ё будет олово, катодом Ц медь.
—оставим схему коррозионного √Ё:
ј(-)Sn │ HCl │ Cu(+)K
или
ј(-)Sn │ H+ │ Cu(+)K
Cоставл€ем уравнени€ электродных процессов и суммарной реакции процесса коррозии:
Ќа A(-)Sn Ц 2ē = Sn2+
Ќа (+)2Ќ+ + 2ē = Ќ2

Sn + 2H+ = Sn2+ + H2
Sn + 2HCl = SnCl2 + H2 Ц суммарна€ реакци€ процесса коррозии.
а)

| –ассчитываем объемный показатель коррозии (KV) по формуле:
KV =   , см3/м2∙час.
ѕри расчете KV принимаем:
S Ц площадь поверхности анода, м2;
τ Ц врем€ процесса коррозии, час; Vгаза Ц объем выделившегос€ газа, см3. , см3/м2∙час.
ѕри расчете KV принимаем:
S Ц площадь поверхности анода, м2;
τ Ц врем€ процесса коррозии, час; Vгаза Ц объем выделившегос€ газа, см3.
|
»з уравнени€ суммарной реакции процесса коррозии следует, что при коррозии выдел€етс€ водород. —ледовательно, Vгаза =  .
.
“огда, KV =  =
=  367,3 см3/м2∙час.
367,3 см3/м2∙час.
–ассчитываем весовой показатель коррозии Km по формуле:
Km =  , г/м2∙час.
, г/м2∙час.
¬ процессе коррозии разрушению подвергаетс€ олово и выдел€етс€ водород.
—ледовательно:
ћэк(ће) = ћэк(Sn) =  =59,5 г/моль,
=59,5 г/моль,
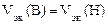 = 11200 см3/моль.
= 11200 см3/моль.
Km =  = 1,95 г/м2∙час.
= 1,95 г/м2∙час.
ќтвет: KV = 367,3 см3/м2∙час.
Km = 1,95 г/м2∙час.
б)

| –ассчитываем весовой показатель коррозии Km по формуле:
Km =  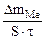 , г/м2∙час.
оррозии подвергаетс€ олово.
“огда потер€ массы металла , г/м2∙час.
оррозии подвергаетс€ олово.
“огда потер€ массы металла
 
|
ѕри расчете Km принимаем:  - [г]; S Ц [м2], τ - [час].
- [г]; S Ц [м2], τ - [час].
“огда: Km = 
 =
=  = 1,2 г/м2∙час.
= 1,2 г/м2∙час.
–ассчитываем глубинный показатель коррозии по формуле:
ѕ = 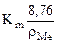 =
=  мм/год.
мм/год.
ќтвет: Km = 1,2 г/м2∙час, ѕ = 1,44 мм/год.
17. ћ≈“јЋЋџ V Ц VI √–”ѕѕџ
|
|
|
”–ќ¬≈Ќ№ ј
1. ћожно ли восстановить оксид хрома (III) до металла при стандартных услови€х:
ј) цинком;
Ѕ) кремнием.
ќтвет подтвердить расчетом  по
по  .
.
| ƒано: Cr2O3; Zn; Si | –≈Ў≈Ќ»≈:
а) —оставл€ем уравнение реакции и рассчитываем стандартное изменение энергии √иббса  вреакции, использу€ I следствие из закона √есса. «начени€ стандартных энергий √иббса образовани€ веществ вреакции, использу€ I следствие из закона √есса. «начени€ стандартных энергий √иббса образовани€ веществ  берем из таблицы
cтандартных термодинамических величин. берем из таблицы
cтандартных термодинамических величин.
|
| Cr -? |
Cr2O3(к) + 3Zn(к) = 2—r(к) + 3ZnO(к) (15.1)
 -1058,97 0 0 3(-320,7)
-1058,97 0 0 3(-320,7)
кƒж/моль
 (15.1) = [
(15.1) = [  (ZnO(к)) + 2
(ZnO(к)) + 2  (—r(к))] Ц
(—r(к))] Ц
- [  (Cr2O3(к)) + 3
(Cr2O3(к)) + 3  (Zn(к))] = 3(-320,7) -
(Zn(к))] = 3(-320,7) -
-(-1058,97) = 96,87 кƒж
“ак как  (15.1) > 0, то при стандартных услови€х реакци€ невозможна.
(15.1) > 0, то при стандартных услови€х реакци€ невозможна.
б) јналогично:
2Cr2O3(к) + 3Si(к) = 4—r(к) + 3Siќ2(к) (15.2)
 2(-1058,97) 0 0 3(-856,7)
2(-1058,97) 0 0 3(-856,7)
кƒж/моль
 (15.2) = [3
(15.2) = [3  (SiO2(к)) + 4
(SiO2(к)) + 4  (Cr(к))] -
(Cr(к))] -
-[2  (Cr2O3(к)) + 3
(Cr2O3(к)) + 3  (Si(к))] = 3(-856,7) Ц
(Si(к))] = 3(-856,7) Ц
-2(-1058,97) = -452,16 кƒж
“ак как  (15.2) < 0, то самопроизвольное протекание реакции при стандартных услови€х возможно.
(15.2) < 0, то самопроизвольное протекание реакции при стандартных услови€х возможно.
ќтвет: ѕри стандартных услови€х возможно восстановление хрома из оксида хрома (III) кремнием.
2. ќпределить рЌ раствора, при котором установилось равновесие: 2CrO  + 2H+ <=> Cr2O
+ 2H+ <=> Cr2O  + H2O –авновесные концентрации ионов Cr2O
+ H2O –авновесные концентрации ионов Cr2O  и CrO
и CrO  соответственно равны 4,5 моль/л и 0,001 моль/л. онстанта равновеси€ с = 1,1Ј1018.
соответственно равны 4,5 моль/л и 0,001 моль/л. онстанта равновеси€ с = 1,1Ј1018.
ƒано:
[Cr2O  ]=4,5 моль/л
[CrO ]=4,5 моль/л
[CrO  ]=0,001 моль/л
с = 1,1Ј1018 ]=0,001 моль/л
с = 1,1Ј1018
|
| рЌ -? |
–≈Ў≈Ќ»≈:
2CrO  + 2H+ <=> Cr2O
+ 2H+ <=> Cr2O  + H2O
+ H2O
рЌ = -lg[H+]
Kc = 
[H+] =  = =
= = 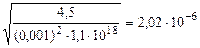 моль/л
моль/л
рЌ = -lg2,02Ј10-6 = 5,7
ќтвет: рЌ = 5,7
3. Ўестивалентный элемент взаимодейству€ с кислородом при н.у., образует оксид, содержащий 20,71% масс. кислорода. ¬ычислить мол€рную массу эквивалента элемента и назвать элемент.

| –≈Ў≈Ќ»≈:
ѕо закону эквивалентов:
nэк(ќ2) = nэк(Ё)
  ћэк(ќ) = 8 г/моль
ћэк(ќ) = 8 г/моль
|
ћэк(Ё) = 
mокс = mэ + 
mэ = 100 -  = 100 Ц 20,71 = 79,29 г.
= 100 Ц 20,71 = 79,29 г.
ћэк(Ё) =  = 30,63 г/моль
= 30,63 г/моль
ћэк(Ё) = 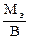 , где ¬ Ц валентность элемента.
, где ¬ Ц валентность элемента.
ћэ = ћэк(Ё)∙¬ = 30,63∙6 = 183,77 г/моль, что соответствует молекул€рной массе атома вольфрама.
ќтвет: ћэк(Ё) = 30,63 г/моль; элемент Ц W.
”–ќ¬≈Ќ№ ¬
1. ”равн€ть реакцию, указать окислитель и восстановитель:
Cr(NO3)3 + Br2 + KOH → K2CrO4 + KBr + KNO3 + H2O ќпределить, какой объем 2 н раствора нитрата хрома (III) расходуетс€ на взаимодействие с бромом массой 4,3 г.
ƒано:
m  = 4,3 г
сэк(Cr(NO3)3)= 2 моль/л = 4,3 г
сэк(Cr(NO3)3)= 2 моль/л
|
| Vр-ра(—r(NO3)3 -? |
–≈Ў≈Ќ»≈:
Cr+3(NO3)3 + Br20 + KOH → → K2Cr+6O4 + KBr-1 + KNO3 + H2O
Ќќ ƒћ

 восст-ль Cr+3 - 3e = Cr+6 2
восст-ль Cr+3 - 3e = Cr+6 2
окисл-ль Br20 + 2e = 2Br- 3
 3Br2 + 2Cr+3 = 6Br- + 2Cr+6
3Br2 + 2Cr+3 = 6Br- + 2Cr+6
ѕереносим найденные коэффициенты в молекул€рное уравнение. ”равниваем число ионов  →6
→6 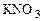 . —читаем число ионов + в продуктах реакции и определ€ем коэффициент перед ќЌ → 16 ќЌ. ”равниваем число атомов Ќ и ќ → 8Ќ2ќ.
. —читаем число ионов + в продуктах реакции и определ€ем коэффициент перед ќЌ → 16 ќЌ. ”равниваем число атомов Ќ и ќ → 8Ќ2ќ.
2Cr(NO3)3 +3Br2 +16KOH = 2K2CrO4 + 6KBr + 6KNO3 + 8H2O
ѕо закону эквивалентов:
nэк(Cr(NO3)3) = nэк(Br2)
Vр-ра(—r(NO3)3 Ј сэк(Cr(NO3)3) = 
Mэк(Br) =  г/моль
г/моль
Vр-ра (—r(NO3)3 Ј 2 =  ; Vр-ра (—r(NO3)3 = 0,0269 л = 26,9 см3
; Vр-ра (—r(NO3)3 = 0,0269 л = 26,9 см3
ќтвет: Vр-ра (—r(NO3)3 = 26,9 см3
2. Ќайти массы воды и кристаллогидрата CrCl3∙6H2O, необходимые дл€ приготовлени€ 1 литра раствора, содержащего 5% (мас.) безводной соли. ѕлотность 5% раствора CrCl3 равна 1,05 г.
|
|
|
ƒано:
Vр-ра = 1 л
ω  = 5%
ρр-ра= 1,05 г/см3 = 5%
ρр-ра= 1,05 г/см3
| –≈Ў≈Ќ»≈
ѕо определению
ω  = =  , %
откуда , %
откуда 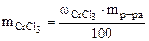 ћасса раствора равна
ћасса раствора равна
|

|

mр-ра = 1Ј103Ј1,05 =1050 г, где 103 коэффициент перевода л в см3. “огда масса CrCl3 в растворе составл€ет:
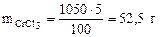
ћасса CrCl3Ј6H2O, в которой содержитс€ 52,5 г CrCl3, может быть найдена по пропорции:
 266 г/моль - 158 г/моль
266 г/моль - 158 г/моль 
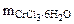 - 52,5 г
- 52,5 г
“огда

«на€ массу раствора и кристаллогидрата, определ€ем массу воды:
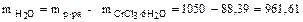 г
г
ќтвет:  г
г
3. Ќаписать уравнени€, при помощи которых можно осуществить следующие превращени€:
K2CrO4 → CrO3 → Cr2O3 → KCrO2 → Cr2(SO4)3
–≈Ў≈Ќ»≈:
1. K2CrO4 + 2H2SO4конц = —rO3 + 2KHSO4 + H2O
t
2. 4—rO3 = 2Cr2O3 + 3O2
t
3. Cr2O3 + 2KOH = 2KCrO2 + H2O
4. 2KCrO2 + 4H2SO4разб = K2SO4 + Cr2(SO4)3 + 4H2O
ћ≈“јЋЋџ VII √–”ѕѕџ
”–ќ¬≈Ќ№ ј
1. ”равн€ть реакцию методом электронного баланса. ”казать окислитель и восстановитель:
FeCl2 + KMnO4 +HCl→FeCl3 + MnCl2+KCl + H2O
| ƒано: ”равнение реакции |
| ”равн€ть реакцию, указать окислитель и восстано- витель |
–≈Ў≈Ќ»≈:
Fe+2Cl2 + KMn+7O4 + HCl → Fe+3Cl3 + +Mn+2Cl2 + KCl + H2O
Ќќ ƒћ

 восст-ль Fe+2 - e = Fe+3 5
восст-ль Fe+2 - e = Fe+3 5
окисл-ль Mn+7 + 5e = Mn+2 1
 |
5Fe+2 + Mn+7 = 5Fe+3 + Mn+2
ѕереносим полученные коэффициенты в молекул€рное уравнение с учетом, что Cl- cодержитс€, также в 5 молекулах FeCl2 и определ€ем необходимое количество молекул HCl дл€ получени€ продуктов реакции. ”равниваем Ќ и ќ → 4Ќ2ќ.
5FeCl2 + KMnO4 + 8HCl = 5FeCl3 + MnCl2 + KCl + 4H2O
”–ќ¬≈Ќ№ ¬
1. ”равн€ть реакцию. ”казать окислитель и восстановитель:
Na2S + KMnO4 + H2O → MnO2 + Na2SO4 + KOH
ќпределить —эк(Na2S), если на взаимодействие с 4 г KMnO4 ушло 250 см3 раствора Na2S.
ƒано:
 4 г 4 г
 = 250 см3 = 0,25 л = 250 см3 = 0,25 л
|
”равн€ть реакцию, указать окислитель и восстановитель

|
–≈Ў≈Ќ»≈:
Na2S-2 + KMn+7O4 + H2O→ Mn+4O2 + Na2S+6O4 + KOH
Ќќ ƒћ

 восст-ль S-2 - 8e = S+6 3
восст-ль S-2 - 8e = S+6 3
окисл-ль Mn+7 + 3e = Mn+4 8
 |
3S-2 + 8Mn+7 = 3S0 + 8Mn+4
ѕереносим полученные коэффициенты в молекул€рное уравнение. ”равниваем число ионов + и определ€ем количество воды → 4Ќ2ќ.
3Na2S +8KMnO4 + 4H2O =8MnO2 +3 Na2SO4 +8KOH
ѕо закону эквивалентов: nэк(Na2S) = nэк(KMnO4)
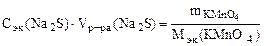 ;
;
 =
=  , г/моль
, г/моль
где ne - число электронов, прин€тых одной молекулой KMnO4.
 г/моль
г/моль
 =
=  моль/л
моль/л
ќтвет:  моль/л.
моль/л.
2. ”равн€ть реакцию. ”казать окислитель и восстановитель:
Mn(OH)4 + HCl → MnCl2 + H2O + Cl2 ќпределить объем газа (н.у.), выдел€ющийс€ при взаимодействии 8 г гидроксида марганца (IV) с хлороводородной кислотой.
ƒано:
m  = 8 г = 8 г
|
V  -? -?
|
–≈Ў≈Ќ»≈:
Mn+4(OH)4 + HCl-1 →Mn+2Cl2 + Cl20 +H2O

 Ќќ ƒћ
Ќќ ƒћ

 восст-ль Cl-1 - e = Cl0 2
восст-ль Cl-1 - e = Cl0 2
окисл-ль Mn+4 + 2e = Mn+2 1

2Cl-1 + Mn+4 = Cl20 + Mn+2
ѕереносим полученные коэффициенты в молекул€рное уравнение, окончательно уравниваем его, учитыва€ при этом количество ионов Cl-1, необходимое дл€ получени€ MnCl2
Mn(OH)4 + 4HCl = MnCl2 + Cl2 + 4H2O
ћ  - Vo
- Vo 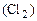
123 г - 22,4 л
8 г - х л
х =  = 1,46 л
= 1,46 л
ќтвет: V  = 1,46 л
= 1,46 л
3. ”равн€ть реакцию. ”казать окислитель и восстановитель:
MnO2 + KClO3 + KOH → K2MnO4 + KCl + H2O. ќпределить массу оксида марганца (IV), необходимую дл€ восстановлени€ 150 см3 1 н раствора K—lO3.
ƒано:
V  =150 см3=
=0,15 л
сэк(KClO3)= 1 моль/л =150 см3=
=0,15 л
сэк(KClO3)= 1 моль/л
|
m  -? -?
|
–≈Ў≈Ќ»≈:
Mn+4O2 + KCl+5O3 + KOH →
→ K2Mn+6O4 + KCl- + H2O
Ќќ ƒћ

 восст-ль Mn+4 - 2e = Mn+6 3
восст-ль Mn+4 - 2e = Mn+6 3
окисл-ль Cl+5 + 6e = Cl- 1
 |
3Mn+4 + Cl+5 = 3Mn+6 + Cl-
ѕереносим полученные коэффициенты в молекул€рное уравнение, уравниваем число +, а затем Ќ и ќ:
3MnO2 + KClO3 + 6KOH = 3K2MnO4 + KCl + 3H2O
|
|
|
ѕо закону эквивалентов:
nэк(KClO3) = nэк(MnO2)
V  Јсэк(KClO3) =
Јсэк(KClO3) = 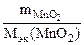
Mэк(MnO2)  г/моль
г/моль
m  = V
= V  Јсэк(KClO3)∙
Јсэк(KClO3)∙ 
m  = 0,15Ј1∙43,5 = 6,5 г
= 0,15Ј1∙43,5 = 6,5 г
ќтвет: m  = 6,5 г
= 6,5 г
ћ≈“јЋЋџ VIII √–”ѕѕџ
”–ќ¬≈Ќ№ ј
1. »спользу€ справочные значени€ изменений стандартных энергий √иббса образовани€ веществ, определить возможность самопроизвольного протекани€ реакции:
Osќ4(к) +4Ќ2(г) = Os(к) + 4Ќ2ќ(ж) при стандартных услови€х. ќтвет мотивировать расчетом ΔrG∞(298K).
| ƒано: ”равнени€ химической реакции. |
 -? -?
|
–≈Ў≈Ќ»≈:
¬озможность самопроизвольного протекани€ реакции при 298 определ€етс€ знаком величины стандартного изменени€ энергии √иббса в реакции:
если  < 0, самопроизвольное протекание реакции при заданных услови€х возможно;
< 0, самопроизвольное протекание реакции при заданных услови€х возможно;
если  > 0, то при заданной температуре реакци€ невозможна..
> 0, то при заданной температуре реакци€ невозможна..
«начение  определ€ем по первому следствию из закона √есса:
определ€ем по первому следствию из закона √есса:
 = [
= [  (Os(к)) + 4
(Os(к)) + 4  (H2O(ж))] Ц
(H2O(ж))] Ц
Ц [  (OsO4(к)) + 4
(OsO4(к)) + 4  (Ќ2(г))]
(Ќ2(г))]
| Osќ4(к) | + 4Ќ2 (г) | = Os(к) | + 4 Ќ2ќ(ж) | |
 кƒж/моль кƒж/моль
| -302,5 | 4(-237,3) |
“огда:
 = 4(-237,3) Ц (-302,5) = -646,7кƒж.
= 4(-237,3) Ц (-302,5) = -646,7кƒж.
ќтвет: так как  < 0, то самопроизвольное протекание реакции возможно.
< 0, то самопроизвольное протекание реакции возможно.
2. ¬ычислить концентрацию ионов железа в 0,01ћ растворе K3[Fe(CN)6], содержащем, кроме того, 0,02 моль/л цианида кали€. онстанта нестойкости иона [Fe(CN)6]3- в водном растворе равна 1.10-31.
ƒано:
с  =0,01 моль/л, сKCN=0,02 моль/л, =0,01 моль/л, сKCN=0,02 моль/л,
 = 1.10-31 = 1.10-31
|
| [Fe3+] -? |
–≈Ў≈Ќ»≈:
ѕервична€ диссоциаци€ комплексных солей протекает по типу диссоциации сильных электролитов:
K3[Fe(CN)6]=[Fe(CN)6]3-+3 + (17.1)
онцентраци€ комплексного иона [Fe(CN)6]3- равна 0,01 моль/л, так как из одной молекулы комплексной соли образуетс€ один комплексный ион.
онстанта нестойкости комплексного иона [Fe(CN)6]3- характеризует следующий равновесный процесс:
[Fe(CN)6]3- <=>Fe3+ + 6CN- (вторична€ диссоциаци€) (17.2)
 (17.3)
(17.3)
ѕрисутствие цианида кали€ в растворе смещает равновесие диссо-циации комплексного иона (17.2) влево вследствие возрастани€ концентрации одноименного иона CN-, образующегос€ при диссоциации KCN:
CN = + + —N-
ѕосле смещени€ равновеси€ устанавливаетс€ новое равновесие. онцентрацию Fe3+ в новом равновесии принимаем равной
х моль/л.
ќбща€ равновесна€ концентраци€ [CN-] равна сумме концентраций CN-, образовавшихс€ при диссоциации CN и [Fe(CN)6]3-:
[CN-] =  +
+ 
из [Fe(CN)6]3- из CN
 = 6х моль/л из [Fe(CN)6]3-
= 6х моль/л из [Fe(CN)6]3-
 =
=  ·α·
·α·  = 0,02Ј1Ј1 = 0,02 моль/л,
= 0,02Ј1Ј1 = 0,02 моль/л,
из CN
так как CN сильный электролит, α=1,  =1.
=1.
“огда:
[CN-] = (6х + 0,02) моль/л.
¬следствие малости х, принимаем: 6х + 0,02 ≈ 0,02 моль/л.
“аким образом, выражение (17.3) примет вид:
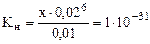 ,
,
откуда х = [Fe3+] =  1,56Ј10-23 моль/л
1,56Ј10-23 моль/л
ќтвет: [Fe3+] = 1,56Ј10-23 моль/л.
3. —оставить схемы электролиза и написать уравнени€ электродных процессов водных растворов солей: а) сульфата кобальта (II), б) хлорида паллади€ (II) (анод инертный). акие продукты выдел€ютс€ на аноде и катоде.

| –≈Ў≈Ќ»≈ а) CoSO4= Co2+ + SO42-, K(-) A(+) инертный Co2+ + 2ē = Co SO42- 2H2O+2ē=H2+2OH- 2H2O-4ē=O2+4H+ |
Ќа катоде выдел€етс€ Co и Ќ2, на аноде выдел€етс€ ќ2.
б) PdCl2 = Pd2+ + 2Cl-
K(-) A(+) инертный
Pd2+ + 2ē = Pd 2Cl - - 2ē = Cl2
H2O H2O
Ќа катоде выдел€етс€ Pd, на аноде выдел€етс€ Cl2.
”–ќ¬≈Ќ№ ¬





 20,71% масс.
¬ = 6
ћэк(Ё) -?
Ё -?
20,71% масс.
¬ = 6
ћэк(Ё) -?
Ё -?

