ѕример 6. ¬ычислить растворимость (моль/л, г/л) оксалата кальци€ —а—2ќ4 в его насыщенном водном растворе, массовую концентрацию ионов —а2+ и массу кальци€ в 100 мл этого раствора.
–ешение. ’имическое равновесие в насыщенном растворе —а—2ќ4 описываетс€ уравнением:
—а—2ќ4↓ D —а2+ + 
«апишем выражение дл€ константы растворимости. ѕоскольку ионна€ сила I Ѓ 0, то можно использовать концентрации, а не активности:
 = [—а2+] Ј [
= [—а2+] Ј [  ]
]
Ќаходим в справочнике [1; 2, табл. 6] значение константы, равное 2,57 ∙ 10Ц9.
ќбозначим растворимость —а—2ќ4 через х моль/л. ѕри диссоциации х молей —а—2ќ4 образуетс€ х молей —а2+ и x молей  . ѕодставл€€ эти значени€ в выражение дл€ константы растворимости, получим:
. ѕодставл€€ эти значени€ в выражение дл€ константы растворимости, получим:
 = [—а2+] Ј [
= [—а2+] Ј [  ] = х ∙ х = 2,57 ∙ 10Ц9
] = х ∙ х = 2,57 ∙ 10Ц9
ќтсюда
x 2 = 2,57 ∙ 10Ц9
x =  = 5,1 ∙ 10Ц5 моль/л.
= 5,1 ∙ 10Ц5 моль/л.
“аким образом, растворимость —а—2ќ4 в его насыщенном водном растворе составл€ет 5,1 ∙ 10Ц5 моль/л.
ƒл€ перехода к другим единицам измерени€ (г/л) необходимо мол€рную растворимость (моль/л) умножить на мол€рную массу —а—2ќ4 (г/моль):

ћол€рна€ концентраци€ ионов —а2+ в насыщенном водном растворе соли —а—2ќ4 така€ же, как мол€рна€ концентраци€ соли:
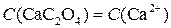
—ледовательно массова€ концентраци€ ионов —а2+ равна:

Ќайдем массу кальци€ в 100 мл насыщенного раствора соли —а—2ќ4:
 »наче:
»наче:
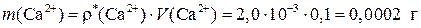
или из пропорции:
в 1000 мл раствора Ц 2,0 ∙ 10Ц3 г кальци€;
в 100 мл раствора Ц x г кальци€;
x = 2,0 ∙ 10Ц3 ∙100 /1000 = 0,0002 г.
–асчет растворимости ћ–— в присутствии разноименных ионов (солевой эффект)
јлгоритм решени€ задач:
1) –ассчитать ионную силу раствора I, которую создает посторонний сильный электролит (разноименные ионы).
2) ƒл€ данной ионной силы вз€ть в справочнике [1; 2, табл. 1, 2] или рассчитать по формулам (1.4), (1.5), (1.6) коэффициенты активности ионов, вход€щих в состав осадка.
3) «аписать равновесие, которое устанавливаетс€ в насыщенном растворе ћ–—, и выражение дл€ термодинамической константы растворимости этого соединени€. ¬з€ть в справочнике [1; 2, табл. 6] численное значение константы.
4) ¬ыразить растворимость ћ–— из выражени€ дл€ термодинамической константы растворимости и провести расчет.
ѕример 7. ¬ычислить растворимость хлорида серебра в 0,01 ћ растворе Ca(NO3)2.
–ешение. –ассчитаем ионную силу в 0,1 моль/л растворе Ca(NO3)2, как описано в примере 1:
I = 0,03 моль/л.
ƒалее найдем коэффициенты активности ионов Ag+ и ClЦ. ѕоскольку в таблицах [1; 2, табл. 1, 2] отсутствует значение ионной силы 0,03 моль/л, то рассчитаем коэффициент активности однозар€дного иона Ag+ по расширенному уравнению ƒеба€Ц’юккел€ (1.5), которое примен€етс€ при ионной силе 0,01 < I < 0,1 моль/л:

ѕо закону ионной силы Ћьюиса-–ендалла коэффициент активности иона ClЦ будет равен коэффициенту активности иона Ag+, поскольку оба иона имеют одинаковый зар€д:

«апишем равновесие, которое устанавливаетс€ в насыщенном растворе AgCl, и выражение дл€ термодинамической константы растворимости AgCl:
|
|
|
AgCl↓ D Ag+ + ClЦ

ќбозначим концентрацию растворенной части хлорида серебра через x моль/л и возьмем из справочника [1; 2, табл. 6] значение константы растворимости AgCl:

—ледовательно, искома€ растворимость AgCl:

ѕример 8. ¬ычислить растворимость арсената бари€ Ba3(AsO4)2 в 0,05 ћ растворе KNO3.
–ешение.
1. –ассчитаем ионную силу в 0,05 ћ растворе KNO3:
KNO3 Ѓ K+ + NO3Ц Þ
— (K+) = C (KNO3) = 0,05 моль/л (α = 1 и n = 1)
C ( ) = C (KNO3) = 0,05 моль/л (α = 1 и n = 1)
) = C (KNO3) = 0,05 моль/л (α = 1 и n = 1)

 Ј (0,05 ∙ 12 + 0,05 ∙ 12) = 0,05 моль/л.
Ј (0,05 ∙ 12 + 0,05 ∙ 12) = 0,05 моль/л.
2. ¬озьмем в таблице [1; 2, табл. 2] коэффициенты активности ионов Ba2+ и AsO43Ц при I = 0,05 моль/л:


3. «апишем равновесие, которое устанавливаетс€ в насыщенном растворе Ba3(AsO4)2, и выражение дл€ термодинамической константы растворимости Ba3(AsO4)2. ¬озьмем в таблице [1; 2, табл. 6] численное значение термодинамической константы растворимости Ba3(AsO4)2, равное 7,8 Ј 10Ц51:
Ba3(AsO4)2↓ D 3Ba2+ + 2AsO43Ц

ќбозначим концентрацию растворенной части Ba3(AsO4)2 через x моль/л. “огда:
[Ba2+] = 3 x моль/л
[AsO43Ц] = 2 x моль/л
ѕодставим эти значени€ в выражение дл€ термодинамической константы растворимости Ba3(AsO4)2:

ѕроведем необходимые вычислени€, выразим и рассчитаем x:
0,125 Ј 0,0441 Ј 27 x 3 Ј 4 x 2 = 7,8 Ј 10Ц51 Þ
x 5 = 1,31 Ј 10Ц50 Þ
 1,1 Ј 10Ц10 моль/л
1,1 Ј 10Ц10 моль/л
“аким образом, мол€рна€ растворимость арсената бари€ Ba3(AsO4)2 в 0,05 ћ растворе KNO3 равна 1,1 Ј 10Ц10 моль/л.






