»о́нное произведе́ние воды́ Ч произведение концентраций ионов водорода Ќ+ и ионов гидроксида OH− в воде или в водных растворах, константа автопротолиза воды. ¬ода, хот€ и €вл€етс€ слабым электролитом, в небольшой степени диссоциирует:
H2O + H2O ↔ H3O+ + OH−или H2O ↔ H+ + OH−
–авновесие этой реакции сильно смещено влево. онстанту диссоциации воды можно вычислить по формуле:
 где:
где:
[H+] Ч концентраци€ ионов гидроксони€ (протонов);
[OH−] Ч концентраци€ гидроксид-ионов;
[H2O] Ч концентраци€ воды (в молекул€рной форме) в воде;
онцентраци€ воды в воде, учитыва€ еЄ малую степень диссоциации, величина практически посто€нна€ и составл€ет (1000 г/л)/(18 г/моль) = 55,56 моль/л.
ѕри 25 ∞C константа диссоциации воды равна 1,8„10−16моль/л. ”равнение (1) можно переписать как:  ќбозначим произведение KЈ[H2O] = Kв = 1,8„10−16 моль/лЈ55,56 моль/л = 10−14моль≤/л≤ = [H+]Ј[OH−] (при 25 ∞C).
ќбозначим произведение KЈ[H2O] = Kв = 1,8„10−16 моль/лЈ55,56 моль/л = 10−14моль≤/л≤ = [H+]Ј[OH−] (при 25 ∞C).
онстанта Kв, равна€ произведению концентраций протонов и гидроксид-ионов, называетс€ ионным произведением воды. ќна €вл€етс€ посто€нной не только дл€ чистой воды, но также и дл€ разбавленных водных растворов веществ. C повышением температуры диссоциаци€ воды увеличиваетс€, следовательно, растЄт и Kв, при понижении температуры Ч наоборот.
ѕрактическое значение ионного произведени€ воды велико, так как оно позвол€ет при известной кислотности (щЄлочности) любого раствора (то есть при известной концентрации [H+] или [OH−]) найти соответственно концентрации [OH−] или [H+]. ’от€ в большинстве случаев дл€ удобства представлени€ пользуютс€ не абсолютными значени€ми концентраций, а вз€тыми с обратными знаком их дес€тичными логарифмами Ч соответственно, водородным показателем (pH) и гидроксильным показателем (pOH).
“ак как Kв Ч константа, при добавлении к раствору кислоты (ионов H+), концентраци€ гидроксид-ионов OH− будет падать и наоборот. ¬ нейтральной среде [H+] = [OH−] =  моль/л. ѕри концентрации [H+] > 10−7 моль/л (соответственно, концентрации [OH−] < 10−7 моль/л) среда будет кислой; ѕри концентрации [OH−] > 10−7 моль/л (соответственно, концентрации [H+] < 10−7 моль/л) Ч щелочной.
моль/л. ѕри концентрации [H+] > 10−7 моль/л (соответственно, концентрации [OH−] < 10−7 моль/л) среда будет кислой; ѕри концентрации [OH−] > 10−7 моль/л (соответственно, концентрации [H+] < 10−7 моль/л) Ч щелочной.
Ёлектролитическа€ диссоциаци€ воды. ¬одородный показатель рЌ
¬ода представл€ет собой слабый амфотерный электролит:
Ќ2ќ Ќ+ + ќЌ-или, более точно:2Ќ2ќ Ќ3ќ+ + ќЌ-
онстанта диссоциации воды при 25о— равна: 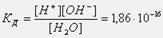 “акое значение константы соответствует диссоциации одной из ста миллионов молекул воды, поэтому концентрацию воды можно считать посто€нной и равной 55,55 моль/л (плотность воды 1000 г/л, масса 1 л 1000 г, количество вещества воды 1000г:18г/моль=55,55 моль, —=55,55 моль: 1 л = 55,55 моль/л). “огда
“акое значение константы соответствует диссоциации одной из ста миллионов молекул воды, поэтому концентрацию воды можно считать посто€нной и равной 55,55 моль/л (плотность воды 1000 г/л, масса 1 л 1000 г, количество вещества воды 1000г:18г/моль=55,55 моль, —=55,55 моль: 1 л = 55,55 моль/л). “огда

Ёта величина посто€нна€ при данной температуре (25о—), она называетс€ ионным произведением воды KW:

ƒиссоциаци€ воды Ц процесс эндотермический, поэтому с повышением температуры в соответствии с принципом Ће-Ўателье диссоциаци€ усиливаетс€, ионное произведение возрастает и достигает при 100о— значени€ 10-13.
|
|
|
¬ чистой воде при 25о— концентрации ионов водорода и гидроксила равны между собой:
[H+] = [OH-] = 10-7 моль/л –астворы, в которых концентрации ионов водорода и гидроксила равны между собой, называютс€ нейтральными. ≈сли к чистой воде прибавить кислоту, концентраци€ ионов водорда повыситс€ и станет больше, чем 10-7 моль/л, среда станет кислой, при этом концентраци€ ионов гидроксила мгновенно изменитс€ так, чтобы ионное произведение воды сохранило свое значение 10-14. “оже самое будет происходить и при добавлении к чистой воде щелочи. онцентрации ионов водорода и гидроксила св€заны между собой через ионное произведение, поэтому, зна€ концентрацию одного из ионов, легко вычислить концентрацию другого. Ќапример, если [H+] = 10-3 моль/л, то [OH-] = KW/[H+] = 10-14/10-3 = 10-11 моль/л, или, если [OH-] = 10-2 моль/л, то [H+] = KW/[OH-] = 10-14/10-2 = 10-12 моль/л. “аким образом, концентраци€ ионов водорода или гидроксила может служить количественной характеристикой кислотности или щелочности среды.
Ќа практике пользуютс€ не концентраци€ми ионов водорода или гидроксила, а водородным рЌ или гидроксильным рќЌ показател€ми. ¬одородный показатель рЌ равен отрицательному дес€тичному логарифму концентрации ионов водорода: рЌ = - lg[H+]
√идроксильный показатель рќЌ равен отрицательному дес€тичному логарифму концентрации ионов гидроксила: рќЌ = - lg[OH-]
Ћегко показать, прологарифмировав ионное произведение воды, что рЌ + рќЌ = 14
≈сли рЌ среды равен 7 - среда нейтральна€, если меньше 7 - кисла€, причем чем меньше рЌ, тем выше концентраци€ ионов водорода. pЌ больше 7 Ц среда щелочна€, чем больше рЌ, тем выше концентраци€ ионов гидроксила. „иста€ вода очень плохо проводит электрический ток, но всЄ же обладает измеримой электропроводностью, котора€ объ€сн€етс€ небольшой диссоциацией воды на ионы водорода и гидроксид-ионы. ѕо величине электропроводности чистой воды можно определить концентрацию ионов водорода и гидроксид-ионов в воде.
ѕоскольку степень диссоциации воды очень мала, то концентраци€ недиссоциированных молекул в воде практически равна общей концентрации воды, поэтому из выражени€ дл€ константы диссоциации воды получакм, что дл€ воды и разбавленных водных растворов при неизменной температуре произведение концентраций ионов водорода и гидроксид-ионов есть величина посто€нна€. Ёта посто€нна€ величина называетс€ ионным произведением воды.
–астворы, в которых концентрации ионов водорода и гидроксид-ионов одинаковы, называютс€ нейтральными. ¬ кисдых растворах больше ионов водорода, в щелочных - гидроксид-ионов. Ќо произведение их концентраций всегда посто€нно. Ёто означает, что если известна концентраци€ ионов водорода в водном растворе, то тем самым и определена и концентраци€ гидроксид-ионов. ѕоэтому как степень кислотности, так и степень щЄлочности раствора можно количественно охарактеризовать концентрацией ионов водорода.






