| –ечовина | 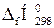 , , 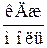
| 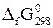 , , 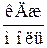
| 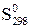 , , 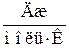
|
| Al2O3(корунд) | Ц1676,0 | Ц1582,0 | 50,92 |
| Al2S3(к) | Ц509,0 | Ц23,0 | 96,20 |
| Al(OH)3(к) | Ц1315,0 | Ц1157,0 | 71,10 |
| AlCl3(к) | Ц 704,6 | Ц 629,0 | 109,29 |
| AgCl (к) | Ц 126,78 | Ц 109,54 | 96,23 |
| BaCO3(к) | Ц 1217 | Ц 1137,2 | 112,13 |
| BaO(к) | Ц 553.9 | Ц 525,4 | 70,30 |
| CH4(г) | Ц 74,85 | Ц 50,85 | 186,27 |
| C2H2(г) | 226,75 | 209,21 | 200,82 |
| C2H4(г) | 52,30 | 68,14 | 219,45 |
| C2H6(г) | Ц 84,67 | Ц 32,93 | 229,49 |
| CH3COOH(р) | Ц 484,09 | Ц 389,36 | 159,83 |
| C2H5OH(г) | Ц 234,80 | Ц 167,96 | 281,38 |
| C2H5OH(р) | Ц 276,98 | Ц 174,15 | 160,67 |
| C6H6(р) | 49,03 | 124,38 | 173,26 |
| —6H12ќ6(к) | Ц 1273 | Ц 919,5 | 212,13 |
| CCl4(р) | Ц 132,84 | Ц 62,66 | 216,19 |
| CaBr2(к) | Ц 674,9 | Ц 656,1 | 130,00 |
| CaC2(к) | Ц 59,9 | Ц 64,9 | 69,96 |
| CaCl2(к) | Ц 796,3 | Ц 748,9 | 108,37 |
| CO(г) | Ц110,5 | Ц137,1 | 197,55 |
| CO2(г) | Ц393,5 | Ц394,4 | 213,66 |
| —аќ(к) | Ц635,5 | Ц604,2 | 38,07 |
| CaCO3(к) | Ц1207 | Ц1129,6 | 91,71 |
| Ca(OH)2(к) | Ц 985,12 | Ц 897,52 | 83,39 |
| Ca3(PO4)2(к) | Ц 4123,6 | Ц 3887,6 | 235,98 |
| CuO(к) | Ц162,0 | Ц129,9 | 42,63 |
| Cu(NO3)2(к) | Ц 307,11 | Ц 114,22 | 191,30 |
| CuSO4(к) | Ц 770,9 | Ц 661,8 | 109,20 |
| CrO3 (к) | Ц 590,36 | Ц 513,44 | 73,22 |
| Cr2O3 (к) | Ц 1140,56 | Ц 1058,97 | 81,17 |
| FeCl3 (к) | Ц 399,70 | Ц 334,20 | 130,10 |
| FeO (к) | Ц 264,85 | Ц 244,30 | 60,75 |
| Fe2O3 (к) | Ц 822,16 | Ц 740,34 | 87,45 |
| Fe3O4 (к) | Ц 1117,13 | Ц 1014,17 | 146,19 |
| HBr (г) | Ц 36,38 | Ц 53,43 | 198,58 |
| HCl (г) | Ц 92,31 | Ц 95,30 | 186,79 |
| HF (г) | Ц 273,30 | Ц 275,41 | 173,67 |
| –ечовина | 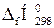 , , 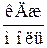
| 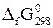 , , 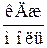
| 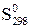 , , 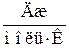
|
| HI (г) | 26,36 | 1,58 | 206,48 |
| H2O (г) | Ц 241,8 | Ц 228,61 | 188,72 |
| H2O (р) | Ц 286,0 | Ц 237,23 | 69,95 |
| H2O2 (р) | Ц 187,86 | Ц 120,52 | 109,60 |
| H2S(г) | Ц21,0 | Ц33,8 | 205,70 |
| KBr(к) | Ц 392,2 | Ц 379,2 | 95,94 |
| KCl (к) | Ц 436,68 | Ц 408,93 | 82,55 |
| KClO3(к) | Ц 391,20 | Ц 289,80 | 142,97 |
| KNO3(к) | Ц 492,46 | Ц 392,75 | 132,88 |
| MgCl2(к) | Ц 644,80 | Ц 595,30 | 89,54 |
| MgO (к) | Ц 601,8 | Ц 569,6 | 27,07 |
| NH3 (г) | Ц 45,94 | Ц 16,48 | 192,66 |
| NH4Cl (к) | Ц 314,22 | Ц 203,22 | 95,81 |
| NH4NO3(к) | Ц 365,43 | Ц 183,93 | 151,04 |
| (NH4)2SO4 | Ц 1180,0 | Ц 901,3 | 220,08 |
| NO(г) | 90,3 | 80,6 | 210,64 |
| NO2(г) | 33,0 | 51,5 | 240,06 |
| NiO (к) | Ц 239,7 | Ц 211,6 | 37,99 |
| NaBr (к) | Ц 361,41 | Ц 349,34 | 86,82 |
| NaCl (к) | Ц 411,12 | Ц 384,13 | 72,13 |
| Na2CO3(к) | Ц 1130,80 | Ц 1048,20 | 138,80 |
| NaNO3(к) | Ц 466,70 | Ц 365,97 | 116,50 |
| P2O5 (к) | Ц 1507,20 | Ц 1371,7 | 140,30 |
| Pb(NO3)2(к) | Ц 451,70 | Ц 258,90 | 217,90 |
| –bќ (черв.) | Ц219,3 | Ц189,1 | 66,11 |
| –bO2 (к) | Ц276,6 | Ц218,3 | 71,92 |
| SO2 (г) | Ц 296,90 | Ц 300,21 | 248,07 |
| SO3 (г) | Ц 395,85 | Ц 371,17 | 256,69 |
| SnO2(к) | Ц 580,8 | Ц 519,3 | 52,30 |
| TiO2 (к) | Ц 943,9 | Ц 888,6 | 50,33 |
| ZnO (к) | Ц 348,11 | Ц 320,7 | 43,51 |
| ZnS(к) | Ц 205,6 | Ц 200,9 | 57,66 |
ѕрим≥тка. «наченн€ 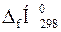 ≥
≥ 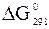 дл€ простих речовин дор≥внюють нулю;
дл€ простих речовин дор≥внюють нулю;
|
|
|
к Ц кристал≥чний, г Ц газопод≥бний, р Ц р≥дкий.
ƒодаток 2
ƒобуток розчинност≥ при 25 0— (n Ц к≥льк≥сть йон≥в)
| —полука | ƒ–, (г-йон/л)n | —полука | ƒ–, (г-йон/л)n | —полука | ƒ–, (г-йон/л)n |
| AgBr AgCN AgCl AgI AgBrO3 Ag2CrO4 Ag2SO4 Al(OH)3 Ag2S BaSO3 BaSO4 | 5,0Ј10-13 1,6Ј10-14 1,7Ј10-10 8,1Ј10-17 6,0Ј10-5 4,4Ј10-12 1,2Ј10-5 4,0Ј10-33 2,5Ј10-25 9,5Ј10-10 1,5Ј10-9 | Ca(OH)2 CaSO4 Cd(OH)2 Co(OH)2 CuCl Cu≤ Fe(OH)2 Hg2Br2 Hg2Cl2 Hg2≤2 HgS | 6,0Ј10-6 2,4Ј10-5 1,7Ј10-14 2,5Ј10-16 3,2Ј10-7 1,1Ј10-12 1,4Ј10-15 4,0Ј10-23 1,0Ј10-18 4,0Ј10-29 1,6Ј10-52 | Hg2SO4 Ni(OH)2 PbBr2 Pb—l2 Pb≤2 Pb(OH)2 PbSO4 TIBr TICl Zn(OH)2 ZnS | 6,2Ј10-7 1,3Ј10-16 4,5Ј10-6 1,5Ј10-5 8Ј10-9 5Ј10-16 1,6Ј10-8 3,6Ј10-6 1,8Ј10-4 4,3Ј10-17 1,3Ј10-23 |
ƒодаток 3
–озчинн≥сть кислот, основ ≥ солей у вод≥
| јн≥они | ат≥они | ||||||||||||||
| 
| 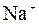
| 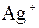
| 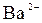
| 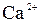
| 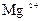
| 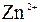
| 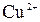
| 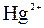
| 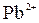
| 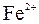
| 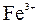
| 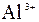
| 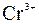
| |
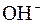
| – | – | Ц | – | ћ | Ќ | Ќ | Ќ | Ц | Ќ | Ќ | Ќ | Ќ | Ќ | |
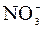
| – | – | – | – | – | – | – | – | – | – | – | – | – | – | – |
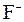
| – | – | – | – | ћ | Ќ | Ќ | – | – | Ц | Ќ | Ќ | Ќ | ћ | Ќ |
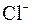
| – | – | – | Ќ | – | – | – | – | – | – | ћ | – | – | – | – |
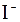
| – | – | – | Ќ | – | – | – | – | – | Ќ | Ќ | – | ? | – | ? |
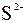
| – | – | – | Ќ | Ц | Ц | Ц | Ќ | Ќ | Ќ | Ќ | Ќ | Ц | Ц | Ц |
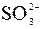
| – | – | – | Ќ | Ќ | Ќ | ћ | ћ | ? | Ќ | Ќ | Ќ | ? | ? | Ц |
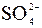
| – | – | – | ћ | Ќ | ћ | – | – | – | Ц | Ќ | – | – | – | – |
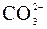
| – | – | – | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | ? | Ќ | Ќ | ? | ? | ? |
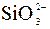
| Ќ | – | – | ? | Ќ | Ќ | Ќ | Ќ | ? | ? | Ќ | Ќ | ? | ? | ? |
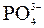
| – | – | – | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ | Ќ |
ѕрим≥тка: – Ц розчин€Їтьс€, M Ц мало розчин€Їтьс€, Ќ Ц практично не розчин€Їтьс€, риска Ц сполука розкладаЇтьс€ водою або не ≥снуЇ,? Ц немаЇ достов≥рних в≥домостей про ≥снуванн€ сполуки
ƒодаток 4
«наченн€ ебул≥оскоп≥чних та кр≥оскоп≥чних констант
ƒе€ких розчинник≥в
| –озчинник | K≈ | Kкр | –озчинник | K≈ | Kкр | –озчинник | K≈ | Kкр |
| вода | 0,52 | 1,86 | фенол | 3,6 | 7,39 | “етрахлорометан (——l4) | 5,3 | 2,98 |
| бензол | 2,57 | 5,1 | ан≥л≥н | 2,6 | 5,87 | ацетатна кислота | 3,1 | 3,9 |
| ацетон | 1,48 | 2,4 | етанол | 1,16 | Ц | н≥тробензен | 5,27 | 6,90 |
| хлороформ | 3,88 | 4,9 | с≥рковуглець | 2,37 | Ц |
ƒодаток 5
—тандартн≥ електродн≥ потенц≥али метал≥в у водних розчинах
| ≈лектродна пара | φ0, ¬ | ≈лектродна пара | φ0, ¬ | ≈лектродна пара | φ0, ¬ |
| Li+/Li | Ц3,045 | Ti2+/Ti | Ц1,630 | Sn2+/Sn | Ц0,136 |
| Rb+/Rb | Ц2,925 | Ti3+/Ti | Ц1,208 | Pb2+/Pb | Ц0,126 |
| K+/K | Ц2,924 | Mn2+/Mn | Ц1,192 | Fe3+/Fe | Ц0,037 |
| Cs+/Cs | Ц2,923 | V2+/V | Ц1,175 | 2H+/H2 | 0,000 |
| Ra2+/Ra | Ц2,916 | Nb3+/Nb | Ц1,100 | Sb3+/Sb | +0,240 |
| Ba2+/Ba | Ц2,905 | Cr2+/Cr | Ц0,852 | Bi3+/Bi | +0,317 |
| Sr2+/Sr | Ц2,888 | Zn2+/Zn | Ц0,763 | Co3+/Co | +0,330 |
| Ca2+/Ca | Ц2,864 | Cr3+/Cr | Ц0,744 | Cu2+/Cu | +0,345 |
| Na+/Na | Ц2,711 | Ga3+/Ga | Ц0,560 | Cu+/Cu | +0,520 |
| Ac3+/Ac | Ц2,600 | Fe2+/Fe | Ц0,440 | Hg22+/2Hg | +0,850 |
| La3+/La | Ц2,522 | Cd2+/Cd | Ц0,403 | Ag+/Ag | +0,799 |
| Ce3+/Ce | Ц2,483 | In3+/In | Ц0,338 | Pb4+/Pb | +0,840 |
| Y3+/Y | Ц2,372 | Ti3+/Ti | Ц0,368 | Hg2+/Hg | +0,852 |
| Mg2+/Mg | Ц2,363 | Tl+/Tl | Ц0,336 | Pd2+/Pd | +0,915 |
| Be2+/Be | Ц1,847 | Co2+/Co | Ц0,277 | Pt2+/Pt | +0,963 |
| U3+/U | Ц1,798 | Ni2+/Ni | Ц0,234 | Au3+/Au | +1,498 |
| Al3+/Al | Ц1,663 | Mo3+/Mo | Ц0,200 | Au+/Au | +1,691 |
|
|
|
ƒодаток 6
—тандартн≥ окисно-в≥дновн≥ потенц≥али де€ких
ќкисно-в≥дновних систем у водних розчинах
| —истема | φ0 , ¬ |
AgCl + ē = Ag + Cl 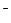
| + 0,222 |
Ag  + ē = Ag + ē = Ag
| + 0,800 |
As 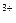 +3ē = As +3ē = As
| + 0,300 |
Br  + 2ē = 2Br + 2ē = 2Br 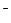
| + 1,087 |
BrO 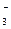 + 3H + 3H  O + 6ē = Br O + 6ē = Br  + 6OH + 6OH 
| + 0,610 |
Cl  + 2ē = 2Cl + 2ē = 2Cl 
| + 1,359 |
2HClO + 2H  + 2ē = Cl + 2ē = Cl  + 2H + 2H  O O
| + 1,630 |
ClO  + 6H + 6H  + 6ē = Cl + 6ē = Cl  + 3H + 3H  O O
| + 1,450 |
ClO  + 3H + 3H  O + 6ē = Cl O + 6ē = Cl  + 6OHЦ + 6OHЦ
| + 0,630 |
CrO  + 4H + 4H  O + 3ē = Cr O + 3ē = Cr 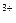 + 8OH + 8OH 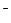
| Ц 0,125 |
CrO 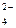 + 4H + 4H  + 3ē = CrO + 3ē = CrO 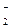 + 2H2O + 2H2O
| + 0,945 |
Cr2O 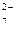 + 14H + 14H  + 6ē = 2Cr + 6ē = 2Cr 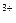 + 7H2O + 7H2O
| + 1,333 |
Cu 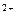 + ē = Cu + ē = Cu 
| + 0,158 |
F  + 2ē = 2F + 2ē = 2F 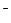
| + 2,870 |
Fe 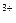 + ē = Fe + ē = Fe 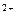
| + 0,771 |
Fe(CN) 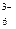 + ē = Fe(CN) + ē = Fe(CN) 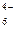
| + 0,356 |
FeO 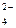 + 8H + 8H  + 3ē = Fe + 3ē = Fe 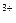 + 4H + 4H  O O
| + 1,700 |
2H  + 2ē = H + 2ē = H 
| + 0,000 |
H  + 2ē = 2H + 2ē = 2H 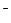
| Ц 2,251 |
H  O O  Ц 2ē = O Ц 2ē = O  + 2H + 2H 
| Ц 0,682 |
H  O O  + 2ē = 2OH + 2ē = 2OH 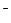
| + 0,880 |
H  O O  + 2H + 2H  + 2ē = 2H + 2ē = 2H  O O
| + 1,776 |
Hg  + 2ē = Hg + 2ē = Hg
| +0,854 |
I  + 2ē = 2I + 2ē = 2I 
| + 0,536 |
HIO + H  + 2ē = I + 2ē = I  + H + H  O O
| + 0,990 |
2IO  + 12H + 12H  + 10ē = I + 10ē = I  + 6H + 6H  O O
| + 1,190 |
IO 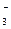 + 3H + 3H  O + 6ē = I O + 6ē = I 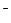 + 6OH + 6OH 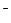
| + 0,260 |
MnO  + 4H + 4H  + 2ē = Mn + 2ē = Mn 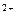 + 2H + 2H  O O
| + 1,228 |
MnO 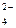 + 4H + 4H  + 2ē = MnO + 2ē = MnO  + 2H + 2H  O O
| + 2,257 |
MnO 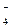 + ē = MnO + ē = MnO 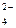
| + 0,564 |
| —истема | φ0 , ¬ |
MnO 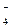 + 2H + 2H  O + 3ē = MnO O + 3ē = MnO  + 4OH + 4OH 
| + 0,600 |
MnO  + 8H + 8H  + 5ē = Mn + 5ē = Mn  + 4H + 4H  O O
| + 1,507 |
MnO  + 4H + 4H  + 3ē = MnO + 3ē = MnO  + 2H + 2H  O O
| + 1,692 |
HNO  + H + H  + ē = NO + H + ē = NO + H  O O
| + 1,004 |
NO 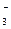 + 2H + 2H  + 2ē = NO + 2ē = NO 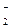 + H + H  O O
| + 0,838 |
NO 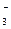 + 2H + 2H  + ē = NO + ē = NO  + H + H  O O
| + 0,772 |
NO 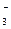 + 4H + 4H  + 3ē = NO + 2H + 3ē = NO + 2H  O O
| + 0,957 |
2NO 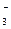 + 12H + 12H  + 10ē = N + 10ē = N  + 6H + 6H  O O
| + 1,24 |
NO 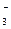 + 10H + 10H  + 8ē = NH + 8ē = NH 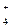 + 3H + 3H  O O
| + 0,880 |
Ni(OH)  + ē = Ni(OH) + ē = Ni(OH)  + OH + OH 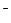
| + 0,49 |
O  + 2H + 2H  + 2ē = O + 2ē = O  + H + H  O O
| + 2,075 |
O  + 2H + 2H  O + 4ē = 4OH O + 4ē = 4OH 
| + 0,401 |
P + 3H  O + 3ē = PH O + 3ē = PH  + 3OH + 3OH 
| Ц 0,890 |
H  PO PO  + 2H + 2H  + 2ē = H + 2ē = H  PO PO  + H + H  O O
| Ц 0,276 |
Pb  + 2ē = Pb + 2ē = Pb 
| + 1,694 |
Pb 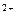 + 2ē = Pb + 2ē = Pb
| Ц 0,130 |
PbO  + 4H + 4H  + 2ē = Pb + 2ē = Pb 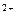 + 2H + 2H  O O
| + 1,455 |
PbO  + SO + SO 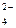 + 4H+ + 2ē = PbSO + 4H+ + 2ē = PbSO  + 2H + 2H  O O
| + 1,685 |
PtCl 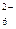 + 2ē = PtCl + 2ē = PtCl 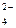 + 2ClЦ + 2ClЦ
| + 0,720 |
S + 2ē = S 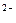
| + 0,480 |
SO 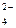 + H + H  O + 2ē = SO O + 2ē = SO 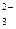 + 2OH + 2OH 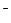
| Ц 0,932 |
SO 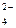 + 4H + 4H  + 2ē = H + 2ē = H  SO SO  + H + H  O O
| + 0,172 |
SO 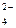 + 8H + 8H  + 6ē = S + 4H + 6ē = S + 4H  O O
| + 0,357 |
SO 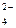 + 8H + 8H  + 8ē = S + 8ē = S 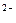 + 4H + 4H  O O
| + 0,149 |
SO 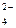 + 10H + 10H  + 8ē = H + 8ē = H  S + 4H S + 4H  O O
| + 0,31 |
S 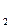 O O 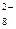 + 2ē = 2SO + 2ē = 2SO 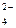
| + 2,010 |
Sn 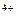 + 2ē = Sn + 2ē = Sn 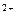
| + 1,151 |
Tl 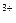 + 2ē = Tl + 2ē = Tl 
| + 1,280 |
V 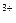 + ē = V + ē = V 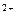
| Ц 0,255 |
Sn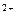 + 2ē = Sn + 2ē = Sn
| Ц 0,14 |
«ћ≤—“
| ѕравила роботи в х≥м≥чн≥й лаборатор≥њЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.......... | |
| –екомендована л≥тература....................................................................................... | |
| ћодуль 1. ќсновн≥ пон€тт€ х≥м≥њ та вченн€ про будову речовини............... | |
| ћодуль 2. «аконом≥рност≥ переб≥гу х≥м≥чних процес≥в.................................... | |
| Ћабораторна робота є 1. ¬изначенн€ теплового ефекту реакц≥њ....................... | |
| Ћабораторна робота є 2........................................................................................ ƒосл≥ди до теми: Ўвидк≥сть х≥м≥чних реакц≥й. атал≥з................................ ƒосл≥ди до теми: ’≥м≥чна р≥вновага................................................................. | |
| ћодуль 3. –озчини................................................................................................. | |
| Ћабораторна робота є 3........................................................................................ ƒосл≥ди до теми: ≈лектрол≥тична дисоц≥ац≥€.................................................................... ƒосл≥ди до тем: …онн≥ реакц≥њ в розчинах електрол≥т≥в. ƒобуток розчинност≥ малорозчинних речовин..................................................................... ƒосл≥ди до тем: …онний добуток води. ¬одневий показник €к к≥льк≥сна характеристика кислотност≥-основност≥ розчин≥в. √≥дрол≥з солей..................... ƒосл≥ди до теми: “верд≥сть води та способи њњ усуненн€.............................. | |
| ћодуль 4. ќкисно-в≥дновн≥ реакц≥њ та електрох≥м≥чн≥ процеси.................... | |
| Ћабораторна робота є 4. ќкисно-в≥дновн≥ реакц≥њ.............................................. | |
| Ћабораторна робота є 5. √альван≥чн≥ елементи................................................... | |
| Ћабораторна робота є 6. ≈лектрол≥з розчин≥в. јкумул€тори........................... | |
| Ћабораторна робота є 7. ороз≥€ метал≥в. ћетоди захисту в≥д короз≥њ............. | |
| ћодуль 5. онструкц≥йн≥ матер≥али................................................................ | |
| Ћабораторна робота є 8......................................................................................... ƒосл≥ди до теми: Ћегк≥ конструкц≥йн≥ метали (алюм≥н≥й, магн≥й, титан)..... ƒосл≥ди до теми: онструкц≥йн≥ метали Ц зал≥зо, кобальт, н≥кель................ ƒосл≥ди до теми: онструкц≥йн≥ метали Ц м≥дь, хром.................................... | |
| ƒодатки..................................................................................................................... |
|
|
|






