ѕ≈–≈ƒћќ¬ј
–обочий зошит призначений дл€ самоп≥дготовки, проведенн€ та оформленн€ лабораторних зан€ть з неорган≥чноњ х≥м≥њ дл€ студент≥в заочноњ форми навчанн€ ≤ курсу фармацевтичних факультет≥в вищих фармацевтичних та медичних заклад≥в осв≥ти IIIЦIV р≥вн≥в акредитац≥њ.
«м≥ст методичних вказ≥вок, що вход€ть до даного робочого зошита, в≥дпов≥даЇ програм≥ з неорган≥чноњ х≥м≥њ, затверджен≥й ћ≥н≥стерством охорони здоровТ€ ”крањни.
–обочий зошит складаЇтьс€ з двох частин. ѕерша м≥стить загальнотеоретичн≥ питанн€ та лабораторн≥ роботи з основних розд≥л≥в загальноњ х≥м≥њ. ƒруга частина присв€чена вивченню х≥м≥чних властивостей елемент≥в та њх сполук, особливо тих, €к≥ використовуютьс€ у фармацевтичн≥й практиц≥. ожний розд≥л вм≥щуЇ еталони та завданн€ дл€ виконанн€ контрольних роб≥т є1 та є2.
¬—“”ѕ
‘ундаментальна х≥м≥чна п≥дготовка Ч основа профес≥йноњ д≥€льност≥ пров≥зора. 10% л≥карських препарат≥в, з €кими пров≥зор стикаЇтьс€ в повс€кденн≥й робот≥, це сполуки неорган≥чного походженн€ р≥зного складу й будови (90% Ч це речовини орган≥чноњ природи), а б≥льш≥сть метод≥в, €к≥ використовуютьс€ у фармацевтичн≥й практиц≥, Ї х≥м≥чними й ф≥зикоЦх≥м≥чними.
Ќа фармацевтичному факультет≥ студент вивчаЇ в≥с≥м х≥м≥чних дисципл≥н. Ѕазовою Ї неорган≥чна х≥м≥€. √оловною метою викладанн€ курсу неорган≥чноњ х≥м≥њ Ї формуванн€ вих≥дного р≥вн€ знань студент≥в, необх≥дного дл€ усп≥шного вивченн€ наступних х≥м≥чних та спец≥альних дисципл≥н, прищепленн€ студентам навичок х≥м≥чного мисленн€, ум≥нн€ передбачати можлив≥сть взаЇмод≥њ речовин та характер продукт≥в њх х≥м≥чного перетворенн€. ¬иконанн€ лабораторного практикуму спри€Ї поглибленню та засвоЇнню теоретичного матер≥алу курсу та формуванню експериментальних навичок самост≥йноњ роботи.
Ќеорган≥чна х≥м≥€ необх≥дна студенту фармацевтичного вузу дл€ освоЇнн€: орган≥чноњ, анал≥тичноњ, ф≥зичноњ, колоњдноњ х≥м≥њ; медикоЦб≥олог≥чних наук Ч патоф≥з≥олог≥њ, б≥ох≥м≥њ, ф≥з≥олог≥њ, фармаколог≥њ; проф≥льних дисципл≥н Ч фармацевтичноњ х≥м≥њ, токсиколог≥чноњ х≥м≥њ, аптечноњ ≥ заводськоњ технолог≥њ л≥к≥в, фармакогноз≥њ.
≥нцева мета вивченн€ неорган≥чноњ х≥м≥њ:
сформувати у студент≥в ц≥л≥сну у€ву про х≥м≥ю €к науку ≥ њњ значенн€ в фармацевтичн≥й галуз≥;
навчити студент≥в загальним принципам п≥дходу до оц≥нки реакц≥йноњ здатност≥ ≥ розум≥нню механ≥зм≥в реакц≥й, покладених в основу синтезу ≥ анал≥зу неорган≥чних речовин;
розкрити прикладн≥ аспекти сучасного стану неорган≥чноњ х≥м≥њ, шл€хи ≥ методи використанн€ њњ дос€гнень у фармацевтичн≥й практиц≥.
ќЅ—я√ ”–—” Ч 306 г: лекц≥њ Ч 16 г, лабораторн≥ зан€тт€ Ч 32 г.
” семестрах I ≥ I≤ програмою передбачена перев≥рка знань у вигл€д≥ двох контрольних роб≥т. ѕо зак≥нченню вивченн€ неорган≥чноњ х≥м≥њ буде проведений перев≥дний ≥спит з усього курсу. ¬≥н вм≥щуЇ в себе:
1. “еоретичн≥ знанн€: основних питань теоретичноњ неорган≥чноњ х≥м≥њ; номенклатури, способ≥в добуванн€ ≥ реакц≥йноњ здатност≥ неорган≥чних сполук.
2. «д≥йсненн€ схем х≥м≥чних перетворень.
|
|
|
3. ѕрактичн≥ навички: €к≥сн≥ реакц≥њ на кат≥они та ан≥они неорган≥чноњ природи; визначенн€ ф≥зичних констант неорган≥чних сполук; очищенн€ р≥дких або кристал≥чних речовин ≥ з п≥дтвердженн€м њхньоњ чистоти.
“≈ћј“»„Ќ»… ѕЋјЌ Ћ≈ ÷≤…
≤ семестр
| є | “ема та зм≥ст лекц≥њ |
| 1. | ¬ступ до вивченн€ неорган≥чноњ х≥м≥њ. ласи неорган≥чних сполук. |
| 2. | ≈нергетика та напр€мок х≥м≥чних реакц≥й. ’≥м≥чна к≥нетика та р≥вновага х≥м≥чних реакц≥й. атал≥з. |
| 3. | –озчини. —пособи вираженн€ концентрац≥њ розчин≥в. ¬ластивост≥ розчин≥в електрол≥т≥в. ƒобуток розчинност≥. рЌ розчин≥в. √≥дрол≥з солей. |
| 5. | ќкисноЦв≥дновн≥ реакц≥њ. ≈лектрол≥з. |
“≈ћј“»„Ќ»… ѕЋјЌ ЋјЅќ–ј“ќ–Ќ»’ «јЌя“№
≤ семестр
| є | “ема та зм≥ст зан€тт€ |
| 1. | ¬ступ. ласи та номенклатура неорган≥чних сполук. ќсновн≥ способи очищенн€ неорган≥чних речовин. |
| 2. | ќсновн≥ закони х≥м≥њ. «акон екв≥валент≥в. –озрахунки за х≥м≥чними р≥вн€нн€ми. ¬изначенн€ екв≥валентноњ маси металу методом вит≥сненн€. |
| 3. | ќсновн≥ теоретичн≥ положенн€ про будову атом≥в. —учасне тлумаченн€ пер≥одичного закону ƒ. I. ћенделЇЇва на основ≥ електронноњ теор≥њ атома. “еор≥€ х≥м≥чного звТ€зку. Ѕудова молекул. |
| 4. | ≈нергетика та напр€мок х≥м≥чних реакц≥й. ’≥м≥чна к≥нетика та р≥вновага х≥м≥чних реакц≥й. атал≥з. |
| 5. | –озчини. —пособи вираженн€ концентрац≥њ розчин≥в. ол≥гативн≥ властивост≥ розчин≥в. ќсмос. |
| 6. | “еор≥€ електрол≥тичноњ дисоц≥ац≥њ. –≥вновага в розчинах слабких електрол≥т≥в. ƒобуток розчинност≥. “еор≥€ сильних електрол≥т≥в. ƒисоц≥ац≥€ води. рЌ розчин≥в. |
| 7. | Ѕуферн≥ системи. √≥дрол≥з солей. |
| 8. | ќкисноЦв≥дновн≥ реакц≥њ. |
–озд≥л 1
¬ступ.
јтомноЦмолекул€рне вченн€. ќсновн≥ пон€тт€ та закони х≥м≥њ. —тех≥ометричн≥ розрахунки.
≈талони розвТ€занн€ типових задач
«адача 1. ƒо розчину що м≥стить н≥трат ср≥бла масою 25,5 г долили розчин що м≥стить сульф≥д натр≥ю масою 7,8 г. яка маса осаду утворитьс€ при цьому?
–озвТ€занн€:
2AgNO3 + Na2S = Ag2Si + 2NaNO3
n (AgNO3) = 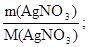 n (AgNO3) =
n (AgNO3) = 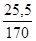 моль = 0,15 моль;
моль = 0,15 моль;
n (Na2S) = 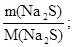 n (Na2S) =
n (Na2S) = 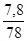 моль = 0,1 моль.
моль = 0,1 моль.
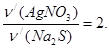
якщо n/(AgNO3) = n(AgNO3); n/(AgNO3) = 0,15 моль, то n/(Na2S) = 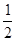 n/(AgNO3);
n/(AgNO3);
n/(Na2S) = 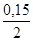 моль = 0,075 моль;
моль = 0,075 моль;

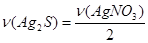 ;
; 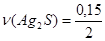 моль = 0,075 моль
моль = 0,075 моль
m(Ag2S) = n(Ag2S)·M(Ag2S); m(Ag2S) = 0,075·248 г = 18,6 г.
«адача 2. —ум≥ш м≥дних ≥ магн≥Ївих ошурок масою 1,5 г обробили надлишком сол€ноњ кислоти. ¬ результат≥ реакц≥њ вид≥ливс€ водень обТЇмом 560 мл (нормальн≥ умови). ¬изначте масову частку м≥д≥ у сум≥ш≥.
–озвТ€занн€:
Mg + 2HCl = MgCl2 + H2
n(H2) = 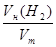 ; n(H2) =
; n(H2) = 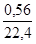 моль = 0,025 моль.
моль = 0,025 моль.
n(Mg) = n(H2); n(Mg) = 0,025 моль
m(Mg) = n(Mg)·M(Mg); m(Mg) = 0,025·24 г = 0,6 г.
m(Cu) = m(сум≥ш≥) Ц m(Mg); m(Cu) = (1,5 Ц 0,6) г = 0,9 г.
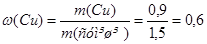 або 60%.
або 60%.
«адача 3. ¬изначте масову частку сол≥ що отримали при зм≥шуванн≥ розчину обТЇмом 40 мл з масовою часткою азотноњ кислоти 0,2 ≥ густиною 1,12 г/мл з розчином обТЇмом 36 мл з масовою часткою г≥дроксиду натр≥ю 0,15 ≥ густиною1,17 г/мл.
–озвТ€занн€:
HNO3 + NaOH = NaNO3 + H2O
m1 = V1r1; m1 = 40·1,12 г = 44,8 г;
m(HNO3) = m1ω(HNO3); m(HNO3) = 44,8·0,2 г = 8,96 г;
ν(HNO3) =  ν(HNO3) =
ν(HNO3) = 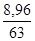 моль = 0,142 моль.
моль = 0,142 моль.
m2 = V2r2; m2 = 36·1,12 г = 42,1 г;
m(NaOH) = m2ω(NaOH); m(NaOH) = 42,1·0,15 г = 6,32 г;
|
|
|
ν(NaOH) = 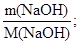 ν(NaOH) =
ν(NaOH) = 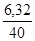 моль = 0,158 моль.
моль = 0,158 моль.
ν(NaNO3) = ν(ЌNќ3); ν(NaNO3) = 0,142 моль.
m(NaNO3) = ν(NaNO3) ·M(NaNO3); m(NaNO3) = 0,142·85 г = 12,1 г.
m3 = m1+m2; m3 = (44,8 + 42,1) г = 86,9 г.
ω(NaNO3) = 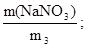 ω(NaNO3) =
ω(NaNO3) = 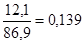 або 13,9%.
або 13,9%.
«адача 4. Ќа реакц≥ю з≥ зразком техн≥чного сульф≥ту натр≥ю масою 9 г витратили розчин масою 40 г з масовою часткою перманганату кал≥ю (KMnO4) 7,9%. ¬изначте масову частку Na2SO3 в техн≥чному сульф≥т≥. –еакц≥€ м≥ж перманганатом кал≥ю ≥ сульф≥том натр≥ю проходить в присутност≥ с≥рчаноњ кислоти.
–озвТ€занн€:
5Na2SO3 + 2KMnO4 + 3H2SO4 = 5Na2SO4 + 2MnSO4 + K2SO4 + 3H2O
| Mn+7 + 5е " Mn+2 | ||
| S+4 Ц 2e " N+6 |
m(KMnO4) = m(рЦну)·ω(KMnO4); m = 40·0,079 г = 3,16 г;
ν(KMnO4) = 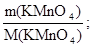 ν(KMnO4) =
ν(KMnO4) = 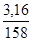 моль = 0,02 моль.
моль = 0,02 моль.
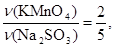 зв≥дки:
зв≥дки:
ν(Na2SO3) = 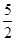 ν(KMnO4); ν(Na2SO3) =
ν(KMnO4); ν(Na2SO3) = 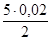 моль = 0,05 моль.
моль = 0,05 моль.
m(Na2SO3) = ν(Na2SO3)·M(Na2SO3); m(Na2SO3) = 0,05·126 г = 6,3 г.
ω(Na2SO3) = 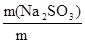 ; ω(Na2SO3) =
; ω(Na2SO3) = 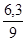 = 0,7 або 70%.
= 0,7 або 70%.
«адача 5. ƒо розчину, в €кому знаходитьс€ н≥трат алюм≥н≥ю масою 42,6 г, додали розчин що м≥стить карбонат натр≥ю масою 37,2 г. ќсад прокалили. ¬изначте масу залишку п≥сл€ прокалюванн€.
–озвТ€занн€:
2Al(NO3)3 + 3Na2CO3 + 3H2O = 2Al(OH)3i + 6NaNO3 + 3CO2h (а)
2Al(OH)3 = Al2O3 + 3H2O (б)
ν(Al(NO3)3) = 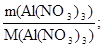 ν(Al(NO3)3) =
ν(Al(NO3)3) = 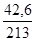 моль = 0,2 моль;
моль = 0,2 моль;
ν(Na2CO3) = 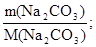 ν(Na2CO3) =
ν(Na2CO3) = 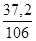 моль = 0,35 моль.
моль = 0,35 моль.
« р≥вн€нн€ (а) вит≥каЇ:
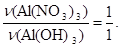
« р≥вн€нн€ (б) вит≥каЇ:
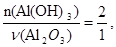
тому:
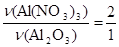 .
.
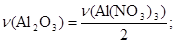
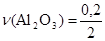 моль = 0,1 моль.
моль = 0,1 моль.
m(Al2O3) = ν(Al2O3)·M(Al2O3); m(Al2O3) = 0,1·102 г = 10,2 г.
–озд≥л 2
’≥м≥чна термодинам≥ка. ≥нетика та катал≥з.
≈талони розвТ€занн€ типових задач
«адача 1. –озрахувати тепловий ефект реакц≥њ: 2NaOH(к.) + CO2(г.) 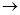 Na2CO3(к.) + H2O, €кщо:
Na2CO3(к.) + H2O, €кщо:  Ќоутв.(NaOH) = Ц426,6 кƒж/моль,
Ќоутв.(NaOH) = Ц426,6 кƒж/моль,  Ќоутв.(CO2) = Ц393,6 кƒж/моль,
Ќоутв.(CO2) = Ц393,6 кƒж/моль,  Ќоутв.(Na2CO3) = = Ц1129,3 кƒж/моль,
Ќоутв.(Na2CO3) = = Ц1129,3 кƒж/моль,  Ќоутв.(H2O) = Ц285,8 кƒж/моль.
Ќоутв.(H2O) = Ц285,8 кƒж/моль.
–озвТ€занн€:
“епловий ефект х≥м≥чноњ реакц≥њ обчислюЇтьс€ по стандартних теплотах утворенн€ за формулою:
 Ќо =
Ќо = 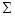
 Ќоутв.(прод. рЦц≥њ) Ц
Ќоутв.(прод. рЦц≥њ) Ц 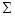
 Ќоутв.(вих. рЦн)
Ќоутв.(вих. рЦн)
з врахуванн€м в≥дпов≥дних стех≥ометричних коеф≥ц≥Їнт≥в, тобто:
 Ќо = (
Ќо = ( Ќоутв.(Na2CO3) +
Ќоутв.(Na2CO3) +  Ќоутв.(H2O)) Ц (2
Ќоутв.(H2O)) Ц (2  Ќоутв.(NaOH) +
Ќоутв.(NaOH) +  Ќоутв.(CO2)) = (Ц1129,3 Ц 285,8) Ц (Ц2·426,6 Ц 393,6) = Ц168,3 кƒж.
Ќоутв.(CO2)) = (Ц1129,3 Ц 285,8) Ц (Ц2·426,6 Ц 393,6) = Ц168,3 кƒж.
“обто це екзотерм≥чна реакц≥€.
«адача 2. „и Ї можливою реакц≥€: SiO2(к.) + 2NaOH(р.) 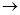 Na2SiO3(к.) + H2O(р.), €кщо:
Na2SiO3(к.) + H2O(р.), €кщо:  Gо(SiO2(к.)) = Ц803,75 кƒж/моль,
Gо(SiO2(к.)) = Ц803,75 кƒж/моль,  Gо(NaOH(р.)) = Ц419,5 кƒж/моль,
Gо(NaOH(р.)) = Ц419,5 кƒж/моль,  Gо(Na2SiO3(к.)) = = Ц1427,8 кƒж/моль,
Gо(Na2SiO3(к.)) = = Ц1427,8 кƒж/моль,  Gо(H2O(р.)) = Ц237,5 кƒж/моль?
Gо(H2O(р.)) = Ц237,5 кƒж/моль?
–озвТ€занн€:
ƒл€ в≥дпов≥д≥ на питанн€ задач≥ необх≥дно обчислити зм≥ну енерг≥њ √≥ббса в дан≥й реакц≥њ за формулою:
 Gо =
Gо = 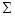
 Gо(прод. рЦц≥њ) Ц
Gо(прод. рЦц≥њ) Ц 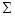
 Gо(вих. рЦн) (з врахуванн€м в≥дпов≥дних стех≥ометричних коеф≥ц≥Їнт≥в) = (Ц1427,8 Ц 237,5) Ц (Ц803,75 Ц 419,5·2) = Ц22,55 кƒж, тобто
Gо(вих. рЦн) (з врахуванн€м в≥дпов≥дних стех≥ометричних коеф≥ц≥Їнт≥в) = (Ц1427,8 Ц 237,5) Ц (Ц803,75 Ц 419,5·2) = Ц22,55 кƒж, тобто  Gо < 0, таким чином реакц≥€ Ї можливою ≥ розчин лугу не можна випарювати в скл€ному посуд≥.
Gо < 0, таким чином реакц≥€ Ї можливою ≥ розчин лугу не можна випарювати в скл€ному посуд≥.
«адача 3. ѕитома теплота топленн€ свинцю ( Ќ) становить 23,4, ƒж/кг. “емпература топленн€ свинцю 327,4о—. «найти зм≥ну ентроп≥њ при топленн≥ 250 г свинцю.
Ќ) становить 23,4, ƒж/кг. “емпература топленн€ свинцю 327,4о—. «найти зм≥ну ентроп≥њ при топленн≥ 250 г свинцю.
–озвТ€занн€: «астосовуючи питому теплоту топленн€, обчислюЇмо теплоту топленн€ 0,25кг свинцю:
| 1кг Pb | Ч 23040 ƒж | х = 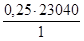 = 5760 ƒж = 5760 ƒж
|
| 0,25кг Pb | Ч х |
‘ормула дл€  Gо =
Gо =  Ќ Ц T
Ќ Ц T  S. “опленн€ Ї р≥вноважним процесом, тому дл€ нього
S. “опленн€ Ї р≥вноважним процесом, тому дл€ нього  Gо=0, тод≥:
Gо=0, тод≥:
 Ќ = T
Ќ = T  S;
S;  S =
S = 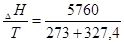 = 9,59 ƒж/ .
= 9,59 ƒж/ .
«адача 4. ѕри 80о— реакц≥€ зак≥нчуЇтьс€ за 8 хв. „ерез ск≥льки хвилин вона зак≥нчитьс€ при 100о—, коли температурний коеф≥ц≥Їнт (g) дор≥внюЇ 2?
–озвТ€занн€: «а правилом ¬анЦ√оффа при зб≥льшен≥ температури швидк≥сть реакц≥њ зб≥льшитьс€ в:
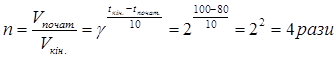
при цьому ≥нтервал переб≥гу реакц≥њ (Dtк≥н.) зменшитьс€ в 4 рази:
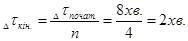
«адача 5. ¬ синтез≥ ам≥аку (N2 + 3H2 = 2NH3) р≥вновага встановилась при наступних концентрац≥€х: [N2] = 2,5моль/л, [H2] = 1,8моль/л, [NH3] = 3,6моль/л. –озрахуйте константу р≥вноваги ц≥Їњ реакц≥њ ≥ вих≥дн≥ концентрац≥њ азоту та водню.
|
|
|
–озвТ€занн€: онстанта х≥м≥чноњ р≥вноваги за р≥вноважними концентрац≥€ми обчислюЇтьс€ за формулою:
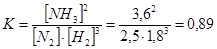
«а р≥вн€нн€м реакц≥њ:
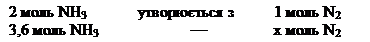
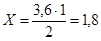 моль (N2(прореаг.) Ч —(прореаг.))
моль (N2(прореаг.) Ч —(прореаг.))
—(вих≥дне) = —(прореаг.) + [—(р≥вноважна)] = 1,8 + 2,5 = 4,3 моль/л
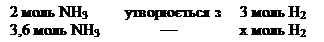 «а р≥вн€нн€м реакц≥њ:
«а р≥вн€нн€м реакц≥њ:
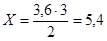 моль (Ќ2(прореаг.) Ч —(прореаг.))
моль (Ќ2(прореаг.) Ч —(прореаг.))
—(вих≥дна) = 1,8 + 5,4 = 7,2моль/л
«адача 6. –еакц≥€ ≥де за р≥вн€нн€м: 4HCl + O2 = 2H2O + 2Cl2. ¬ €кому напр€мку зм≥ститьс€ х≥м≥чна р≥вновага, €кщо концентрац≥њ ус≥х реагуючих речовин зб≥льшити в 2 рази?
–озвТ€занн€: «а законом д≥ючих мас швидк≥сть пр€моњ ≥ оберненоњ реакц≥њ дор≥внюють:
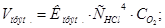
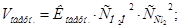
ѕри зб≥льшен≥ концентрац≥њ ус≥х речовин в 2 рази:
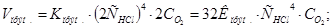
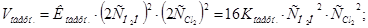
Ўвидк≥сть пр€моњ реакц≥њ зросла в 32 рази, а швидк≥сть оберненоњ Ч т≥льки в 16 раз≥в, отже х≥м≥чна р≥вновага зм≥ститьс€ в напр€мку пр€моњ реакц≥њ.
«адача 7. –≥вновага х≥м≥чноњ реакц≥њ: 2NO + O2 = 2NO2 встановилась при сл≥дуючи концентрац≥€х реагуючих речовин: [NO] = 0,5моль/л, [O2] = 0,7моль/л, [NO2] = 2,1моль/л. як зм≥нитьс€ швидк≥сть обох реакц≥й, €кщо тиск в систем≥ зменшити в 2 рази? уди зм≥ститьс€ х≥м≥чна р≥вновага?
–озвТ€занн€: ƒо зменшенн€ тиску:
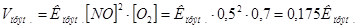
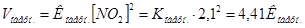
ѕри зменшен≥ тиску в 2 рази концентрац≥њ ус≥х речовин зменшатьс€ в 2 рази ≥ тод≥:
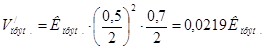
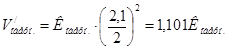
Ўвидк≥сть пр€моњ реакц≥њ зменшилась в:
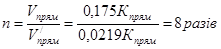 , а оберненоњ в:
, а оберненоњ в:
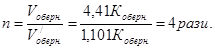
Ўвидк≥сть оберненоњ реакц≥њ буде б≥льше, тому х≥м≥чна р≥вновага реакц≥њ зм≥ститьс€ в напр€мку оберненоњ реакц≥њ.
–озд≥л 3
Ѕудова речовини. Ѕудова атома та його електронних оболонок. ѕер≥одичний закон ƒ.≤.ћенделеЇва та його тлумаченн€ на основ≥ електронноњ будови атом≥в. ’≥м≥чний звТ€зок та будова молекул
≈талони розвТ€занн€ типових задач
«адача 1. «образити електронноЦграф≥чн≥ формули (орб≥тальн≥ структури) атома хлору в нормальному та збуджених станах. ¬изначити можлив≥ валентност≥ та ступен≥ окисленн€. Ќаписати формули можливих сполук з оксигеном та г≥дрогеном.
¬≥дпов≥дь:
1s2 2s22p6 3s23p5
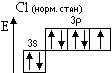
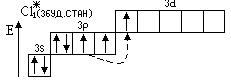
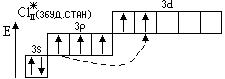
¬алентн≥сть = ≤ ¬алентн≥сть = ≤≤≤ ¬алентн≥сть = V
—т. окисл. =  1 —т. окисл. = +3 —т. окисл. = +5
1 —т. окисл. = +3 —т. окисл. = +5
Ц1 +1 +3 +5
HCl; —l2O (—l2O3) (Cl2O5)
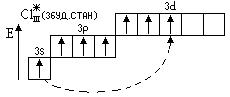
¬алентн≥сть = VII
—т. окисл. = +7
+7
Cl2O7
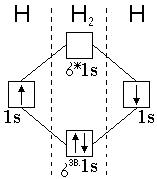
«авданн€ 2. Ќапиш≥ть енергетичну схему заповненн€ електронами ћќ в молекул≥ водню.
–озвТ€занн€:
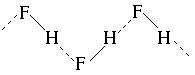
«авданн€ 3. ѕоказати водневий звТ€зок м≥ж молекулами фтороводню.
–озвТ€занн€:
–озд≥л 4
¬ченн€ про розчини —пособи вираженн€ концентрац≥њ розчин≥в. ол≥гативн≥ властивост≥ розчин≥в. ќсмос.
≈талони розвТ€занн€ типових задач
“аблиц€ звТ€зку вираженн€ р≥зних способ≥в концентрац≥й
| ћетод оц≥нки вм≥сту розчиненоњ речовини | w | —м, моль/л | —н, моль/л | “, г/мл |
| ћасова частка, w | Ч | 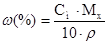
| 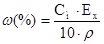
| 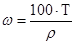
|
| ћол€рна концентрац≥€, —м | 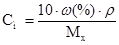
| Ч | 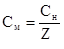
| 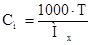
|
| Ќормальн≥сть, Cн | 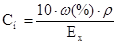
| 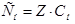
| Ч | 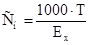
|
| “итр, “х | 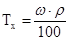
| 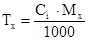
| 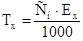
| Ч |
«адача 1. —к≥льки грам борноњ; кислоти та води потр≥бно дл€ приготуванн€ 250г розчину з масовою часткою борноњ кислоти 3%?
–озвТ€занн€: ‘ормула розрахунку масовоњ частки:
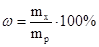
«в≥дси:
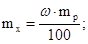
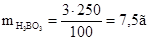
¬оди потр≥бно вз€ти: 250 Ц 7,5 = 242,5г
«адача 2. —к≥льки грам≥в хлориду натр≥ю потр≥бно дл€ приготуванн€ 1л розчину з —ћ = 2моль/л?
–озвТ€занн€: ‘ормула розрахунку мол€рноњ концентрац≥њ:
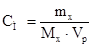 , зв≥дки: mx = Cћ·Mx·Vp = 2·58,5·1 = 117г
, зв≥дки: mx = Cћ·Mx·Vp = 2·58,5·1 = 117г
«адача 3. —к≥льки грам≥в ћnќ4 необх≥дно дл€ приготуванн€ 2л розчину з —н = 0,5 моль/л, €кщо анал≥з проводитьс€ в кислому середовищ≥?
–озвТ€занн€:
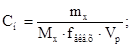 зв≥дки: mx = Cн·Mx·fекв.х·Vp = 0,5·158·
зв≥дки: mx = Cн·Mx·fекв.х·Vp = 0,5·158· 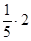 = 31,6г.
= 31,6г.
«адача 4. –озрахувати мол€льну концентрац≥ю розчину, приготовленого з 2г ќЌ ≥ 200г води.
–озвТ€занн€: ‘ормула розрахунку мол€льноњ концентрац≥њ:
|
|
|
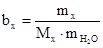 ; зв≥дки:
; зв≥дки: 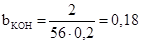 моль/кг.
моль/кг.
«адача 5. –озрахувати титр розчину с≥рчаноњ кислоти з w = 50% ≥ r = 1,4г/мл.
–озвТ€занн€: ‘ормула розрахунку титра розчину:
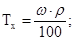 зв≥дки:
зв≥дки: 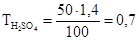 г/мл.
г/мл.
«адача 6. «найти мол€рну концентрац≥ю екв≥валента розчину с≥рчаноњ кислоти з масовою часткою 10% (густина 1,22, fекв. =1/2).
–озвТ€занн€: ‘ормула переходу в≥д масовоњ частки до мол€рной концентрац≥њ екв≥валента:
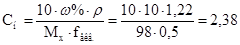 (моль/л).
(моль/л).
«адача 7 яка масова частка у в≥дсотках сульфату зал≥за (II) у насиченому при 80 ∞— розчин≥, €кщо розчинн≥сть сол≥ при ц≥й температур≥ дор≥внюЇ 100 г.
–озвТ€занн€:
оеф≥ц≥Їнт розчинност≥ (розчинн≥сть) виражаЇ к≥льк≥сть грам≥в розчиненоњ речовини, що м≥ститьс€ у 100 г розчинника (Ќ2ќ).
1. «находимо масу розчину:
m(розчину) = m(речовини) + m(розчинника)
m(розчину) = 100 г + 100 г = 200 г
2. ќбчислюЇмо масову частку розчиненоњ речовини у в≥дсотках:
200 г розчину Ч 100 г FеSќ4
100 г розчину Ч X г FеSќ4
X = 50 г
¬≥дпов≥дь: масова частка сульфату зал≥за (II) дор≥внюЇ 50 %.
«адача 8. ќбчислити обТЇм розчину з масовою часткою с≥рчаноњ кислоти 56% (r = 1,460 г/мл) та обТЇм води, €к≥ необх≥дн≥ дл€ приготуванн€ 100мл розчину з масовою часткою с≥рчаноњ кислоти 20% (r = 1,143 г/мл).
–озвТ€зок: ѕри зм≥шуванн≥ розчин≥в користуютьс€ Дправилом хрестаФ або Дд≥агональною схемоюФ. ¬ середин≥ хреста записують масову частку розчину (w3), €кий необх≥дно приготувати. «л≥ва записують концентрац≥њ w1 ≥ w2. —права записують р≥зницю м≥ж w3 ≥ w2, w3 ≥ w1.
—хема маЇ загальний вигл€д:
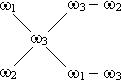
| ¬ наведеному приклад≥:
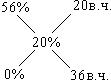
|
«г≥дно з≥ схемою на 20 в.ч. розчину з w = 5б%, необх≥дно вз€ти 36 в.ч. води, а маса розчину складаЇ:
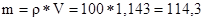 г,
г,
що зг≥дно з≥ схемою складаЇ 20+36=56 в.ч.
ћасу с≥рчаноњ кислоти, €ку потр≥бно вз€ти дл€ приготуванн€ розчину, можна знайти за пропорц≥Їю:
| 114,3г 20%Цго рЦну | складаЇ | 56 в.ч. |
| ’г | Ч | 20 в.ч. |
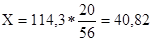 г
г
а обТЇм отриманого розчину: 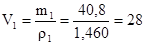 (мл)
(мл)
ƒл€ приготуванн€ розчину необх≥дно води: 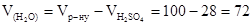 мл
мл
ѕриготуванн€ розчину розведенн€м б≥льш концентрованого розчину.
«адача 9. —к≥льки розчину сол≥ з масовою часткою 18% необх≥дно додати до 46г води, щоб утворивс€ 15% розчин.
–озвТ€зок:
| 0,18х = | (46 + х) 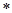 0,15 0,15
|
| х = |
ƒл€ приготуванн€ 15% розчину з w = 18% необх≥дно вз€ти 230г 18%Цного розчину ≥ 46г води.
«адача 10. Ќа нейтрал≥зац≥ю 25 мл розчину H2SO4 витрачено 22,5 мл 0,152 н розчину NаќЌ ќбчислити нормальн≥сть ≥ титр розчину кислоти.
–озвТ€занн€:
«г≥дно з законом екв≥валент≥в:
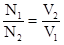 ;
; 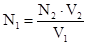 ;
; 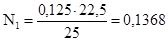 моль/л
моль/л
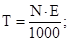
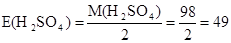 г/моль
г/моль
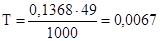 г/мл
г/мл
¬≥дпов≥дь: нормальн≥сть 0,1368 моль/л; титр 0,0067 г/мл.
«адача 11. –озрахувати –осм. розчину хлориду натр≥ю з масовою часткою 5,85% при 0∞—. —туп≥нь дисоц≥ац≥њ хлориду натр≥ю 0,96, а r = 1,04г/мл.
–озвТ€зок: ѕереведемо масову частку в мол€рну концентрац≥ю.
 моль/л
моль/л
–озрахуЇмо ≥зотон≥чний коеф≥ц≥Їнт:
≥ = 1 + a(n Ц 1) = 1 + 0,96(2 Ц 1) = 1,96
ќсмотичний тиск розраховуЇмо за формулою дл€ електрол≥т≥в:
–осм. = iCRT = 1,96 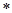 1моль/л
1моль/л 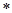 0,082атм/моль*
0,082атм/моль* 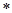 273 = 4,36атм
273 = 4,36атм
«адача 12. –озрахувати –осм. розчину глюкози з масовою часткою 5%, €кщо t=27ќ—, r=1г/мл.
–озвТ€зок: ѕереведемо масову частку в мол€рну концентрац≥ю.
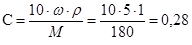 моль/л.
моль/л.
осмотичний тиск обчислюЇмо за формулою неелектрол≥т≥в:
–осм. = C R T
Pосм. = 0,28 * 0,082 * (273+27) = 6,8атм
«адача 13. –озрахувати осмотичну концентрац≥ю кров≥. јбо: –озрахувати мол€рну концентрац≥ю водного розчину глюкози, €кий Ї ≥зотон≥чним кров≥.
–озвТ€зок: 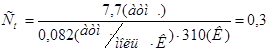 моль/л
моль/л
«адача 14. онстанта дисоц≥ац≥њ бромнуватистоњ кислоти (Ќ¬rќ) дор≥внюЇ 2,≤ 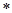 10Ц9. ќбчисл≥ть њњ ступ≥нь дисоц≥ац≥њ в 0,01ћ розчин≥.
10Ц9. ќбчисл≥ть њњ ступ≥нь дисоц≥ац≥њ в 0,01ћ розчин≥.
–озвТ€зок: « закону розведенн€ ќствальда:
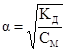 ; тод≥
; тод≥ 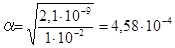 ; a = 4,58·10-4, або a = 0,046%.
; a = 4,58·10-4, або a = 0,046%.
«адача 15. ƒобуток розчинност≥ хромату ср≥бла Ag2CrO4 дор≥внюЇ 1,1·10Ц12. ќбчисл≥ть розчинн≥сть ц≥Їњ сол≥ в моль/л та г/л.
–озвТ€занн€:
«аписують р≥вн€нн€ електрол≥тичноњ дисоц≥ац≥њ хромату ср≥бла:
Ag2CrO4 D 2Ag+ + CrO 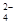
та вираз його добутку розчинност≥:
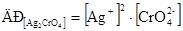
ѕозначають розчинн≥сть Ag2CrO4 через S моль/л хроматЦ≥он≥в та 2S моль/л кат≥он≥в ср≥бла. ÷≥ значенн€ концентрац≥њ ≥он≥в п≥дставл€ють у вираз добутку розчинност≥ Ag2CrO4:
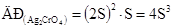
ќбчислюють розчинн≥сть сол≥ в моль/л:
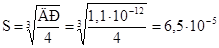 моль/л.
моль/л.
ќск≥льки р≥вноважна концентрац≥€ хроматЦ≥он≥в у розчин≥ чисельно дор≥внюЇ розчинност≥ хромату ср≥бла, то розчинн≥сть Ag2CrO4 дор≥внюЇ 6,5·10Ц5 моль/л. ѕеремножуючи розчинн≥сть сол≥ на њњ мол€рну масу, обчислюють розчинн≥сть хромату ср≥бла в г/л:
(Ag2CrO4) = 6,5·10Ц5·331,8 = 2,2·10Ц2г/л.
Ч в розчинах сильних електрол≥т≥в:
Ч кислот:
pH = Цlg[H+] = Цlg—н(кЦти) = ЦlgZ·Cм(кЦти)
—н(кЦти) = antilg pH = 10ЦpH; —м = 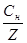 .
.
|
|
|
Ч луг≥в:
рќЌ = Цlg[OHЦ] = ЦlgCн(лугу) = ЦlgZ·Cм(лугу)
рЌ = 14 Ц рќЌ
—н(лугу) = antilg pOH = 10ЦpOH; —м = 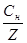 .
.
Ч в розчинах слабких електрол≥т≥в:
Ч кислот: (дисоц≥ац≥€ переважно ≥де по ≤ ступеню)
pH = Цlg[H+] = Цlga·—м(кЦти) = Цlg 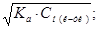
Cм(кЦти) = 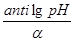 =
= 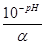 ; —н = Z·Cм
; —н = Z·Cм
Ч основ: (дисоц≥ац≥€ ≥де по ≤ ступеню)
рќЌ = Цlg[OHЦ] = Цlga·—м(основи) = Цlg 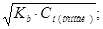
Cм(основи) = 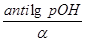 ; —н = Z·Cм
; —н = Z·Cм
«адача 16. ќбчислити рЌ розчину з концентрац≥Їю ≥он≥в водню 4,2·10Ц5моль/л.
–озвТ€занн€:
[H+] = 4,2·10Ц5; рЌ = Цlg[H+] = Цlg4,2·10Ц5 = 5Цlg4,2 = 4,38
«адача 17. ќбчислити рЌ розчину сол€ноњ кислоти з —н = 0,15 моль/л ≥ розчину г≥дроксиду натр≥ю з —н = 0,2 моль/л, a = 1.
–озвТ€занн€:
1) рЌ = Цlg[H+]; [H+] = a·—кЦти = 1·0,15 = 0,15моль/л; рЌ = Цlg1,5·10Ц1 = 1Цlg1,5 = 0,824
2) рЌ + рќЌ = 14; рЌ = 14ЦрќЌ; [OHЦ] = a·Cосн. = 1·0,2 = 0,2моль/л; рќЌ = Цlg[ќHЦ] =
= Цlg0,2 = 0,7; рЌ = 14Ц0,7 = 13,3.
«адача 18. ќбчислити рЌ розчину мурашиноњ кислоти з —н = 0,1 моль/л, g = 1,8·10Ц4.
–озвТ€занн€:
рЌ = Цlg[H+]; [H+] = a·C; g = a2·C; a= 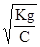 ; [H+] =
; [H+] = 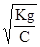 ·C =
·C = 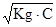 ;
;
pH = Цlg 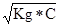 = Цlg
= Цlg 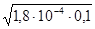 = Цlg4,24·10Ц3 = 2,37.
= Цlg4,24·10Ц3 = 2,37.
«адача 19. як зм≥нитьс€ рЌ води, €кщо 80 мл њњ долити до 20 мл розчину г≥дроксиду натр≥ю з —н = 0,1 моль/л, a = 1.
–озвТ€занн€:
рЌ(води) = 7. ѕ≥сл€ розведенн€ V2 = 80 + 20 = 100мл
V1·C1 = V2·C2; C2 = 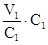 =
= 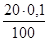 = 0,02; pOH = Цlg[OHЦ];
= 0,02; pOH = Цlg[OHЦ];
[OHЦ] = a·C = 1·0,2 = 2·10Ц2, pOH = Цlg2·10Ц2 = 1,7;
pH = 14 Ц 1,7 = 12,3; DpH = 12,3 Ц 7 = 5,30.
«адача 20. ќбчислити [Ќ+] кров≥, €кщо рЌ = 7,36.
–озвТ€занн€:
«наход€ть антилогарифм числа 7,36; [Ќ+] = 4,4·10Ц8моль/л.
–озрахунки рЌ в розчинах солей.
Ч с≥ль утворена сильною кислотою та слабкою основою (NH4Cl, Fe(NO3)3)
| Kb | Kb: д1, д2, д3. оли основа багатокислотна, то дл€ визначенн€ рЌ середньоњ сол≥ беретьс€ останн€ д ( д3) |
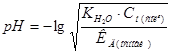 ;
;
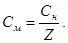
Ч с≥ль утворена сильною основою та слабкою кислотою (K2CO3, Na3PO4)
| а: д1, д2 | д1, д2, д3. оли слабка кислота буде багатоосновною, то дл€ розрахунку рќЌ середньоњ сол≥ беруть останню д (дл€ K2CO3 Ч д2) |
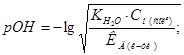
pH = 14 Ц pOH
«адача 21. ќбчисл≥ть рЌ 0,5Ќ розчину карбонату натр≥ю, €кщо 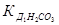 = 4,5·10Ц7,
= 4,5·10Ц7, 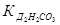 = 4,7·10Ц11.
= 4,7·10Ц11.
–озвТ€занн€:
арбонат натр≥ю Ч це с≥ль, утворена слабкою двоосновною кислотою, тому на перш≥й стад≥њ г≥дрол≥зу утворюЇтьс€ кисла с≥ль та сильна основа. ѕодальшому переб≥гу г≥дрол≥зу перешкоджаЇ накопиченн€ в розчин≥ г≥дроксидЦ≥он≥в. «аписують ≥онн≥ та молекул€рн≥ р≥вн€нн€ реакц≥њ г≥дрол≥зу:
CO32Ц + H2O D HCO3Ц + OHЦ
Na2CO3 + H2O D NaHCO3 + NaOH Cм = 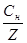 =
= 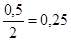 моль/л
моль/л
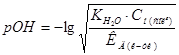 =
= 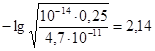
рЌ = 14 Ц 2,14 = 11,86
«адача 22. ќбчислити рЌ буферноњ системи, €ка складаЇтьс€ ≥з 100мл розчину оцтовоњ кислоти з — = 0,1моль/л ≥ 200мл розчину ацетату натр≥ю з — = 0,2моль/л, д = 1,75  10Ц5.
10Ц5.
–озвТ€занн€:
рЌ = Цlg д + lg 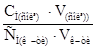 = Цlg1,75·10Ц5+lg
= Цlg1,75·10Ц5+lg 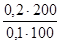 = 5,36.
= 5,36.
«адача 23. ќбчислити обТЇм розчину ацетату натр≥ю з — = 0,1моль/л ≥ обТЇм оцтовоњ кислоти з C = 0,1моль/л, €к≥ необх≥дно зм≥шати, щоб приготувати 3л ацетатного буферу з рЌ = 5,24 ( д = 1,75·10Ц5).
–озвТ€занн€:
рЌ = Цlg д+lg 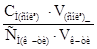 = lg1,75·10Ц5+lg
= lg1,75·10Ц5+lg 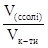 ;
;
lg 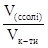 = 5,24Ц4,76 = 0,48.
= 5,24Ц4,76 = 0,48.
јнтилогарифм числа 0,48 дор≥внюЇ 3. [V(сол≥)] / [V(кислоти)] = 3/1.
ќтже, потр≥бно 3 частини розчину сол≥ ≥ одну частину розчину кислоти дл€ приготуванн€ буферноњ системи з рЌ = 5,24. ќбТЇм розчину сол≥ дор≥внюЇ: 3000·3/4 = 2250мл, а кислоти 3000·1/4 = 750мл.
–озд≥л 5
ќкисноЦв≥дновн≥ реакц≥њ (ќ¬–)
≈талони розвТ€занн€ типових задач
«авданн€. «ак≥нчити р≥вн€нн€ реакц≥й.
а) Cl 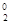 + 2NaOH " NaClЦ + NaCl+O + H2O
+ 2NaOH " NaClЦ + NaCl+O + H2O
Clо + 1 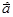 " ClЦ " ClЦ
| ||
Clо Ц 1 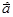 " Cl+ " Cl+
|
б) Pb+4O2 + 2KIЦ + 2H2SO4 " Pb+2SO4 + I 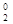 + K2SO4 + 2H2O
+ K2SO4 + 2H2O
Pb+4 + 2 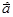 " Pb+2 " Pb+2
| ||
2IЦ Ц 2 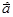 " 2Io " 2Io
|
в) 3Fe+2O + 10HN+5O3 " 3Fe+3(NO3)3 + N+2O + 5H2O
Fe+2 Ц 1 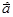 " Fe+3 " Fe+3
| ||
N+5 + 3 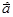 " N+2 " N+2
|
г) 2KMn+7O4 + 5KN+3O2 + 3H2SO4 " 2Mn+2SO4 + 5KN+5O3 + K2SO4 + 3H2O
Mn+7 + 5 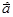 " Mn+2 " Mn+2
| ||
N+3 Ц 2 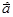 " N+5 " N+5
|
 д) 6Ag+1N+5O
д) 6Ag+1N+5O 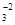 " 6AgO + 6N+4O2h + 3O
" 6AgO + 6N+4O2h + 3O  h
h
Ag+1+ 1 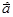 " Ag0 " Ag0
| 
| ||
N+5 + 1 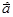 "N+4 "N+4
| |||
| 3OЦ2 Ц 6e " 3OO |
—корочуЇмо на Д3Ф: 2Ag+1N+5O 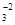 " 2Ag0 + 2N+4O2h + 3O
" 2Ag0 + 2N+4O2h + 3O 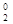 h
h
–озд≥л 6
омплексн≥ сполуки
≈талони розвТ€занн€ типових задач
ѕриклади тестового контролю:
5.1. Ќазв≥ть комплексну сполуку [Cu(NH3)4]SO4:
а) сульфаттетраам≥ак м≥д≥ (≤≤);
б) сульфаттетрам≥н м≥д≥ (≤≤);
в) м≥д≥сульфатетрам≥н (≤≤).
5.2. Ќапиш≥ть формулу ≥он≥зац≥йного ≥зомеру сполуки [Co(H2O)5]Cl3:
а) [CoCl3]. [H2O]5; б) [Co(H2O)5Cl]Cl2; в) [Co(H2O)3]Cl3.2H2O.
5.3. ¬изначте зар€д комплексного ≥ону в сполуц≥ [Cr(H2O)4Cl2]Cl.2H2O:
а) 4+; б) 2+; в) 3+.
5.4. Ќапиш≥ть формулу загальноњ нест. комплексного ≥ону [NiCl4]2Ц:
а) Kнест.= 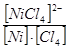 ; б) Kнест.=
; б) Kнест.= 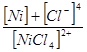 ; в) Kнест.=
; в) Kнест.= 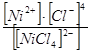 .
.
5.5. ¬изначте зар€д центрального ≥ону в сполуц≥ Fe2[Fe(CN)6]:
а) 2+; б) 6+; в) 3+.
5.6. Ќапиш≥ть формулу комплексноњ сполуки, €кщо координац≥йне число становить чотири Zn(OH)2+2NaOH а) [ZnNa2(OH)2](OH)2; б) Na2[Zn(OH)4]; в) Zn[Na2(OH)4]..7. Ќапиш≥ть продукти первинноњ дисоц≥ац≥њ сполуки Na3[AlF6]:
а) 3Na+ + [AlF6]3Ц; б) Na 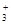 + Al3+ + 6FЦ; в) 3Na+ + Al3+ + F
+ Al3+ + 6FЦ; в) 3Na+ + Al3+ + F 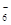 .
.
ЋјЅќ–ј“ќ–Ќ»… ѕ–ј “» ”ћ
ѕ–ј¬»Ћј –ќЅќ“» ≤ “≈’Ќ≤ ј Ѕ≈«ѕ≈ » ¬ ЋјЅќ–ј“ќ–≤ѓ Ќ≈ќ–√јЌ≤„Ќќѓ ’≤ћ≤ѓ
«агальн≥ правила
1. ѕриступати до виконанн€ лабораторного практикуму дозвол€Їтьс€ лише п≥сл€ того, €к студенти уважно ознайомл€тьс€ з темою роботи, засво€ть теоретичний матер≥ал за допомогою п≥дручник≥в, методичних пос≥бник≥в та конспект≥в лекц≥й, зробл€ть запис р≥вн€нь в≥дпов≥дних реакц≥й в лабораторному журнал≥.
2. „ерговий студент одержуЇ необх≥дн≥ дл€ роботи вс≥Їњ групи прилади та реактиви ≥ розм≥щуЇ њх на робочих м≥сц€х.
3. ” х≥м≥чн≥й лаборатор≥њ дозвол€Їтьс€ працювати лише при на€вност≥ б≥лого халата та шапочки. ожному студенту в≥дводитьс€ пост≥йне м≥сце роботи, €ке в≥н повинен тримати в чистот≥, не захаращувати його сторонн≥ми предметами, що не мають в≥дношенн€ до даноњ роботи. Ѕезладд€ та неакуратн≥сть при виконанн≥ досл≥д≥в часто призвод€ть до необх≥дност≥ повторенн€ експерименту.
4. ѕри нагр≥ванн≥ та кипТ€т≥нн≥ розчин≥в у проб≥рц≥ треба користуватись проб≥ркотримачем ≥ тримати проб≥рку так, щоб њњ отв≥р був направлений у протилежний б≥к в≥д тих, хто працюЇ поруч.
5. Ќе можна нахил€тис€ над проб≥ркою, в €к≥й нагр≥вають чи кипТ€т€ть р≥дину, щоб бризги не потрапили в обличч€.
6. ” тих випадках, коли виникаЇ необх≥дн≥сть перев≥рити запах речовин у проб≥рках в €ких збер≥гаютьс€ р≥дини, необх≥дно легким рухом долон≥ руки направити пот≥к пов≥тр€ в≥д проб≥рки до себе ≥ обережно понюхати.
7. –еактиви, дистильовану воду, електричну енерг≥ю в лаборатор≥њ сл≥д використовувати економно.
8. ¬с≥ роботи з речовинами, при взаЇмод≥њ €ких утворюютьс€ шк≥длив≥ дл€ орган≥зму гази або речовини з неприЇмним запахом, необх≥дно проводити в спец≥ально в≥дведених дл€ ц≥Їњ мети прим≥щенн€х з посиленою вентил€ц≥Їю, або п≥д т€гою. атегорично заборон€Їтьс€ працювати з вказаними речовинами на робочому м≥сц≥.
9. –озчини с≥рководню, кислот, луг≥в тощо необх≥дно зливати в спец≥ально в≥дведений посуд, щоб запоб≥гти руйнуванню канал≥зац≥йноњ системи в лаборатор≥њ. –озчини, €к≥ м≥ст€ть сполуки ср≥бла, ртут≥, свинцю, йодидЦйони необх≥дно зливати в окремий посуд дл€ њх подальшоњ регенерац≥њ.
10. атегорично заборон€Їтьс€ виконувати досл≥ди, €к≥ не в≥днос€тьс€ до даноњ лабораторноњ роботи.
11. ѕ≥сл€ зак≥нченн€ роботи необх≥дно помити проб≥рки, здати њх черговому, прибрати своЇ робоче м≥сце, виключити нагр≥вн≥ прилади, електричне осв≥тленн€, воду, вимити руки.
ѕравила поводженн€ з реактивами
Ч дл€ проведенн€ досл≥ду розчини та тверд≥ речовини беруть у таких к≥лькост€х, €ких вимагаЇ методика експерименту;
Ч реактиви збер≥гають у закритих кришками скл€нках дл€ запоб≥ганн€ њх забрудненн€;
Ч тверд≥ реактиви обережно в≥дбирають шпателем; р≥дк≥ реактиви, €к≥ знаход€тьс€ в крапельниц€х, в≥дм≥р€ють крапл€ми;
Ч надлишок реактиву не висипають ≥ не виливають у посуд, з €кого в≥н був вз€тий, щоб запоб≥гти забрудненню реактив≥в;
Ч концентрован≥ розчини кислот та луг≥в, токсичних речовин знаход€тьс€ п≥д т€гою, де з ними ≥ працюють.
–обота з кислотами ≥ лугами
1. ѕ≥д час роботи з концентрованими кислотами та лугами необх≥дно бути обережним та сл≥дкувати за тим, щоб вони не потрапили на шк≥ру ≥ од€г.
2. ѕри розведенн≥ концентрованоњ с≥рчаноњ кислоти необх≥дно обережно ≥ поступово приливати кислоту до води, а не навпаки. ÷е повТ€зано з тим, що при розведенн≥ с≥рчаноњ кислоти вид≥л€Їтьс€ велика к≥льк≥сть тепла. “ому при додаванн≥ води до кислоти розчин може розбризкуватись ≥ потрапити на шк≥ру ≥ од€г.
–обота з шк≥дливими ≥ отруйними речовинами
ѕри робот≥ з шк≥дливими ≥ отруйними речовинами (ц≥ан≥ди, сол≥ бар≥ю, ртут≥, свинцю, мишТ€ку, метал≥чна ртуть, с≥рководень тощо) необх≥дно сл≥дкувати за тим, щоб шк≥длив≥ або отруйн≥ речовини не потрапили в орган≥зм через шлунковоЦкишковий тракт. ” звТ€зку з цим њсти в лаборатор≥њ категорично заборон€Їтьс€. ѕ≥сл€ роботи в лаборатор≥њ необх≥дно добре вимити руки. Ѕалони з ртуттю або заповнен≥ нею прилади необх≥дно пом≥стити на спец≥альн≥ п≥дставки, щоб у випадку пошкодженн€ прилад≥в основна маса ртут≥ потрапила на п≥дставку, а не на робочий ст≥л чи п≥длогу. якщо ртуть все ж розлилась, њњ треба дуже швидко з≥брати за допомогою м≥дних дроту або пластинки, а пот≥м засипати с≥ркою. ѕрацювати з ртуттю дозвол€Їтьс€ лише у спец≥альних прим≥щенн€х.






