р≥м параметр≥в стану в термодинам≥ц≥ використовують величини, €к≥ називають термодинам≥чними функц≥€ми. ƒо термодинам≥чних функц≥й в≥днос€тьс€ внутр≥шн€ енерг≥€ (U), теплота (Q), механ≥чна робота (ј), ентальп≥€ (Ќ), ентроп≥€ (S) та ≥нш≥.
¬заЇмозвТ€зок термодинам≥чних характеристик систем розгл€даютьс€ на основ≥ досл≥джень закон≥в термодинам≥ки.
ѕриклад 2. —формулюйте перший закон термодинам≥ки. ќбгрунтуйте його суть на прикладах.
¬≥дпов≥дь:
ƒосв≥д людства св≥дчить про те, що не можна побудувати машину, €ка працювала б, не витрачаючи енерг≥њ. “аку у€вну машину, €ку марно прагнули винайти, назвали в≥чним двигуном (perpetuum mobile) першого роду. ÷е привело до важливого наукового узагальненн€, у вигл€д≥ першого закону (принципа, начала) термодинам≥ки, €кий формулюють по-р≥зному:
- в≥чний двигун першого роду неможливий;
- теплота, €ка поглинаЇтьс€ системою, витрачаЇтьс€ на зб≥льшенн€ внутр≥шньоњ енерг≥њ ≥ на механ≥чну роботу, €ку виконуЇ система.
÷ей закон представл€Ї собою окремий випадок закону збереженн€ енерг≥њ. …ого використовують до процес≥в, €к≥ супроводжуютьс€ перетворенн€м теплоти п≥д час виконанн€ певноњ роботи.
–озгл€немо два випадки:
¬ипадок перший.
”€в≥м цил≥ндр з невагомим поршнем (рис. 9.1). ” цил≥ндр≥ м≥ститьс€ 1 моль газу при визначеному тиску ≥ початков≥й температур≥ “1. ѕоршень закр≥плено в положенн≥ а так, що в≥н не перем≥щуЇтьс€, тобто газ не може виконувати роботу проти зовн≥шнього тиску.

1 Ц цил≥ндр з газом; 2 Ц невагомий поршень;
3 Ц вантаж, що зр≥вноважуЇ тиск газу –.
–исунок 9.1 Ц ≤люстрац≥€ пон€тт€ ентальп≥њ
якщо п≥двести теплоту Qv (≥ндекс v вказуЇ на незм≥нний обТЇм), вона витрачаЇтьс€ на п≥двищенн€ температури системи до “2, тобто на зм≥ну њњ внутр≥шньоњ системи.
” першому стан≥ внутр≥шн€ енерг≥€ маЇ значенн€ U1, в другому U2. ќск≥льки немаЇ умов, за €ких внутр≥шн€ енерг≥€ дор≥внюЇ нулю, то обчислюють не абсолютн≥ величини, а њх зм≥ну ∆U п≥д час переходу ≥з стану 1 у стан 2. якщо зм≥на внутр≥шньоњ енерг≥њ Ї результатом п≥дведенн€ теплоти Qv, то це записуЇтьс€ р≥вн€нн€м:
Qv = U2-U1 = ∆U
«ростанн€ запасу внутр≥шньоњ енерг≥њ можна оц≥нити за зм≥ною температури системи, коли в≥домо теплоЇмн≥сть с њњ складових частин.
“еплоЇмн≥сть системи (речовини) Ц це в≥дношенн€, к≥лькост≥ теплоти, €ку система вбираЇ при нагр≥ванн≥ або в≥ддаЇ при охолодженн≥, до в≥дпов≥дноњ зм≥ни температури. “еплоЇмн≥сть, в≥днесена до одиниц≥ маси речовини, називаЇтьс€ питомою, до мол€ Ц мольною.
якщо мольна теплоЇмн≥сть визначаЇтьс€ при сталому обТЇм≥ вона називаЇтьс€ ≥зохорною ≥ позначаЇтьс€ €к —v, €кщо при сталому тисков≥, - ≥зобарною ≥ позначаЇтьс€ —p.
¬ цьому випадку записуЇтьс€ р≥вн€нн€:
Qv=Cv(T2-T1)
де Cv - мольна теплоЇмн≥сть газу при сталому обТЇм≥;
“1 Ц початкова температура системи;
“2 Ц к≥нцева температура системи.
¬ипадок другий
ƒо газу п≥дводитьс€ теплота, ≥ в≥н виконуЇ роботу. ѕоршень перем≥щуЇтьс€ з положенн€ а в положенн€ б, тод≥ закон збереженн€ енерг≥њ записують так:
|
|
|
Qp = ∆U+A
де Qp Ц п≥дведена теплота при збереженн≥ сталого тиску в цил≥ндр≥;
ј Ц робота, що њњ виконуЇ поршень. ѓѓ можна обчислити так:
ј = –(V2-V1)
де V1 Ц початковий обТЇм газу;
V2 Ц к≥нцевий обТЇм газу.
–обота, з €кою найчаст≥ше маЇмо справу в х≥м≥њ, звТ€зана ≥з розширенн€м системи. “аке розширенн€ в≥дбуваЇтьс€, наприклад, п≥д час реакц≥њ взаЇмод≥њ кальц≥й карбонату з хлоридною кислотою, в результат≥ чого вид≥л€Їтьс€ газ карбон (≤V) оксид. (рис. 9.2). ¬ цьому випадку робота (ј), €ка виконуЇтьс€ системою визначаЇтьс€ формулою:
ј = - –∆V
де – Ц зовн≥шн≥й тиск,
∆V Ц зм≥на обТЇму системи.
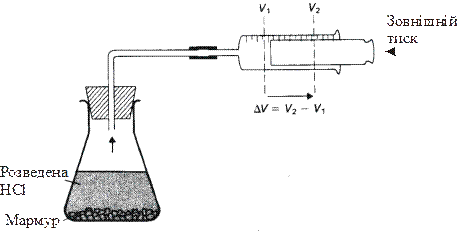
–исунок 9.2 Ц –обота, що виконуЇтьс€ х≥м≥чною системою
ƒл€ багатьох х≥м≥чних реакц≥й, €к≥ виконуютьс€ в лабораторних умовах, зовн≥шн≥й тиск сп≥впадаЇ ≥з атмосферним тиском. «нак Ум≥нусФ у наведеному р≥вн€нн≥ в≥дпов≥даЇ тому, що робота виконуЇтьс€ системою, а значить, система втрачаЇ енерг≥ю.
«м≥ну внутр≥шньоњ енерг≥њ системи можна визначити €к р≥зницю м≥ж к≥лькост€ми п≥дведеноњ теплоти ≥ виконаноњ роботи. ѕотр≥бно зазначити, що теплота ≥ робота Ц не види енерг≥њ, а дв≥ форми переходу енерг≥њ в≥д одноњ системи до ≥ншоњ.
–≥вн€нн€ можна записати так:
Qp = U2 - U1 + P(V2 - V1) = (U2 + PV2) - (U1 + PV1)
—ума (U+PV) Ї властив≥стю системи, функц≥Їю стану, €ку прийн€то позначати л≥терою Ќ, ≥ називати ентальп≥Їю (в≥д грецького слова, що означаЇ нагр≥ваю). ўе цю функц≥ю називають тепловм≥стом.
–≥вн€нн€ можна записати так: Qp = H2 - H1 = ∆H
≈нтальп≥ю можна розгл€дати, €к енерг≥ю системи що розширюЇтьс€ п≥д час нагр≥ванн€.
ќтже, досл≥дженн€ першого закону термодинам≥ки призводить до взаЇмозвТ€зку таких властивостей системи (функц≥€ стану), €к ентальп≥€ ≥ внутр≥шн€ енерг≥€.
ѕриклад 1.
¬изначити зм≥ну внутр≥шньоњ енерг≥њ при випаровуванн≥ 25г води при 200—.
–озвТ€занн€.
1. ѕрипустимо, що вод€на пара п≥дл€гаЇ законам ≥деального газу тод≥ обТЇмом р≥дини пор≥вн€но з обТЇмом газу можна знехтувати.
2. ¬икористовуЇмо дов≥дников≥ дан≥: теплота пароутворенн€ води дор≥внюЇ 2451.8 ƒж/г.
3. «а р≥вн€нн€м V = (mRT)/(PM)
ќбчислюЇмо обТЇм вод€ноњ пари при 200— ≥ тисков≥ 1 атм:
V =(25Ј0.082Ј393)/18=33,375 л
4. –обота розширенн€ газу:
ј = PV
A = 33,375 лЈатм
1лЈатм = 101,25 ƒж
ќтже,
A = 33,375Ј101,25 = 3378,58 ƒж
5. ≥льк≥сть теплоти, що вбираЇ система при випаровуванн≥ 25 г води:
Qp = 2451,72Ј25 = 61295,6 ƒж
6. «м≥на внутр≥шньоњ енерг≥њ при випаровуванн≥ води:
∆U=Qp - A
∆U = 61295.6 Ј 3378.5 = 57917.02 ƒж
¬≥дпов≥дь: зм≥на внутр≥шньоњ енерг≥њ ∆U дор≥внюЇ
57917.02 ƒж або 57,92 кƒж
ѕриклад 3. як≥ процеси вивчаЇ термох≥м≥€? ќбірунтуйте своњ судженн€ на прикладах.
¬≥дпов≥дь:
“ермох≥м≥€ Ц це використанн€ першого закону термодинам≥ки в х≥м≥чних процесах. ѕ≥д час розробок нових технолог≥чних процес≥в та проектуванн€ обладнанн€ необх≥дно знати теплов≥ ефекти х≥м≥чних реакц≥й. Ѕ≥ологи повинн≥ також знати чи достатньо енерг≥њ, €ка вид≥л€Їтьс€ в результат≥ реакц≥њ, щоб п≥дтримати життЇздатн≥сть кл≥тини. Ќа ц≥ питанн€ дозвол€ють в≥дпов≥сти термох≥м≥чн≥ розрахунки. ќтже, термох≥м≥€ вивчаЇ теплов≥ ефекти х≥м≥чних реакц≥й, тобто к≥льк≥сть теплоти, €ка вид≥л€Їтьс€ або поглинаЇтьс€ п≥д час х≥м≥чних реакц≥й.
ѕерш за все дамо в≥дпов≥дь на питанн€: чому х≥м≥чн≥ реакц≥њ супроводжуютьс€ певним тепловим ефектом?
ўоб дати в≥дпов≥дь на це питанн€, розгл€немо приклад взаЇмод≥њ водню ≥з хлором:
|
|
|
H2 + Cl2 = 2 HCl
ƒл€ утворенн€ двох моль хлороводню повинн≥ роз≥рватись х≥м≥чн≥ звТ€зки в 1 мол≥ водню ≥ 1 мол≥ хлору. ѕор≥вн€Їмо енерг≥њ х≥м≥чних звТ€зк≥в
H-H 436кƒж/моль
Cl-Cl 242,3кƒж/моль
H-Cl 431,4кƒж/моль
ќтже дл€ розриву в молекулах х≥м≥чних звТ€зк≥в м≥ж воднем та хлором необх≥дно затратити:
436 + 242,3 = 678,3 кƒж
”творенн€ х≥м≥чного звТ€зку в 2 мол€х хлороводню супроводжуЇтьс€ вид≥ленн€м:
431,4 Ј 2 = 862,8 кƒж
“аким чином, сумарна теплота, €ка вид≥л€Їтьс€ дор≥внюЇ:
862,8 - 678,3 = 184,5 кƒж
ќтже, теплов≥ ефекти реакц≥њ залежать в≥д природи речовин, тобто в≥д њх х≥м≥чних звТ€зк≥в. ѕ≥д час х≥м≥чних реакц≥й проходить перетворенн€ внутр≥шньоњ енерг≥њ системи, €ка завжди знаходитьс€ в прихован≥й форм≥, в теплову.
–еакц≥њ, п≥д час €ких внутр≥шн€ енерг≥€ речовини перетворюЇтьс€ у теплову (тепло вид≥л€Їтьс€) називаЇтьс€ екзотерм≥чними.
–еакц≥њ, п≥д час €ких теплова енерг≥€ перетворюЇтьс€ у внутр≥шню енерг≥ю (тепло поглинаЇтьс€) називаЇтьс€ ендотерм≥чними.
ѕриклад 1.
ѕроведено розчиненн€ бар≥й хлорату Ba(ClO3)2 у вод≥ з температурою 30о—. який цей процес: екзотерм≥чний чи ендотерм≥чний? який знак маЇ величина ∆Ќ? ¬≥дпов≥дь обгрунтуйте.
–озвТ€занн€.
1. ѕониженн€ температури розчину п≥д час розчиненн€ сол≥ вказуЇ на те, що процес розчиненн€ йде з поглинанн€м тепла ≥з навколишнього середовища. «начить, цей процес ендотерм≥чний.
2. ўоб температура ц≥Їњ системи, €ка складаЇтьс€ ≥з сол≥ ≥ води, залишалас€ 300—, до системи необх≥дно п≥двести тепло. «начить величина ΔЌ повинна бути позитивною.
’≥м≥чн≥ р≥вн€нн€, в €ких зазначено тепловий ефект реакц≥й, називаютьс€ термох≥м≥чними р≥вн€нн€ми.
Ќаприклад, р≥вн€нн€ екзотерм≥чних реакц≥й:
CH4(г)+2O2(г) → CO2(г)+2Ќ2ќ(г)+891 кƒж
Cu(т)+Cl2(г) → CuCl2(т)+223 кƒж
–≥вн€нн€ ендотерм≥чних реакц≥й:
N2(г)+ќ2(г) → 2NO(г)-180,5 кƒж
CO2(г)+—(г) → 2—ќ(г)-160 кƒж
“еплов≥ ефекти реакц≥й визначають здеб≥льшого дл€ речовин, що окиснюютьс€ п≥д час реакц≥й, у к≥лькост≥ речовини 1 моль. “ому в термох≥м≥чних р≥вн€нн€х допускаютьс€ ≥ дробов≥ коеф≥ц≥Їнти:
H2(г)+0.5O2(г) →H2O(г)+285.8 кƒж
O2(г) →2/3O3(г) Ц 96 кƒж
якщо, в термох≥м≥чному р≥вн€нн≥ не використовуютьс€ дробов≥ коеф≥ц≥Їнти, то к≥льк≥сть теплоти зазначаЇтьс€ з розрахунку к≥лькост≥ моль речовини, що окиснюЇтьс€. Ќаприклад:
2C2H2+5O2→4CO2+2H2O+2600 кƒж
C2H2+2.5O2→2CO2+H2O+1300 ƒж
ѕри розрахунках теплоти утворенн€ та теплоти згор€нн€ речовин використанн€ дробових коеф≥ц≥Їнт≥в обовТ€зкове. Ќаприклад:
Ca(к)+1/2 O2(г) = CaO(к) ∆Ќ=-635,1 кƒж/моль
1/2 N2(г)+1/2 O2(г) = NO(г) ∆Ќ=90,4 кƒж/моль
“еплотою (ентальп≥ею) утворенн€ називаЇтьс€ к≥льк≥сть теплоти, €ка вид≥л€Їтьс€ або вбираЇтьс€ п≥д час утворенн€ 1 моль речовини з простих речовин. ≥льк≥сть теплоти, що вид≥л€Їтьс€ або поглинаЇтьс€ при розкладанн≥ 1моль складноњ речовини називаЇтьс€ теплотою розкладанн€. Ќаприклад
H2O(р) = H2 (г) + 1/2 O2 (г) ∆Ќ=285.9 кƒж/моль
Ќа практиц≥ ≥нод≥ визначають теплоти згор€нн€ орган≥чних речовин. “еплотою згор€нн€ називаЇтьс€ к≥льк≥сть теплоти, €ка вид≥л€Їтьс€ п≥д час згор€нн€ 1 моль речовини.
≥нцевими продуктами њх згор€нн€ найчаст≥ше Ї вода ≥ карбон (IV) оксид, ≥нод≥ сульфур (IV) оксид.
Ќаприклад:
C2H2+2½O2=2—O2+H2O ∆Ќ=-1301.5 кƒж/моль
“еплота згор€нн€ харчових продукт≥в у живому орган≥зм≥ Ї джерелом енерг≥њ, за рахунок €коњ зд≥йснюЇтьс€ його життЇд≥€льн≥сть.
“еплотою розчиненн€ Ц називають к≥льк≥сть теплоти, що вид≥л€Їтьс€ або поглинаЇтьс€ при розчиненн≥ одного мол€ речовини в такому обТЇм≥ розчинника, коли доливанн€ його не спричинюЇ зм≥ни теплового ефекту.
“еплота розчиненн€ залежить в≥д природи розчинника ≥ природи розчиненоњ речовини. ” термох≥м≥чному в≥дношенн≥ процес розчиненн€ складний. ƒе€к≥ стад≥њ цього процесу можуть бути ендотерм≥чними ≥ ∆Ќ њх додатна (руйнуванн€ структури твердих т≥л, поширенн€ молекул розчиненоњ речовини в обТЇм≥ розчинника, дисоц≥ац≥€ молекул на йони), а де€к≥ екзотерм≥чн≥ ≥ ∆Ќ њх в≥дТЇмне (х≥м≥чна взаЇмод≥€ розчиненоњ речовини з розчинником).
|
|
|
¬ основному тепловий ефект розчиненн€ твердих речовин складаЇтьс€ з двох складових частин
∆Ќрозч.= ∆Ќ1 + (-∆Ќ2)
де ∆Ќ1 Ц теплота, що витрачаЇтьс€ на руйнуванн€ кристал≥чноњ реш≥тки;
∆Ќ2 Ц теплота г≥дратац≥њ (сольватац≥њ), тобто теплота, що вид≥л€Їтьс€ при х≥м≥чн≥й взаЇмод≥њ молекул розчиненоњ речовини з молекулами розчинника. ѕриклад:
а) розчиненн€ у вод≥ кал≥й г≥дроксиду супроводжуЇтьс€ вид≥ленн€м тепла:
KOH(к)+хЌ2ќ(р)=KOH ∙ хЌ2ќ(р) ∆Ќрозч.=-55,3 кƒж/моль
б) п≥д час розчиненн€ кухонноњ сол≥ у вод≥ в≥дбуваЇтьс€ охолодженн€ системи:
NaCl(к)+xH2O(р)=NaCl Ј xH2O(р) ∆Ќрозч.=5 кƒж/моль
“ак≥ речовини використовують дл€ одержанн€ низьких температур у холодильн≥й промисловост≥ ≥ називають кр≥ог≥дратними сум≥шами.
ќдиницею вим≥рюванн€ теплового ефекту реакц≥њ Ї ƒж/моль або кƒж/моль. ≥льк≥сть теплоти, що вид≥л€Їтьс€ при х≥м≥чних реакц≥€х, пр€мо пропорц≥йна к≥лькост≥ речовини, що бере участь у х≥м≥чн≥й реакц≥њ.
¬ термох≥м≥чних р≥вн€нн€х х≥м≥чних реакц≥й тепловий ефект позначають ∆Ќ. ” в≥дпов≥дност≥ до прийн€тоњ у термодинам≥ц≥ системи знак≥в дл€ екзотерм≥чних реакц≥й ∆Ќ<0 а дл€ ендотерм≥чних - ∆Ќ>0.
≥льк≥сть теплоти, що вид≥л€Їтьс€ або поглинаЇтьс€ п≥д час х≥м≥чних реакц≥й, звТ€зана ≥з зм≥нами ентальп≥њ та внутр≥шньоњ енерг≥њ наступними сп≥вв≥дношенн€ми:
∆Ќ= -Qp
∆U= -Qv
де ∆Ќ Ц зм≥на ентальп≥њ; Qp Ц к≥льк≥сть теплоти, що вид≥л€Їтьс€ чи поглинаЇтьс€ при пост≥йному тиску; Qv Ц к≥льк≥сть теплоти, що вид≥л€Їтьс€ чи поглинаЇтьс€ при пост≥йному обТЇм≥; ∆U Ц зм≥на внутр≥шньоњ енерг≥њ.
якщо реакц≥€ в≥дбуваЇтьс€ за стандартних умов (температура 298,15 або 25 0—, тиск 101325 ѕа, концентрац≥€ ус≥х речовин у розчин≥ або газ≥ 1моль/л≥тр), то зм≥ну ентальп≥њ позначають символом ∆Ќ0. –озгл€немо термох≥м≥чне р≥вн€нн€ реакц≥њ водню з киснем
2Ќ2(г)+ќ2(г) =2Ќ2ќ(г) ∆Ќ0 = - 483кƒж/моль
” дужках вказано агрегатний стан речовини, в≥д €кого залежить тепловий ефект реакц≥њ. ÷ей запис означаЇ, що п≥д час взаЇмод≥њ 2 моль водню з 1 моль кисню утворюЇтьс€
2 моль пари води ≥ вид≥л€Їтьс€ (за стандартних умов)
483,6 кƒж теплоти.
≤з зб≥льшенн€м к≥лькост≥ реагуючих речовин тепловий ефект реакц≥њ пропорц≥йно зростаЇ.
«м≥на ентальп≥њ пр€моњ ≥ оборотньоњ реакц≥й р≥вна за величиною, але протилежна за знаком.
ѕриклад 2
ћаЇмо х≥м≥чний процес, €кий описуЇтьс€ термох≥м≥чним р≥вн€нн€м
—ќ2(г) + 2Ќ2ќ (г) = —Ќ4(г) +2ќ 2(г) ∆Ќ=802 кƒж
ѕотр≥бно довести, що ∆Ќ пр€моњ реакц≥њ + ∆Ќ зворотноњ реакц≥њ = 0
якщо б п≥д час згор€нн€ —Ќ4 вид≥лилось б≥льше теплоти, н≥ж п≥д час взаЇмод≥њ —ќ2 ≥ Ќ2ќ, то можна б було одержати необмежен≥ к≥лькост≥ енерг≥њ. —палюючи де€ку к≥льк≥сть —Ќ4, достатньо було б лише зберегти ту частину одержаноњ енерг≥њ, €ка необх≥дна дл€ в≥дновленн€ —Ќ4, а решту використати дл€ одержанн€ корисноњ роботи. ѕ≥сл€ в≥дновленн€ —Ќ4 його можна було б знову спалити ≥ так повторювати процес до неск≥нченост≥. ќднак це вступаЇ в протир≥чч€ ≥з законом збереженн€ енерг≥њ та нашим практичним досв≥дом.
«м≥на ентальп≥њ реакц≥њ залежить також в≥д агрегатного стану реагент≥в ≥ продукт≥в. якщо в реакц≥њ згор€нн€ метану вода була б р≥диною, а не газопод≥бною, то ∆Ќ дор≥внювало б - 790 кƒж зам≥сть - 802 кƒж. ¬ цьому випадку в навколишнЇ середовище передаЇтьс€ б≥льше теплоти, тому що при конденсац≥њ 2 моль газопод≥бноњ води в р≥дкий стан додатково вид≥л€Їтьс€ ще 88 кƒж:
2 Ќ2ќ(г) → 2 Ќ2ќ(р) ∆Ќ=-88 кƒж
|
|
|
ѕриклад 3.
ѕ≥д час згор€нн€ магн≥ю масою 6 г у кисн≥ вид≥лилас€ теплота у к≥лькост≥ 150 кƒж. —клад≥ть термодинам≥чне р≥вн€нн€ реакц≥њ.
1. —кладаЇмо р≥вн€нн€ реакц≥њ гор≥нн€ магн≥ю у кисн≥
2Mg+O2=2MgO
2. ќбчислюЇмо к≥льк≥сть речовини магн≥ю, що згор≥в
ν(Mg) = 6г/(24г/моль) = 0,25 моль
3. ќбчислюЇмо к≥льк≥сть теплоти, €ка б вид≥лилась п≥д час згор€нн€ двох моль магн≥ю
∆Ќ = (2моль/0,25моль)Ј(-150 кƒж) = Ц 1200 кƒж
4. «аписуЇмо р≥вн€нн€ реакц≥њ
2Mg(т) + ќ2 (г) = 2Mgќ(т) ∆Ќ = Ц 1200кƒж
ѕриклад 4.
√ор≥нн€ с≥рки в кисн≥ описуЇтьс€ термох≥м≥чним р≥вн€нн€м реакц≥њ:
S(т)+ќ2 (г) ↔ Sќ2 (г) ∆Ќ = Ц 297 кƒж
¬изначити масу с≥рки та обТЇм кисню за нормальних умов, €кщо п≥сл€ њх взаЇмод≥њ вид≥лилось 891 кƒж теплоти.
1. ќбчислюЇмо к≥льк≥сть речовини с≥рки, поск≥льки зг≥дно р≥вн€нн€ реакц≥њ 297 кƒж в≥дпов≥даЇ 1 моль S та 1 моль ќ2:
ν(S) = ν(O2) = ((891 кƒжЈ1 моль)/297 кƒж) = 3 моль
2. ќбчислюЇмо масу с≥рки ≥ обТЇм кисню:
m(S) = 32 г/мольЈ3 моль = 96 г
V(O2) = 22.4Ј3 = 67,2 л
¬≥дпов≥дь:маса с≥рки Ц 96 г, обТЇм кисню Ц 67,2 л.
ѕриклад 4. —формулюйте другий закон термодинам≥ки.
¬≥дпов≥дь:
ѕерший закон термодинам≥ки виражаЇ незнищенн≥сть ≥ екв≥валентн≥сть р≥зних форм енерг≥њ при взаЇмних переходах, встановлюЇ сп≥вв≥дношенн€, €к≥ звТ€зують внутр≥шню енерг≥ю ≥з к≥льк≥стю теплоти ≥ виконаною роботою, тобто дозвол€Ї скласти енергетичний баланс процесу.
ќднак, перший закон не розгл€даЇ питанн€, €к≥ звТ€зан≥ ≥з можлив≥стю проходженн€ ≥ напр€мком процесу. ” навколишньому св≥т≥ р≥зн≥ ф≥зичн≥ ≥ х≥м≥чн≥ процеси в≥дбуваютьс€ у певному напр€м≥. Ќаприклад, теплота переходить в≥д б≥льш нагр≥того т≥ла до менш нагр≥того, газ поширюЇтьс€ у область низького тиску. ¬с≥ ц≥ процеси в≥дбуваютьс€ самов≥льно (або спонтанно), без затрат енерг≥њ ззовн≥, ≥ т≥льки в певному напр€мку. «воротн≥ процеси самов≥льно не в≥дбуваютьс€ (газ не стискаЇтьс€ сам по соб≥, теплота не може перейти в≥д холодного т≥ла до б≥льш нагр≥того ≥ т.д.). ўоб спонтанний процес в≥дбувавс€ оборотньо потр≥бна енерг≥€ ззовн≥. Ќаприклад, щоб п≥дн€ти воду, €ка падаЇ з висоти ≥ обертаЇ турб≥ну, на попередн≥й р≥вень, потр≥бно затратити енерг≥ю.






