| „астинка | —имвол | ћаса спокою | «ар€д | ||
| абсолютна кг | в≥дносна а.о.м. | в≥дносний | електричний л | ||
| протон | 
| 1,673 ∙10-27 | 1.007276 | +1 | 1,602∙10-19 |
| нейтрон | 
| 1,675 ∙10-27 | 1.008665 | ||
| електрон | ℮ 
| 9,109 ∙10-31 | 0,000549 | Ц1 | 1,602∙10-19 |
“аблиц€ 6
ќ—Ќќ¬Ќ≤ “»ѕ» –јƒ≤ќј “»¬Ќ»’ ѕ≈–≈“¬ќ–≈Ќ№
| ядерн≥ реакц≥њ | –≥вн€нн€ реакц≥њ | ¬изначенн€ |
α Ц розпад
самов≥льне перетворенн€ €дер ≥з випусканн€м
α Ц частин  He) He)
|  Ra = Ra = 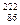 Rn + Rn +  He He
| ¬иникаЇ ≥зотоп елемента, зм≥щеного на дв≥ кл≥тинки в≥д вих≥дного до початку ѕер≥одичноњ системи ≥ масове число його на 4 одиниц≥ менше |
| β - розпад |  U = U =  Np + e Np + e
| ѕриводить до виникненн€ ≥зотопу елемента, зм≥щеного на одну кл≥тинку в≥д вих≥дного до к≥нц€ ѕер≥одичноњ системи ≥з тим же масовим числом |
| ≈лектронний захват |  + е = + е = 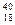 Ar Ar
| –еакц≥€ супроводжуЇтьс€ захватом €дром електрона, €кий знаходитьс€ на найближчому до €дра п≥др≥вн≥ |
| —понтанний под≥л €дер |  U = U =  Kr + Kr +  Ba Ba
| –еакц≥€ супроводжуЇтьс€ утворенн€м двох нових рад≥оактивних елемент≥в |
“аблиц€ 7
ѕ–ј¬»Ћј — ЋјƒјЌЌя ≈Ћ≈ “–ќЌЌ»’ ‘ќ–ћ”Ћ
| Ќазва | ‘ормулюванн€ | «астосуванн€ |
| ѕринцип м≥н≥муму енерг≥њ | Ќайст≥йк≥шому стану електрона в атом≥ в≥дпов≥даЇ м≥н≥мальна енерг≥€ | ≈лектрон займаЇ јќ з найнижчою енерг≥Їю |
| ѕринцип ѕаул≥ | јтом не може мати двох електрон≥в з однаковим значенн€м вс≥х чотирьох квантових чисел | N = 2n2, (число електрон≥в на р≥вн≥); максимальне число електрон≥в на п≥др≥вн€х: s = 2; p =6; d =10; f =14 |
| ѕерше правило лечковського | ≈нергетичн≥ п≥др≥вн≥ заповнюютьс€ за зростанн€м суми (n + l) | ¬казуЇ посл≥довн≥сть заповненн€ п≥др≥вн≥в |
| ƒруге правило лечковського | ѕри р≥вност≥ суми (n + l) перевага за значенн€м n | |
| ѕравило √унда | —умарне сп≥нове число електрон≥в певного п≥др≥вн€ повинно бути максимальним | ¬казуЇ пор€док заповненн€ р≥вноц≥нних јќ |
“аблиц€ 8
Ќј«¬ј “ј ≤Ќ“≈–ѕ–≈“ј÷≤я ¬јЌ“ќ¬»’ „»—≈Ћ
| Ќазва | —имвол | ўо визначаЇ | ћожлив≥ значенн€ |
| √оловне | n | ≈нерг≥ю орб≥тал≥ (енергетичний р≥вень) | ц≥л≥ числа в≥д 1 до ∞ |
| ѕоб≥чне (орб≥тальне) | l | ‘орму орб≥тал≥ (енергетичний п≥др≥вень) | ц≥л≥ числа в≥д 0 до n-1 |
| ћагн≥тне | ml | ќр≥Їнтац≥ю орб≥тал≥ у простор≥ | Ц lЕ0Е+ l ml = 2l + 1 |
| —п≥нове | ms | ¬ласний магн≥тний момент електрона | ½ ≥ -½ |
“аблиц€ 9
¬јЌ“ќ¬»… —“јЌ ≈Ћ≈ “–ќЌ≤¬,
™ћЌ≤—“№ ≈Ќ≈–√≈“»„Ќ»’ –≤¬Ќ≤¬ ≤ ѕ≤ƒ–≤¬Ќ≤¬
| ≈лект-ронна оболон-ка | ≈нерге-тичний р≥вень N | ≈нерге-тичний п≥д-р≥вень | ћожлив≥ значенн€ Ml | „исло орб≥талей | „исло електрон≥в max | |||
| l | 2l + 1 | n2 | (2l + 1)2 | 2n2 | ||||
| K | s | |||||||
| L | s p | 0; Ц1, 0, +1 | ||||||
| M | s p d | 0; Ц1, 0, +1; Ц2,Ц1,0,+1,+2 | ||||||
| N | s p d f | 0; Ц1, 0, +1; Ц2,Ц1,0, +1,+2 Ц3,Ц2,Ц1, 0,+1,+2,+3 |
’≤ћ≤„Ќ»… «¬Тя«ќ
|
|
|
“аблиц€ 10
¬»ƒ» «¬Тя« ≤¬
| є | ¬ид звТ€зку | ¬изначенн€ | ћехан≥зм утворенн€ |
| овалентний звТ€зок | «вТ€зок атом≥в за допомогою сп≥льних електронних пар, тобто перекриванн€ јќ | ќбм≥нний ЌЦ + ∙Ќ+→Ќ׃Ќ | |
| овалентний звТ€зок | «вТ€зок м≥ж атомами, один з €ких маЇ непод≥лену пару електрон≥в, а ≥нший в≥льну јќ | ƒонорно-акцепторний Ќ-׃ + □Ќ+→Ќ Ц Ќ | |
| ≤онний | «вТ€зок м≥ж ≥онами, що зд≥йснюЇтьс€ завд€ки електростатичному прит€ганню | 2Na + Cl2=2NaCl Na -1e → Na+ Cl2 + 2e → 2Cl- | |
| ћетал≥чний | «вТ€зок у металах та сплавах м≥ж позитивно зар€дженими ≥онами та валентними електронами, що Ї сп≥льними дл€ вс≥х ≥он≥в | ||
| ¬одневий (м≥жмолекул€рний) | “рицентровий звТзок м≥ж двома електронегатив-ними атомами через атом водню | H+- F-∙∙∙∙ H+- F- |
“аблиц€ 11






