| ¬ещество (состо€ние) |  кƒж/моль
кƒж/моль
|  ƒж/(мольЈ )
ƒж/(мольЈ )
|  кƒж/моль
кƒж/моль
| —р0 298, ƒж/(мольЈ *) | |||
| ѕростые вещества | |||||||
| Ag (к) | 42,55 | 25,44 | |||||
| Al (к) | 28,33 | 24,35 | |||||
| As (к, серый) | 35,61 | 24,74 | |||||
| Au (к) | 47,40 | 25,36 | |||||
| Br2 (г) | 30,91 | 254,37 | 3,14 | 36,07 | |||
| Br2 (ж) | 152,21 | 75,69 | |||||
| C (графит) | 5,74 | 8,54 | |||||
| Ca (к) | 41,42 | 26,36 | |||||
| Cd (к) | 51,76 | 25,94 | |||||
| —l2 (г) | 222,98 | 33,93 | |||||
| Co (к) | 30,04 | 24,81 | |||||
| Cu (к) | 33,14 | 24,43 | |||||
| F2 (г) | 202,67 | 31,30 | |||||
| Fe (к) | 27,15 | 24,98 | |||||
| H2 (г) | 130,52 | 28,83 | |||||
| J2 (г) | 62,43 | 260,60 | 19,39 | 36,90 | |||
| K (к) | 64,18 | 29,58 | |||||
| Mg (к) | 32,68 | 24,89 | |||||
| N2 (г) | 191,50 | 29,12 | |||||
| Ni (к) | 29,87 | 26,07 | |||||
| O2 (г) | 205,04 | 29,37 | |||||
| P (к, белый) | 41,09 | 23,82 | |||||
| P (к, красный) | −17,45 | 22,80 | −12,00 | 21,39 | |||
| Pb (к) | 64,81 | 26,82 | |||||
| S(г) | 278,90 | 167,70 | − | ||||
| Si (к) | 18,33 | 19,99 | |||||
| Sn (к) | 51,55 | 26,99 | |||||
| Zn (к) | 41,63 | 25,44 | |||||
| Ќеорганические вещества | |||||||
| AgBr (к) | −100,40 | 107,11 | -97,02 | 52,30 | |||
| Ag2O(к) | −30,54 | 121,75 | -10,90 | 65,86 | |||
| Al2O3 (к, корунд) | −1675,69 | 50,92 | −1582,27 | 79,04 | |||
| ¬—l 3 (г) | −402,96 | 290,08 | −387,98 | 62,63 | |||
| ¬F3 (г) | −1136,58 | 254,01 | −1119,93 | 50,46 | |||
| —O (г) | −110,53 | 197,55 | −137,15 | 29,14 | |||
| —ќ2 (г) | −393,51 | 213,66 | −394,37 | 37,11 | |||
| —а—ќ3 (к) | −1206,83 | 91,71 | −1128,35 | 83,47 | |||
| —а—l2 (к) | −795,92 | 108,37 | −749,34 | 72,59 | |||
| —аќ (к) | −635,50 | 39,70 | −604,20 | 67,03 | |||
| CaSO4 (к) | −1432,70 | 106,70 | −1320,30 | 99,60 | |||
| Ca3PO4 (к) | −4137,60 | 236,00 | −3899,50 | 227,80 | |||
| ѕродолжение табл. 2 | |||||
| ¬ещество (состо€ние) |  кƒж/моль
кƒж/моль
|  ƒж/(моль Ј )
ƒж/(моль Ј )
|  кƒж/моль
кƒж/моль
| —р0 298, ƒж/(мольЈ *) | |
| Ca3PO4 (к) | −4137,60 | 236,00 | −3899,50 | 227,80 | |
| CaSO4Ј0,5H2O(к) | −1573,00 | 134,00 | −1435,00 | 121,00 | |
| CaSO4 Ј2H2O (к) | −2021,10 | 194,00 | −1795,90 | 186,20 | |
| —uќ (к) | −162,00 | 42,63 | −134,26 | 42,30 | |
| CuS (к) | −48,50 | 66,50 | −53,60 | 47,84 | |
| CuSќ4 (к) | −771,10 | 93,93 | −661,80 | 63,64 | |
| Fеќ (к) | −264,85 | 60,75 | −244,30 | 49,92 | |
| Fе2 ќ3 (к) | −822,16 | 87,45 | −740,34 | 103,76 | |
| Fe3O4(к) | −1117,70 | 146,40 | −1014,20 | 143,40 | |
| FеS (к) | −100,40 | 60,29 | −100,80 | 50,54 | |
| Ќ¬r (г) | −36,38 | 198,58 | −53,43 | 29,14 | |
| Ќ—l (г) | −92,31 | 186,79 | −95,30 | 29,14 | |
| ЌF (г) | −273,30 | 173,67 | −275,41 | 29,14 | |
| Ќ2ќ (ж) | −285,83 | 69,95 | −273,23 | 75,30 | |
| Ќ2O (г) | −241,81 | 188,72 | −228,61 | 33,61 | |
| Ќ2S(г) | −20,60 | 205,70 | −33,50 | 33,44 | |
| ћgќ (к) | −601,49 | 27,07 | −569,27 | 37,20 | |
| NH3 (г) | −45,94 | 192,66 | −16,48 | 35,16 | |
| NЌ4NO3 (к) | −365,43 | 151,04 | −183,93 | 151,04 | |
| Nќ (г) | 91,26 | 210,64 | 87,58 | 29,58 | |
| NO2 (г) | 34,19 | 240,06 | 52,29 | 36,66 | |
| N2O (г) | 82,01 | 219,83 | 104,12 | 38,62 | |
| N2O4 (г) | 11,11 | 304,35 | 99,68 | 79,16 | |
| N2O5 (г) | 13,30 | 355,65 | 117,14 | 95,28 | |
| NaCl(к) | −411,12 | 72,13 | -384,13 | 50,81 | |
| Na2CO3(к) | -1130,80 | 138,80 | −1048,20 | 70,63 | |
| Niќ (к) | −239,74 | 37,99 | −211,60 | 44,31 | |
| NiS (к) | −92,88 | 67,38 | − | 54,68 | |
| NiSO4 (к) | −889,10 | 97,10 | −763,80 | 138,30 | |
| –bќ (к) | −219,28 | 66,11 | −189,10 | 45,81 | |
| –bO 2 (к) | −276,56 | 71,92 | −217,56 | 64,77 | |
| SO2 (г) | −296,90 | 248,07 | −300,21 | 39,87 | |
| SO3 (г) | −395,85 | 256,69 | −371,17 | 50,09 | |
| SiO2 (стекло) | −903,49 | 46,86 | −850,71 | 44,35 | |
| Snќ (к) | −285,98 | 56,48 | −265,88 | 44,35 | |
| Snќ2 (к) | −580,74 | 52,30 | −519,83 | 52,59 | |
| “iO2 (к) | −944,75 | 50,33 | −889,49 | 55,04 | |
| Znќ (к) | −348,11 | 43,51 | −318,10 | 40,25 | |
| ZnCO3(к) | −812,53 | 80,33 | −730,66 | 80,08 | |
| ZnS (к) | −205,40 | 65,30 | −200,70 | 45,52 | |
| ќрганические вещества | |||||
| ”глеводороды | |||||
| —Ќ4 (г) метан | −74,85 | 186,27 | −50,85 | 35,71 | |
| —2Ќ2(г) ацетилен | 226,75 | 200,82 | 209,21 | 43,93 | |
| —2Ќ4 (г) этилен | 52,30 | 219,45 | 68,14 | 43,56 | |
|
|
|
| ѕродолжение табл. 2 | |||||
| —2Ќ6 (г) этан | −84,67 | 229,49 | −32,93 | 52,64 | |
| —3Ќ 6 (г) пропилен | 20,41 | 266,94 | 62,70 | 63,89 | |
| —3Ќ8 (г) пропан | −103,85 | 269,91 | −23,53 | 73,51 | |
| —4Ќ]0 (г) н-бутан | −126,15 | 310,12 | −17,19 | 97,45 | |
| C5H12 (г) пентан | −146,40 | 348,40 | − | 122,60 | |
| —6Ќ6 (г) бензол | 82,93 | 269,20 | 129,68 | 81,67 | |
| —6Ќ6 (ж) бензол | 49,03 | 173,26 | 124,38 | 135,14 | |
| —6Ќ12 (ж) циклогексан | −156,23 | 204,35 | 26,60 | 156,48 | |
| —6Ќ12 (г) циклогексан | −124,14 | 298,24 | 31,70 | 106,27 | |
| —6Ќ14 (г) гексан | −167,20 | 386,80 | − | 146,70 | |
| —6Ќ14 (ж) гексан | −198,63 | 295,80 | −4,34 | 195,00 | |
| C7H16 (г) н-гептан | −187,80 | 425,30 | − | 224,70 | |
| C8H18 (г) н-октан | −208,40 | 463,70 | − | 254,00 | |
| ислородосодержащие соединени€ | |||||
| CH3OH (г) метиловый спирт | −201,20 | 329,70 | − | 81,80 | |
| CH3OH (ж) метиловый спирт | −238,30 | 126,80 | −166,23 | 123,31 | |
| CH3COOH (ж) уксусна€ кислота | −487,10 | 160,00 | −392,50 | 123,40 | |
| C2H5OH (г) этиловый спирт | −235,30 | 282,00 | − | 113,00 | |
| C2H5OH (ж) этиловый спирт | −277,60 | 160,70 | −174,80 | 113,00 | |
| C3H7OH (г) пропиловый спирт | −260,40 | 317,60 | − | 143,50 | |
| C6H12O6 (к) глюкоза | −1273,00 | 212,00 | −919,50 | 218,87 | |
| (CH3)2CO (г) ацетон | −216,40 | 294,90 | − | 125,00 | |
| * ѕримечание: -- градус ельвина. | |||||
“аблица 3
|
|
|
онстанты диссоциации ( д) слабых электролитов
в водных растворах (при 25OC)
| Ёлектролит | ”равнение диссоциации | д, моль/л |
| јзотиста€ кислота | HNO2  H+ + NO2− H+ + NO2−
| 4,0 Ј10-4 |
| Ѕорна€ кислота | H3BO3  H+ + H2BO3−
H2BO3− H+ + H2BO3−
H2BO3−  H+ + HBO32−
HBO32− H+ + HBO32−
HBO32−  H+ + BO33− H+ + BO33−
| 5,8 Ј10-10 1,8 Ј10-13 1,6 Ј10-14 |
| ¬ода | H2O  H+ + OH− H+ + OH−
| 1,8 Ј10-16 |
| ремниева€ кислота | H2SiO3  H+ + HSiO3−
HSiO3− H+ + HSiO3−
HSiO3−  H+ + SiO32− H+ + SiO32−
| 2,2 Ј10-10 1,6 Ј10-12 |
| ѕродолжение табл. 3 | ||
| ќртофосфорна€ кислота | H3PO4  H+ + H2PO4−
H2PO4− H+ + H2PO4−
H2PO4−  H+ + HPO42−
HPO42− H+ + HPO42−
HPO42−  H+ + PO43− H+ + PO43−
| 7,5 Ј10-3 6,2 Ј10-8 2,2Ј10-13 |
| —ерниста€ кислота | H2SO3  H+ + HSO3−
HSO3− H+ + HSO3−
HSO3−  H+ + SO32− H+ + SO32−
| 1,3 Ј10-2 5,0 Ј10-6 |
| —ероводородна€ кислота | H2S  H+ + HS−
HS− H+ + HS−
HS−  H+ + S2− H+ + S2−
| 5,7 Ј10-8 1,2 Ј10-15 |
| ўавелева€ кислота | H2C2O4  H+ + HC2O4−
HC2O4− H+ + HC2O4−
HC2O4−  H+ + C2O42− H+ + C2O42−
| 5,9 Ј10-2 6,4 Ј10-5 |
| —инильна€ кислота | HCN  H+ + CN− H+ + CN−
| 7,2 Ј10-10 |
| ”гольна€ кислота | H2CO3  H+ + HCO3−
HCO3− H+ + HCO3−
HCO3−  H+ + CO32− H+ + CO32−
| 4,3 Ј10-7 5,6 Ј10-11 |
| ”ксусна€ кислота | CH3COOH  H+ + CH3COO− H+ + CH3COO−
| 1,8 Ј10-5 |
| ‘тороводородна€ кислота | HF  H+ + F− H+ + F−
| 7,4 Ј10-4 |
| √идроксид алюмини€ | Al(OH)3  Al(OH)2+ + OH−
Al(OH)2+ Al(OH)2+ + OH−
Al(OH)2+  AlOH2+ + OH−
AlOH2+ AlOH2+ + OH−
AlOH2+  Al3+ + OH− Al3+ + OH−
| 7,4 Ј10-9 2,1 Ј10-9 1,4 Ј10-9 |
| √идроксид аммони€ | NH4OH  NH4+ + OH− NH4+ + OH−
| 1,8 Ј10-5 |
| √идроксид серебра | AgOH  Ag+ + OH− Ag+ + OH−
| 9,6 Ј10-4 |
| √идроксид кобальта | —o(OH)2  CoOH+ + OH−
CoOH+ CoOH+ + OH−
CoOH+  Co2+ + OH− Co2+ + OH−
| 7,9Ј10-5 7,9Ј10-6 |
| √идроксид оловаЦII | Sn(OH)2  SnOH+ + OH−
SnOH+ SnOH+ + OH−
SnOH+  Sn2+ + OH− Sn2+ + OH−
| 3,5 Ј10-10 1,3 Ј10-12 |
| √идроксид железаЦIII | Fe(OH)3  Fe(OH)2+ + OH−
Fe(OH)2+ Fe(OH)2+ + OH−
Fe(OH)2+  FeOH2+ + OH−
FeOH2+ FeOH2+ + OH−
FeOH2+  Fe3+ + OH− Fe3+ + OH−
| 4,8 Ј10-11 1,8 Ј10-11 1,4 Ј10-12 |
| √идроксид железаЦII | Fe(OH)2  FeOH+ + OH−
FeOH+ FeOH+ + OH−
FeOH+  Fe2+ + OH− Fe2+ + OH−
| − 1,3 Ј10-4 |
| √идроксид медиЦII | Cu(OH)2  Cu2+ + 2OH− Cu2+ + 2OH−
| 3,4 Ј10-7 |
| √идроксид никел€ | Ni(OH)2  Ni2++ 2OH− Ni2++ 2OH−
| 2,5 Ј10-5 |
| √идроксид свинца | Pb(OH)2  PbOH+ + OH−
PbOH+ PbOH+ + OH−
PbOH+  Pb2+ + OH− Pb2+ + OH−
| 9,5 Ј10-4 3,0 Ј10-8 |
| √идроксид хрома | —r(OH)3  Cr3+ + 3OH− Cr3+ + 3OH−
| 1,0 Ј10-10 |
| √идроксид цинка | Zn(OH)2  ZnOH+ + OH−
ZnOH+ ZnOH+ + OH−
ZnOH+  Zn2+ + OH− Zn2+ + OH−
| 5,0 Ј10-5 1,5 Ј10-9 |
“аблица 4
Cтепень диссоциации (a) электролитов
в водных растворах при 18 о—
| Ёлектролит (—эк = 0,1 моль экв/л) | ‘ормула | a, % |
| √идроксид аммони€ | NH4OH | 1,3 |
| ”ксусна€ кислота | CH3COOH | 1,3 |
| —ероводородна€ кислота | H2S | 0,07 |
| Ѕорна€ кислота | H3BO3 | 0,01 |
| —инильна€ кислота | HCN | 0,007 |
“аблица 5
ажуща€с€ степень диссоциации (a каж.) электролитов
в водных растворах при 18 о—
| Ёлектролит (—эк.= 0,1 моль экв/л) | ‘ормула вещества | a каж., % | Ёлектролит (—эк.= 0,1 моль экв/л) | ‘ормула вещества | a каж., % | |||
| ислоты | —оли | |||||||
| јзотна€ | HNO3 | ’лорид натри€ | NaCl | |||||
| —ол€на€ | HCl | |||||||
| —ерна€ | H2SO4 | ’лорид кали€ | KCl | |||||
| ‘осфорна€ | H3PO4 | ’лорид магни€ | MgCl2 | |||||
| —ерниста€ | H2SO3 | ’лорид алюмини€ | AlCl3 | |||||
| ѕлавикова€ | HF | —ульфат меди | CuSO4 | |||||
| ќсновани€ | ќсновани€ | |||||||
| √идроксид кали€ | KOH | √идроксид натри€ | NaOH | |||||
|
|
|
“аблица 7
ѕроизведение растворимости (ѕ–)
малорастворимых электролитов в воде (при t = 25ќ—)
Am+n Bn-m (тв)  n Am+(p-р)+m Bn−(p-р);
n Am+(p-р)+m Bn−(p-р);
ѕ–=[Am+]nЈ[Bn−]m, мольn+m/л n+m;
[Am+] Ц равновесна€ концентраци€ иона, моль/л.
| ¬ещество (электролит) | ѕ– | ¬ещество (электролит) | ѕ– |
| AgCl | 1,6 Ј10-10 | CaSO4 | 4,4 Ј10-5 |
| AgBr | 4,4 Ј10-13 | Cd(OH)2 | 4,3 Ј10-15 |
| AgJ | 9,7 Ј10-17 | Cu(OH)2 | 5,0 Ј10-19 |
| Ag2CO3 | 8,2 Ј10-12 | Cr(OH)3 | 6,7 Ј10-31 |
| Ag2SO4 | 4,4 Ј10-5 | Fe(OH)2 | 1,6 Ј10-15 |
| Ag2S | 1,6 Ј10-49 | Fe(OH)3 | 3,8 Ј10-38 |
| AgOH | 1,9 Ј10-8 | Mn(OH)2 | 2,3 Ј10-13 |
| Al(OH)3 | 5,1 Ј10-33 | Ni(OH)2 | 1,6 Ј10-14 |
| BaCO3 | 8,1 Ј10-9 | PbBr2 | 5,0Ј10-5 |
| BaSO4 | 1,1 Ј10-10 | PbCl2 | 2,1Ј10-5 |
| MgCO3 | 1,0 Ј10-5 | PbJ2 | 9,8 Ј10-9 |
| Mg(OH)2 | 5,5 Ј10-12 | Pb(OH)2 | 1,0 Ј10-15 |
| CaCO3 | 4,8 Ј10-9 | PbSO4 | 1,6 Ј10-8 |
| CaF2 | 4,0 Ј10-11 | PbS | 3,6 Ј10-29 |
| Ca(OH)2 | 6,3Ј10-6 | Zn(OH)2 | 1,3 Ј10-17 |
“аблица 8
ƒиэлектрическа€ проницаемость ( )
)
некоторых веществ
| ¬ещество | 
| ¬ещество | 
|
| ¬акуум | 1,0 | Ётиловый спирт | 26,0 |
| ¬ода дистиллированна€ | 81,0 | Ѕензол | 2,6 |
“аблица 9
риоскопические ( к) и эбулиоскопические ( э) константы
некоторых растворителей (кгЈ *Јмоль−1)
| –астворитель | Ё | |
| ¬ода | 1,86 | 0,52 |
| Ѕензол | 5,10 | 2,57 |
| јнилин | 5,87 | 3,69 |
| Ётиловый спирт | 1,99 | 1,16 |
| јцетон | 2,40 | 1,50 |
| ’лороформ | 4,90 | 3,89 |
| ”ксусна€ кислота | 3,90 | 3,09 |
| „етыреххлористый углерод | 29,8 | 5,02 |
| ƒиоксан | 4,63 | 3,01 |
| ÷иклогексан | 20,80 | 2,92 |
| Ќитробензол | 6,88 | 5,20 |
* ѕримечание: − градус ельвина.
“аблица 10
¬одородный показатель растворов (рЌ)
| онцентраци€ ионов | ўелочна€ среда
(усиление щелочных свойств)

| Ќейтраль-на€ среда | ислотна€ среда
(усиление кислотных свойств)

| ||||||
| [OH− ] | 1(100) | 10−2 | 10−4 | 10−6 | 10−7 | 10−8 | 10−10 | 10−12 | 10−14 |
| [H+] | 10−14 | 10−12 | 10−10 | 10−8 | 10−7 | 10−6 | 10−4 | 10−2 | 100 |
| рH |
“аблица 11
—тандартные окислительно-восстановительные потенциалы (≈о298)
¬ водных растворах
| ќкислительно-восстановительна€ пара (ќ¬ѕ) | ”равнение реакции восстановлени€ | ≈о298, ¬ |
| —оединени€ азота | ||
| (Ќ+) NO-3/NO2 (Ќ2ќ) | NO−3 + 2H+ +  ЂNO2+ H2O ЂNO2+ H2O
| +0,78 |
| (Ќ+) NO-3/HNO2 (Ќ2ќ) | NO−3 + 3H+ + 2  ЂHNO2+ H2O ЂHNO2+ H2O
| +0,93 |
| (Ќ+) NO-3/NO-2 (Ќ2ќ) | NO−3 + 2H+ + 2  ЂNO2− + H2O ЂNO2− + H2O
| +0,94 |
| (Ќ+) NO-3/NO (Ќ2ќ) | NO−3 + 4H+ + 3  ЂNO+ 2H2O ЂNO+ 2H2O
| +0,96 |
| (Ќ+) NO-2 /N2 (ќЌ-) | NO−2 + 2H+ + 2  ЂN2 + 2OH− ЂN2 + 2OH−
| +0,99 |
| (Ќ+) 2NO2/N2 (Ќ2ќ) | 2NO2 + 8H+ + 8  ЂN2 + 4H2O ЂN2 + 4H2O
| +1,36 |
| (Ќ+) 2HNO2/N2 (Ќ2ќ) | 2HNO2 + 6H+ + 6  ЂN2 + 4H2O ЂN2 + 4H2O
| +1,45 |
| (Ќ+) 2NO/N2 (Ќ2ќ) | 2NO + 4H+ + 4  ЂN2 + 2H2O ЂN2 + 2H2O
| +1,68 |
| —оединени€ бора | ||
| (Ќ+) H3BO3/B (Ќ2ќ) | H3BO3 + 3H+ + 3  ЂB + 3H2O ЂB + 3H2O
| −0,71 |
| —оединени€ брома | ||
| BrO-3/Br- (OH-) | BrO−3 + 3H2O + 6  ЂBr− + 6OH− ЂBr− + 6OH−
| +0,60 |
| Br2/2Br- | Br2 + 2  Ђ2Br− Ђ2Br−
| +1,09 |
| (Ќ+) BrO-3/Br- (Ќ2ќ) | BrO−3 + 6H+ + 6  ЂBr− + 3H2O ЂBr− + 3H2O
| +1,44 |
| (Ќ+) 2BrO-3/Br2 (Ќ2ќ) | 2BrO−3 + 12H+ + 10  ЂBr2 + 6H2O ЂBr2 + 6H2O
| +1,52 |
| —оединени€ йода | ||
| (Ќ2ќ) JO-3/J- (OH-) | JO−3 + 6H2O + 6  ЂJ− + 6OH− ЂJ− + 6OH−
| +0,26 |
| J2/2J- | J2 + 2  Ђ2J− Ђ2J−
| +0,53 |
| (H+) JO-3/J- (Ќ2ќ) | JO−3 + 6H+ + 6  ЂJ− + 3H2O ЂJ− + 3H2O
| +1,09 |
| (H+) 2JO-3/J2 (Ќ2ќ) | 2JO−3 + 12H+ + 10  ЂJ2 + 6H2O ЂJ2 + 6H2O
| +1,20 |
| —оединени€ кислорода | ||
| (Ќ2ќ) H2O/H2 (OH-) | 2H2O + 2  ЂH2 ЂH2 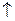 + 2OH− + 2OH−
| −0,83 |
| (Ќ2ќ) O2/H2O2 (OH-) | O2 + 2H2O + 2  ЂH2O2 + 2OH− ЂH2O2 + 2OH−
| −0,08 |
| (H+) O2/H2O2 | O2 + 2H+ + 2  ЂH2O2 ЂH2O2
| +0,68 |
| (H+) O2/2H2O (Ќ2ќ) | O2 + 4H+ + 4  Ђ2H2O Ђ2H2O
| +1,23 |
| (H+) H2O2/2H2O (Ќ2ќ) | H2O2 + 2H+ + 2  Ђ2H2O Ђ2H2O
| +1,78 |
| —оединени€ марганца | ||
| Mn2+/Mn | Mn2+ + 2  ЂMn ЂMn
| −1,18 |
| MnO4-/ MnO42- | MnO4- +  ЂMnO42− ЂMnO42−
| +0,56 |
| MnO4-/ MnO2(OH-) | MnO4− + 2H2O + 3  ЂMnO2 + 4OH− ЂMnO2 + 4OH−
| +0,57 |
| MnO42- / MnO2(OH-) | MnO42- + 2H2O + 2 ЂMnO2 + 4OH- ЂMnO2 + 4OH-
| +0,58 |
|
|
|
| ѕродолжение табл. 11 | |||
| ќкислительно-восстановительна€ пара (ќ¬ѕ) | ”равнение реакции восстановлени€ | ≈о298, ¬ | |
| (H+) MnO2/Mn2+ | MnO2 + 4H+ + 2  ЂMn2+ + 2H2O ЂMn2+ + 2H2O
| +1,24 | |
| (H+)MnO4-/ Mn2+ (Ќ2ќ) | MnO4− + 8H+ + 5  ЂMn2+ + 4H2O ЂMn2+ + 4H2O
| +1,53 | |
| (H+)MnO4-/ ћnO2(Ќ2ќ) | MnO4− + 4H+ + 3  ЂMnO2 + 2H2O ЂMnO2 + 2H2O
| +1,73 | |
| —оединени€ свинца | |||
| Pb2+/Pb | Pb2+ + 2  ЂPb ЂPb
| −0,13 | |
| (H+) PbO2/ Pb2+ (Ќ2ќ) | PbO2+ 4H+ +SO42− +2  ЂPbSO4+ H2O ЂPbSO4+ H2O
| +1,46 | |
| Pb4+/ Pb2+ | Pb4++ 2  ЂPb2+ ЂPb2+
| +1,69 | |
| —оединени€ серы | |||
| (Ќ2ќ)SO42-/ SO32-(OH-) | SO42−+ H2O + 2  ЂSO32−+ 2OH− ЂSO32−+ 2OH−
| −0,93 | |
| (H+) SO42-/ S2- (Ќ2ќ) | SO42− + 8H+ + 8  ЂS2−+ 4H2O ЂS2−+ 4H2O
| −0,15 | |
| (H+) S/H2S | S + 2H+ + 2  ЂH2S ЂH2S
| +0,17 | |
| (H+)SO42-/ SO32- (Ќ2ќ) | SO42− + 2H+ + 2  ЂSO32− + H2O ЂSO32− + H2O
| +0,20 | |
| —оединени€ фосфора | |||
| (H+) PO43- /P (Ќ2ќ) | PO43- + 8H+ + 5  ЂP + 4H2O ЂP + 4H2O
| −0,38 | |
| (H+) PO43- /PO33- (Ќ2ќ) | PO43− + 2H+ + 2  ЂPO33− + H2O ЂPO33− + H2O
| −0,28 | |
| (H+) P/PH3 | – + 3Ќ+ + 3  Ђ–Ќ3 Ђ–Ќ3
| −0,04 | |
| —оединени€ хлора | |||
| Cl2/2Cl- | Cl2 + 2  Ђ2Cl− Ђ2Cl−
| +1,36 | |
| (H+) ClO3-/Cl- (Ќ2ќ) | ClO3− + 6H+ + 6  ЂCl− + 3H2O ЂCl− + 3H2O
| +1,45 | |
| (H+) 2ClO3-/Cl2 (Ќ2ќ) | 2ClO3− + 12H+ + 10  ЂCl2 + 6H2O ЂCl2 + 6H2O
| +1,47 | |
| (H+) 2ClO-/Cl2 (Ќ2ќ) | 2ClO− + 4H+ + 2  ЂCl2 + 2H2O ЂCl2 + 2H2O
| +1,63 | |
| —оединени€ хрома | |||
| Cr3+/Cr | Cr3+ + 3  ЂCr ЂCr
| −0,74 | |
| (Ќ2ќ)CrO42- /Cr(OH)3 | CrO42− + 4H2O + 3 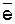 ЂCr(OH)3 + 5OH− ЂCr(OH)3 + 5OH−
| −0,13 | |
| (H+) CrO42-/Cr (Ќ2ќ) | CrO42- + 8H+ + 6 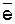 ЂCr + 4H2O ЂCr + 4H2O
| +0,37 | |
| (H+)CrO42-/CrO2-(Ќ2ќ) | CrO42− + 4H+ + 3  ЂCrO2− + 2H2O ЂCrO2− + 2H2O
| +0,95 | |
| (H+)Cr2O72-/2Cr3+(Ќ2ќ) | Cr2O72− + 14H+ + 6  Ђ2Cr 3++ 7H2O Ђ2Cr 3++ 7H2O
| +1,33 | |
| (H+) CrO42-/Cr3+ (Ќ2ќ) | CrO42−+ 8H+ + 3  ЂCr3++ 4H2O ЂCr3++ 4H2O
| +1,48 | |
| ƒругие соединени€ | |||
| (H+) As/AsH3 | As + 3H+ + 3  ЂAsH3 ЂAsH3
| −0,54 | |
| Sn2+/Sn | Sn2+ + 2  ЂSn ЂSn
| −0,14 | |
| Fe3+/Fe2+ | Fe3+ +  ЂFe2+ ЂFe2+
| +0,77 | |
| F2/2F- | F2 + 2  Ђ2F− Ђ2F−
| +2,85 | |
“аблица 12
—тандартные потенциалы металлических электродов
(р€д напр€жений)
| Ёлектрод | Ёлектродный процесс | ≈о, ¬ 
|
| Li+/ Li | Li+ + ē  Li Li
| −3,040 |
| Rb+/ Rb | Rb+ + ē  Rb Rb
| −2,925 |
| +/ | ++ ē 
| −2,925 |
| Cs+/ Cs | Cs+ + ē  Cs Cs
| −2,923 |
| Ba2+/ Ba | Ba2+ + 2ē  Ba Ba
| −2,906 |
| Ca2+/ Ca | Ca2+ + 2ē  Ca Ca
| −2,866 |
| Na+/ Na | Na+ + ē  Na Na
| −2,714 |
| Mg2+/ Mg | Mg2+ + 2ē  Mg Mg
| −2,363 |
| Be2+/ Be | Be2++ 2ē  ¬е ¬е
| −1,847 |
| Al3+/ Al | Al3+ + 3ē  Al Al
| −1,662 |
| Ti2+/ Ti | Ti2+ + 2ē  Ti Ti
| −1,628 |
| Mn2+/ Mn | Mn2+ + 2ē  Mn Mn
| −1,180 |
| Zn2+/ Zn | Zn2+ + 2ē  Zn Zn
| −0,763 |
| Cr3+/ Cr | Cr3+ + 3ē  Cr Cr
| −0,744 |
| Fe2+/ Fe | Fe2+ + 2ē  Fe Fe
| −0,440 |
| Cd2+/ Cd | Cd2+ + 2ē  Cd Cd
| −0,403 |
| Co2+/ Co | Co2+ + 2ē  Co Co
| −0,277 |
| Ni2+/ Ni | Ni2+ + 2ē  Ni Ni
| −0,250 |
| Mo3+/Mo | Mo3+ + 3ē  Mo Mo
| −0,200 |
| Sn2+/ Sn | Sn2+ + 2ē  Sn Sn
| −0,136 |
| Pb2+/ Pb | Pb2+ + 2ē  Pb Pb
| −0,126 |
| Fe3+/ Fe | Fe3+ + 3ē  Fe Fe
| −0,036 |
| 2H+/ H2 | 2H+ + 2ē  H2 H2
| +0,000 |
| Sb2+/ Sb | Sb2+ + 2ē  Sb Sb
| +0,200 |
| Bi3+/ Bi | Bi3+ + 3ē  Bi Bi
| +0,220 |
| Cu2+/ Cu | Cu2+ + 2ē  Cu Cu
| +0,337 |
| Hg22+/ 2Hg | Hg22++ 2ē  2Hg 2Hg
| +0,790 |
| Ag+/ Ag | Ag+ +ē  Ag Ag
| +0,799 |
| Hg2+/ Hg | Hg2+ +2ē  Hg Hg
| +0,854 |
| Pd2+/ Pd | Pd2+ + 2ē  Pd Pd
| +0,987 |
| Pt2+/ Pt | Pt2+ + 2ē  Pt Pt
| +1,190 |
| Au3+/ Au | Au3+ + 3ē  Au Au
| +1,498 |
¬ќѕ–ќ—џ
дл€ подготовки к зачету по курсу Ђ’ими€ї






