–аствор Ц это гомогенна€ термодинамически устойчива€ во времени система, состо€ща€ минимум из двух компонентов и продуктов их взаимодействи€.
Ћюбой раствор состоит из двух веществ Ц растворител€ и растворЄнного вещества, причЄм растворител€ в растворе всегда содержитс€ в большем количестве, чем растворЄнного вещества.
1. ћј——ќ¬јя ƒќЋя –ј—“¬ќ–®ЌЌќ√ќ ¬≈ў≈—“¬ј
ћассова€ дол€ растворЄнного вещества (ωр.в., %) показывает сколько граммов растворЄнного вещества содержитс€ в 100 граммах раствора.



ω % (р.в.) = m(р.в.) *100 % ,
m (р Ц ра) (1)
 где m (р Ц ра) = m (р.в.) + m (р Ц л€)
где m (р Ц ра) = m (р.в.) + m (р Ц л€)
ћежду массой раствора, объЄмом и плотностью существует соотношение, т.к.
 (г/см3) или (г/мл), то т(р Ц ра) = ρ*V,
(г/см3) или (г/мл), то т(р Ц ра) = ρ*V,
|
а) приготовление растворов с заданной массовой долей растворЄнного вещества ω3 смешением двух растворов: с большей ω1 и меньшей ω2 массовой долей того же самого вещества.
ѕри этом требуетс€ найти массу более концентрированного (m1) и менее концентрированного (m2) растворов. »спользуетс€ Ђправило крестаї, суть которого отражает схема:
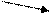
 ω1 m1 = ω3 Ц ω2
ω1 m1 = ω3 Ц ω2
ω3

 ω2 m2 = ω1 Ц ω3
ω2 m2 = ω1 Ц ω3
ѕусть, например, требуетс€ приготовить раствор с массовой долей 10% массой 70 г из 5 % и 40 %.
ѕодставим эти числа в приведЄнную ниже схему. ѕолучаем:
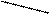
 40 m1 = 10 Ц 5 = 5
40 m1 = 10 Ц 5 = 5

 10
10
5 m2 = 40 Ц 10 = 30
ќткуда 
Ќапример, вз€в 10 г раствора ω = 40% и 60 г раствора с ω = 5%, получим 70г. раствора с ω = 10%.
б) приготовление растворов путЄм разбавлени€ концентрированных растворов.
“ак как при разбавлении растворов масса (р.в.) неизменна (m*ω) = const, то существует обратно пропорциональна€ зависимость между отношением масс растворов и их массовыми дол€ми
|
2. ћќЋя–Ќјя ќЌ÷≈Ќ“–ј÷»я –ј—“¬ќ–ј
ћол€рна€ концентраци€ раствора (—м, моль/л.) показывает число моль растворЄнного вещества, содержащегос€ в 1 л раствора.
—ћ = n моль, если V в мл, то —ћ = n*1000 ,
V л. V мл
где n = m (р.в.),
ћ (р.в.)
|
(2)
ћол€рным называют раствор, в 1 л которого содержитс€ 1 моль растворЄнного вещества.
Ёквивалентом (Ё) называетс€ такое количество элемента или вещества (по массе), которое без остатка взаимодействует с 1 моль атомов или ионов водорода, с 1 моль электронов в окислительно-восстановительной реакции или 1 моль любого вещества в любой химической реакции.
Ёквивалент Ц величина переменна€ дл€ одного и того же вещества и рассчитываетс€ по уравнению реакции или по формуле:
|
f (кислоты) = 1 ,
число ионов (Ќ+), участвующих в х.р.
f (основани€) = 1 ,
число ионов (ќЌ‾ ), участвующих в х.р.
f (кислоты) = 1 ,
число ионов (ћеn+) * n
|
|
|
где n- зар€д иона металла.
3. «ј ќЌ Ё ¬»¬јЋ≈Ќ“ќ¬
|
¬ид:, где —н1 и —н2 Ц мол€рна€ концентраци€ эквивалентов анализируемого и стандартного (концентраци€ которого точно известна) растворов, а V1 и V2 Ц соответственно их объЄмы.
4. ѕ≈–≈¬ќƒ ќƒЌќ√ќ —ѕќ—ќЅј ¬џ–ј∆≈Ќ»я ќЌ÷≈Ќ“–ј÷»»
–ј—“¬ќ–ј ¬ ƒ–”√ќ…
Ќужно помнить, что масса растворЄнного вещества Ц неизменна€ и обща€ дл€ формул любого способа выражени€ концентрации величина, следовательно, при помощи еЄ и производ€т перевод.
а) перевод массовой доли растворЄнного вещества в мол€рную концентрацию.
|
б) перевод массовой доли растворЄнного вещества в мол€рную концентрацию эквивалента.
|
–≈Ў≈Ќ»≈ “»ѕќ¬џ’ «јƒј„ ѕќ “≈ћ≈: Ђ—ѕќ—ќЅџ ¬џ–ј∆≈Ќ»…
ќЌ÷≈Ќ“–ј÷»… –ј—“¬ќ–ќ¬ї
«адача є 1.
120 мл. раствора с массовой долей NaCl 16 % и плотностью 1,16 г/см3 прилили воду объЄмом 80 мл. –ассчитайте ω%, —м, —н полученного раствора.
 ƒано: –ешение:
ƒано: –ешение:
Vр-ра = 120 мл 1. Ќаходим массу 16 % раствора, зна€,
ρ1 (р-ра) = 1,16 г/см3 что
 ω1 %(NaCl) = 16 % ρ = m , m = ρ *V,
ω1 %(NaCl) = 16 % ρ = m , m = ρ *V,
V(H2O) = 80 мл V
ρ2 = 1,071 г/см3m (р-ра) = 120 *1,16 = 139,2 г.
ћ(NaCl) = 58,5 г/моль 2. »з формулы: ω % (р.в.) = m (р.в.) *100 % ,
 ω2 %=? m(р Ц ра)
ω2 %=? m(р Ц ра)
—н =? определ€ем m (р.в.) в 16 % растворе:
—м =? m (р.в.) = ω1% *m (р-ра) ,
100%
m (р.в.) = 16 *139,2 = 22,27 г
100 %
3. ѕосле прибавлени€ воды масса раствора изменилась, т.к. ρ (Ќ2ќ) = 1 г/см3, то m (р-ра) = 139,2 + 80 = 219,2 г.
4. Ќаходим массовую долю растворЄнного вещества в новом растворе:
ω2% = 
5. ќпредел€ем —м, зна€ формулу перерасчета:
—м = 
—м = 
6. ќпредел€ем —н, но т.к. f(NaCl) = 1 и Ё = ћ (NaCl), то
—н = —м = 1,86 н.
ќтвет: ω2% = 10,16%, —м = 1,86 ћ, —н = 1,86 н.
«адача є2
Ќа нейтрализацию 20 мл 0,2 н раствора сол€ной кислоты израсходовано 15 мл гидроксида кали€. –ассчитайте —н ( ќЌ).
 ƒано: –ешение:
ƒано: –ешение:
V(HCl) = 20 мл 1. «на€ закон эквивалентов,
C(HCl) = 0,2 н —н (к-ты) *V(к-ты) = —н (щ) *V(щ),
 V(KOH) = 15 мл находим —( ќЌ)
V(KOH) = 15 мл находим —( ќЌ)
—н ( ќЌ) = 
ќтвет: —н ( ќЌ) = 0,26 н.






