»зучение данной темы по рекомендуемой литературе.
–≈ ќћ≈Ќƒ”≈ћјя Ћ»“≈–ј“”–ј
ќсновна€ литература:
1.јхметов, Ќ.—. ќбща€ и неорганическа€ хими€: учебник /Ќ.—. јхметов. - ћ.: ¬ысша€ школа, 2005.
2. ’омченко, √.ѕ. Ќеорганическа€ хими€: учебник дл€ с.-х. вузов / √.ѕ. ’омченко, ». . ÷итович. Ц —ѕб.: »“ √ранит: ќќќ »ѕ Ђ ќ—“јї, 2009. Ц 464 с.
3. ÷итович, ». . урс аналитической химии: учебник / ». . ÷итович.- —ѕб.: Ћань, 2007.
4. —аргаев, ѕ.ћ. Ќеорганическа€ хими€ [Ёлектронный ресурс]/ ѕ.ћ. —аргаев.- —ѕб.: Ћань, 2013.- –ежим доступа: http://www.Lanbook.ru, свободный.
ƒополнительна€ литература:
1. √линка, Ќ.Ћ. «адачи и упражнени€ по общей химии: учебное пособие/ Ќ.Ћ. √линка. - ћ.: но–ус, 2012.- 240 с.
2. √линка, Ќ.Ћ. ќбща€ хими€: учебное пособие / Ќ.Ћ. √линка.- ћ.: ёрайт, 2010. Ц 886 с.
3. ≈ршов, ё.ј. ќбща€ хими€. Ѕиофизическа€ хими€. ’ими€ биогенных элементов: учебник / ё.ј. ≈ршов [и д.р.]- ћ.: ¬ысша€ школа, 2009.Ц 560 с.
4. елина, ё. ќбща€ и неорганическа€ хими€ в таблицах и схемах: учебное пособие/ ё. елина, Ќ. ¬. Ѕезручко.- ћ.: ‘еникс, 2008.
5. н€зев, ƒ. ј. Ќеорганическа€ хими€: учебник/ ƒ. ј. н€зев, —. Ќ. —марыгин.- ћ.: ёрайт, 2012. Ц 886 с.
–есурсы интернета:
1. ќткрытый колледж http://www.college.ru/chemistry/index/php.
2. –убрикон: энциклопедии, словари, справочники http://www.en.edu.ru/db/msg/7030/sp/3368/2794/2761/
3. »нформационно-справочные и образовательные системы по химии http://www.chemnet.ru/, http://www.alhimik.ru/, http://www.chemport.ru/,http://www.chemworld.narod.ru/
ѕриложени≈
ѕриложение 1
ќсновные физические посто€нные
| ѕосто€нна€ | ќбозначение | «начение |
| „исло јвогардо | NA | 6,02 Ј 1023 моль-1 |
| «ар€д электрона | e | 1,60 Ј 10-19 л |
| ћол€рный объем газа | Vm | 22,4 л/моль* |
| ѕосто€нна€ Ѕольцмана | k | 1,38 Ј 10-23 ƒж/ |
| ѕосто€нна€ ѕланка | h | 6,63 Ј 10-34 ƒжЈс |
| ѕосто€нна€ ‘араде€ | F | 9,65 Ј 104 л/моль |
| ”ниверсальна€ газова€ посто€нна€ | R | 8,31 ƒж/(мольЈ ) |
* ѕри нормальных услови€х.
ѕриложение 2
√рупповые названи€ химических элементов
| јктиноиды | Ac, Th, Pa, U, Np, Pu, Am, Cm, Bk, Cf, Es, Fm, Md, No, Lr |
| Ѕлагородные газы | He, Ne, Ar, Kr, Xe, Rn |
| √алогены | F, Cl, Br, I, At |
| Ћантаноиды | La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu |
| ’алькогены | O, S, Se, Te, Po |
| —емейство железа | Fe, Co, Ni |
| —емейство платины | Ru, Rh, Pd, Os, Ir, Pt |
| ўелочные металлы | Li, Na, K, Rb, Cs, Fr |
| ўелочноземельные металлы | Ca, Sr, Ba, Ra |
ѕриложение 3
ћножители и приставки дл€ образовани€ дес€тичных кратных и дольных единиц
| ћножитель | ѕриставка | ќбозначение | ћножитель | ѕриставка | ќбозначение | |
| 1012 | тера | “ | 10-1 | деци | д | |
| 109 | гига | √ | 10-2 | санти | с | |
| 106 | мега | ћ | 10-3 | милли | м | |
| 103 | кило | к | 10-6 | микро | мк | |
| 102 | гекто | г | 10-9 | нано | н | |
| 101 | дека | да | 10-10 | пико | п |
ѕриложение 4
“ермодинамические константы некоторых веществ
|
|
|
| ¬ещество | 
| 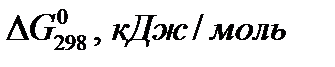
| 
| ||||
| Al (к) | 28,31 | ||||||
| Al2O3 (к) | Ц 1675,0 | Ц 1576,4 | 50,94 | ||||
| Ц 981,57 | Ц 944,75 | 199,4 | ||||
| C (алмаз) | 1,897 | 2,866 | 2,38 | ||||
| — (графит) | 5,74 | ||||||
| CO (г) | Ц 110,5 | Ц 137,27 | 197,4 | ||||
| CO2(г) | Ц 393,51 | Ц 394,38 | 213,6 | ||||
| COCl2(г) | Ц 223,0 | Ц210,5 | 289,2 | ||||
| CS2 (г) | Ц 115,3 | 65,1 | 237,8 | ||||
| CS2 (ж) | Ц 87,8 | 63,6 | 151,0 | ||||
| C2H2 (г) | 226,75 | 209,2 | 200,8 | ||||
| C2H4 (г) | 52,28 | 68,12 | 219,4 | ||||
| CH4 (г) | Ц 74,85 | Ц 50,79 | 186,19 | ||||
| C2H6 (г) | Ц 84,67 | Ц 32,89 | 229,5 | ||||
| C6H6 (ж) | 49,04 | 124,50 | 173,2 | ||||
| CH3 OH (ж) | Ц 238,7 | Ц 166,31 | 126,7 | ||||
| C2H5 OH (ж) | Ц 227,6 | Ц 174,77 | 160,7 | ||||
| Ca (к) | 41,62 | ||||||
| Caќ (к) | Ц 635,1 | Ц 604,2 | 39,7 | ||||
| Ca(OH)2 (к) | Ц 986,2 | Ц 896,76 | 83,4 | ||||
| CaCќ3(к) | Ц 1206,0 | Ц 1128,8 | 92,9 | ||||
| Cl2 (г) | 223,0 | ||||||
| HCl (г) | Ц 92,30 | Ц 95,27 | 186,7 | ||||
| HCl (ж) | Ц 167,5 | Ц 131,2 | 55,2 | ||||
| Cu (к) | 33,3 | ||||||
| Cu2O (к) | Ц 167,36 | Ц 146,36 | 93,93 | ||||
| CuO (к) | Ц 165,3 | Ц 127,19 | 42,64 | ||||
| Cu2S (к) | Ц 82,01 | Ц 86,19 | 119,24 | ||||
| Fe (к) | 27,15 | ||||||
| FeO (к) | Ц 263,68 | Ц 244,35 | 58,79 | ||||
| FeCl2(к) | Ц 341,0 | Ц 302,08 | 119,66 | ||||
| Fe2O3(к) | Ц 821,32 | 740,99 | 89,96 | ||||
| Fe3O4(к) | Ц 1117,1 | Ц 1015,5 | 146,2 | ||||
| Ge (к) | 31,09 | ||||||
| GeO (к) | Ц 305,4 | Ц 276,1 | 50,2 | ||||
| GeO2 (к) | Ц 539,74 | Ц 531,4 | 52,30 | ||||
| H2 (г) | 130,6 | ||||||
| H2O (г) | Ц 241,84 | Ц 228,8 | 188,74 | ||||
| H2O (ж) | 285,84 | Ц 237,5 | 69,96 | ||||
| N2 (г) | 191,5 | ||||||
| N2ќ (г) | 81,55 | 103,6 | 220,0 | ||||
| Nќ (г) | 90,37 | 86,69 | 210,62 | ||||
| Nќ2(г) | 33,89 | 51,84 | 240,45 | ||||
| NЌ3(г) | Ц 46,19 | Ц 16,64 | 192,5 | ||||
| NЌ4Cl (к) | Ц 315,39 | Ц 343,64 | 94,56 | ||||
| NЌ4OH(ж) | Ц 366,69 | Ц 263,8 | 179,9 | ||||
| NaOH (к) | Ц 426,6 | Ц 377,0 | 64,18 | ||||
| NaCl (к) | Ц 410,9 | Ц 384,0 | 72,36 | ||||
| O2 (г) | 205,03 | ||||||
| Pb (к) | 64,9 | ||||||
| PbO (к) | Ц 217,86 | Ц 188,49 | 67,4 | ||||
| PbO2 (к) | Ц 276,6 | Ц 218,99 | 76,44 | ||||
| SO2 (г) | Ц 296,9 | Ц 300,37 | 248,1 | ||||
| H2S (г) | Ц 20,15 | Ц 33,02 | 205,64 | ||||
| H2S (ж) | Ц 39,33 | Ц 27,36 | 122,2 | ||||
| Sn (к) | 51,55 | ||||||
| SnO (к) | Ц 286,0 | Ц 257,32 | 56,74 | ||||
| SnO2 (к) | Ц 580,8 | Ц 519,65 | 52,34 | ||||
| Ti (к) | 30,6 | ||||||
| TiO2 (к) | Ц 938,6 | Ц 852,2 | 49,92 |
ѕриложение 5
оэффициенты активности ионов при различных ионных силах раствора
| »онна€ сила раствора | «ар€д иона z | »онна€ сила раствора IC | «ар€д иона z | ||||
| ±1 | ±2 | ±3 | ±1 | ±2 | ±3 | ||
| 0,05 0,1 0,2 | 0,84 0,81 0,80 | 0,50 0,44 0,41 | 0,21 0,16 0,14 | 0,3 0,4 0,5 | 0,81 0,82 0,84 | 0,42 0,45 0,50 | 0,14 0,17 0,21 |
ѕриложение 6
онстанты диссоциации некоторых слабых электролитов
в водных растворах, 
| Ёлектролит | д | 
| |
| HNO2 | 4.0Ј10 Ц 4 | 3.40 | |
| NH4OH | 1.8Ј10 Ц 5 | 4.75 | |
| H2SiO3 | K1 | 2.2Ј10 Ц 10 | 9.66 |
| K2 | 1.6Ј10 Ц 12 | 11.80 | |
| H2SO4 | K2 | 1.2Ј10 Ц 2 | 1.92 |
| H2SO3 | K1 | 1.6Ј10 Ц 2 | 1.80 |
| K2 | 6.3Ј10 Ц 8 | 7.21 | |
| H2S | K1 | 6.0Ј10 Ц 8 | 7.22 |
| K2 | 1.0Ј10 Ц 14 | 14.0 | |
| H2CO3 | K1 | 4.5Ј10 Ц 7 | 6.35 |
| K2 | 4.7Ј10 Ц 11 | 10.33 | |
| CH3COOH | 1.8Ј10 Ц 5 | 4.75 | |
| H3PO4 | K1 | 7.5Ј10 Ц 3 | 2.12 |
| K2 | 6.3Ј10 Ц 8 | 7.20 | |
| K3 | 1.3Ј10 Ц 12 | 11.89 | |
| HF | 6.6Ј10 Ц 4 | 3.18 | |
| HCN | 7.9Ј10 Ц 10 | 9.10 |
|
|
|
ѕриложение 7
—тепень диссоциации кислот, щелочей и солей
в водных растворах,  .
.
| Ёлектролит и его ионы | α % | |
| 1Ќ | 0,1Ќ | |
| ислоты | ||
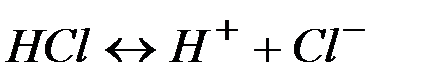
| ||

| ||

| ||

| - | 0,07 |

| - | 0,17 |

| 0,4 | 1,3 |

| ||
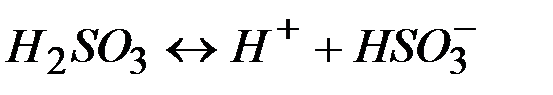
| - | |

| - | 0,01 |
| ќсновани€ | ||

| ||

| ||

| - | |
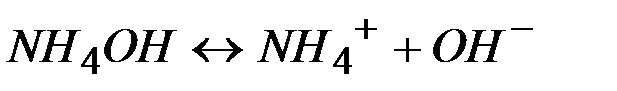
| 0,4 | 1,3 |
| —оли | ||
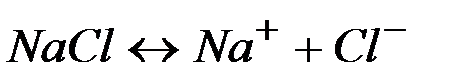
| ||
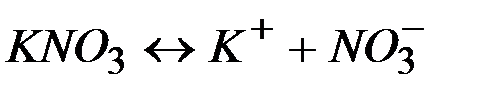
| ||

| ||

|
ѕриложение 8
»онное произведение воды (Kw) при разных значени€х температуры
| t, 0C | Kw Ј 10-14 | t, 0C | Kw Ј 10-14 | |
| 0,11 | 2,40 | |||
| 0,17 | 2,95 | |||
| 0,30 | 5,50 | |||
| 0,46 | 9,55 | |||
| 0,69 | 15,8 | |||
| 1,00 | 25,8 | |||
| 1,48 | 38,8 | |||
| 2,09 | 55,0 |
ѕриложение 9
ѕлотности водных растворов некоторых растворов при 200C
| ¬ещество | ћасса % | |||||||||
| 1н. | ||||||||||
| NaCl | 1,01 | 1,04 | 1,07 | 1,12 | 1,15 | 1,19 | 1,04 | |||
| KCl | 1,01 | 1,04 | 1,06 | 1,10 | 1,13 | 1,16 | 1,04 | |||
| Na2SO4 | 1,02 | 1,05 | 1,09 | 1,15 | 1,06 | |||||
| K2SO4 | 1,02 | 1,05 | 1,08 | 1,07 | ||||||
| MgSO4 | 1,02 | 1,06 | 1,10 | 1,17 | 1,22 | 1,27 | ||||
| NaNO3 | 1,03 | 1,07 | 1,14 | 1,23 | 1,05 | |||||
| BaCl2 | 1,04 | 1,09 | 1,20 | 1,27 | 1,09 | |||||
| NH4NO3 | 1,01 | 1,04 | 1,08 | 1,13 | 1,18 | 1,23 | ||||
| (NH4) 2SO4 | 1,01 | 1,06 | 1,12 | 1,17 | 1,23 | |||||
| NH4Cl | 1,00 | 1,03 | 1,06 | 1,07 | ||||||
| FeSO4 | 1,02 | 1,10 | 1,17 | 1,20 | ||||||
| Al2(SO4)3 | 1,02 | 1,10 | 1,23 | 1,31 |
ѕриложение 10
ѕроизведени€ растворимости некоторых малорастворимых электролитов при 25о—.
| Ёлектролит | ѕ– (KS) | Ёлектролит | ѕ– (KS) |
| AgBr | 6,3×10-13 | CdS | 7,9×10-27 |
| AgBrO3 | 5,5.10-5 | CoS (18oC) | 2,0.10-27 |
| AgCl | 1,56×10-10 | Cu(OH)2 | 5,6.10-20 |
| Ag2CrO4 | 1,1×10-12 | CuS | 4,0×10-38 |
| AgI | 1,5×10-16 | Fe(OH)2 | 8,0×10-16 |
| Ag2SO4 | 7,7.10-5 | Fe(OH)3 (18oC) | 3,8×10-38 |
| Ag2S | 5,7.10-51 | FeS | 3,7.10-19 |
| Al(OH)3 | 1,9×10-33 | Mg(OH)2 | 5,5×10-12 |
| BaCO3 | 7,0×10-9 | MnS розовый | 2,5×10-10 |
| BaCrO4 | 2,3.10-10 | NiS (18oC) | 2,0.10-28 |
| BaC2O4 | 1,2×10-7 | PbCl2 | 2,4×10-5 |
| Ba3(PO4)2 | 6,0.10-39 | PbI2 | 8,7.10-9 |
| BaSO4 | 1,08×10-10 | PbS (18oC) | 1,1.10-29 |
| CaCO3 | 4,8×10-9 | PbSO4 | 2,2×10-8 |
| CaC2O4 | 2,6×10-9 | SrCO3 | 1,1×10-10 |
| CaF2 | 4,0.10-11 | SrSO4 | 2,3×10-7 |
| Ca3(PO4)2 | 1,0.10-29 | Zn(OH)2 (20oC) | 4,0.10-16 |
| CaSO4 | 6,1×10-5 | ZnS | 1,6×10-24 |
ѕриложение 11
онстанты гидролиза некоторых протолитов в водных растворах при 250—
| Ёлектролит | онстанта протолиза, моль/л | |
| Ќазвание | ‘ормула | |
| ѕероксид водорода | Ќ2ќ2 | 1~10−12 (Ќ2ќ2, Ќќ2−,Ќ2ќ) 2~10−25 (Ќ2ќ −, Ќќ22−,Ќ2ќ) |
| ÷иановодород | HCN | =7∙10−10 (ЌCN, CN−,Ќ2ќ) |
| ћетакремниева€ кислота | Ќ2SiO3 | 1~10−10 (Ќ2 SiO3, ЌSiќ3−,Ќ2ќ) 2~10−12 (ЌSiO3 −, Siќ32−,Ќ2ќ) |
| Cероводород | Ќ2S | 1=6 ∙10−8 (Ќ2S, ЌS−,Ќ2ќ) 2=1 ∙10−14 (ЌS −, S2−,Ќ2ќ) |
| ”гольна€ кислота | Ќ2—ќ3 | 1=4,5 ∙10−7 (Ќ2—ќ3, Ќ—ќ3−,Ќ2ќ) 2=4,7∙10−11 (Ќ—ќ3−, —ќ32−,Ќ2ќ) |
| √идрат аммиака | NH3 ∙HOH | o=2 ∙10−5 (NЌ3∙HOH, NЌ4+,Ќ2ќ) |
| ”ксусна€ кислота | —Ќ3—ќќЌ | =2 ∙10−5 (—Ќ3—ќќЌ,—Ќ3—ќќ−,Ќ2ќ) |
| јзотиста€ кислота | ЌNO2 | =4∙10−4 (ЌNќ2, Nќ2−,Ќ2ќ) |
| ‘тороводород | HF | =7∙10−4 (ЌF, F−,Ќ2ќ) |
| ќртофосфорна€ кислота | Ќ3–ќ4 | 1=8∙10−3 (Ќ3–ќ4, Ќ2–ќ4−,Ќ2ќ) 2=6∙10−8 (Ќ2–ќ4−, Ќ–ќ42−,Ќ2ќ) 3=1∙10−12 (Ќ–ќ42−,–ќ43−, Ќ2ќ) |
| —ерниста€ кислота | Ќ2SO3 | 1=2∙10−2 (Ќ2SO3, ЌSќ3−,Ќ2ќ) 2=6∙10−8 (ЌSO3 −, Sќ32−,Ќ2ќ) |
| јзотна€ кислота | HNO3 | =43,6(ЌNќ3, Nќ3−,Ќ2ќ) |
| ћарганцова€ кислота | HMnO4 | =200(ЌMnќ4, Mnќ4−,Ќ2ќ) |
| —ерна€ кислота | H2SO4 | 1=1000(Ќ2SO4, ЌSќ4−,Ќ2ќ) 2=1∙10−2 (ЌSO4 −, Sќ42−,Ќ2ќ) |
| ’лороводород | HCI | =1∙107 (Ќ—I,—I−,Ќ2ќ) |
| Ѕромоводород | HBr | =1∙109 (ЌBr, Br−,Ќ2ќ) |
| …одоводород | HJ | =1∙1011 (ЌJ, J−,Ќ2ќ) |
|
|
|
ѕриложение 12
—войства важнейших двухцветных кислотно-основных индикаторов
| »ндикатор | ѕереход окраски | »нтервал pH |
| ћетиловый оранжевый | расна€ → оранжева€ | 3,1 Ц 4,0 |
| Ћакмоид | расна€ → син€€ | 4,0 Ц 6,4 |
| ћетиловый красный | расна€ → желта€ | 4,2 Ц 6,2 |
| Ѕромтимоловый | ∆елта€ → син€€ | 6,0 Ц 7,6 |
ѕриложение 13
—тандартные электродные потенциалы (∆≈º) некоторых металлов
| Ёлектрод | ≈0, ¬ | Ёлектрод | ≈0, ¬ | |
| Li+/Li Rb+/Rb K+/K Cs+/Cs Ba2+/Ba Ca2+/Ca Na+/Na Mg2+/Mg AI3+/AI Ti2+/Ti Zr4+/Zr Mn2+/Mn V2+/V Cr2+/Cr Zn2+/Zn Cr3+/Cr Fe2+/Fe | −3,045 −2,925 −2,924 −2,923 −2,90 −2,87 −2,714 −2,37 −1,70 −1,603 −1,58 −1,18 −1,18 −0,913 −0,763 −0,74 −0,44 | Cd+/Cd Co2+/Co Ni2+/Ni Sn2+/Sn Pb2+/Pb Fe3+/Fe 2H+/H Sb3+/Sb Bi3+/Bi Cu2+/Cu Cu+/Cu Fe Hg22+/2Hg Ag+/Ag Hg2+/Hg Pt2+/Pt Au3+/Au Au+/Au | −0,403 −0,277 −0,25 −0,136 −0,127 −0,037 −0,000 +0,20 +0,215 +0,34 +0,52 +0,77 +0,79 +0,80 +0,85 +1,19 +1,50 +1,70 |
ѕриложение 14
—тандартные восстановительные потенциалы,  при
при  .
.
| ѕроцесс |  , ¬ , ¬
| |

| +2,87 | |
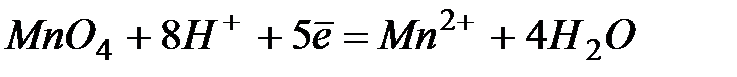
| +1,51 | |
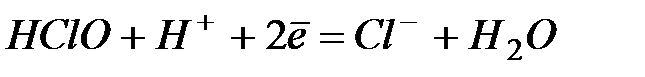
| +1,50 | |

| +1,36 | |

| +1,33 | |

| +1,23 | |

| +1,23 | |
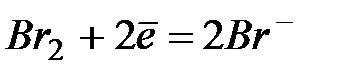
| +1,07 | |
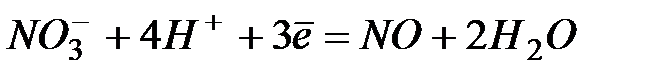
| +0,96 | |

| +0,87 | |

| +0,815 | |

| +0,80 | |

| +0,77 | |

| +0,63 | |

| +0,60 | |

| +0,56 | |

| +0,54 | |

| +0,36 | |

| +0,31 | |

| +0,26 | |

| +0,17 | |

| 0,000 | |
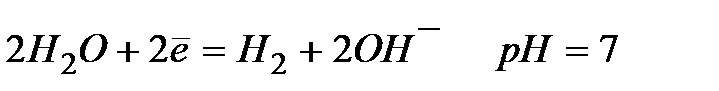 (pH=9) (pH=9)
| -0,413 (-0,53) | |

| -0,54 | |

| -0,83 | |

| -0,86 | |

| -0,91 | |

| -1,22 | |
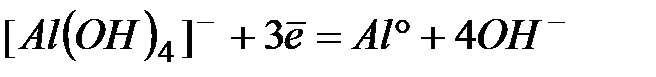
| -2,35 | |
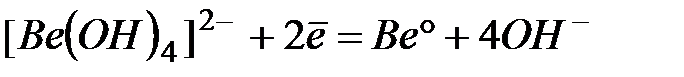
| -2,55 |
ѕриложение 15
онстанты нестойкости комплексных ионов в водных растворах при 25о—.
|
|
|
| —хема диссоциации комплексного иона | нестойкости | р |
| [Ag(NH3)2]+DAg+ + 2NH3 | 5,89×10-8 | 7,23 |
| [Ag(NO2)2]-D Ag+ + 2NO2- | 1,3×10-3 | 2,89 |
| [Ag(S2O3)2]3-D Ag+ + 2S2O32- | 2,5×10-14 | 13,60 |
| [Ag(CN)2]-D Ag+ + 2CN- | 1,4×10-20 | 19,85 |
| [AgI2]-D Ag+ + 2I- | 5,5.10-12 | 11,74 |
| [Al(OH)4(H2O)2]-D Al3+ + 2OH- + 2H2O | 1,0.10-33 | 33,0 |
| [AlF6]3-D Al3+ + 6F- | 5,01.10-18 | 17,30 |
| [AuCl4]-D Au3+ + 4Cl- | 5,0.10-22 | 21,30 |
| [Be(OH)4]2-D Be2+ + 4OH- | 1,0.10-15 | 15,0 |
| [BeF4]2-D Be2+ + 4F- | 4,17 .10-17 | 16,30 |
| [CaЁƒ“ј]2-D Ca2+ + Ёƒ“ј | 2,57.10-11 | 10,59 |
| [Cd(CN)4]2-D Cd2+ + 4CN- | 7,76.10-18 | 17,11 |
| [Cd(En)2]2+D Cd2+ + 2En | 6,0.10-11 | 10,22 |
| [Cd(NH3)6]2+D Cd2+ + 6NH3 | 2,76.10-5 | 4,56 |
| [Co(C2O4)3]3-D Co3+ + C2O42- | 5,0.10-12 | 11,30 |
| [Co(En)3]3+D Co3+ + 3En | 2,04.10-49 | 48,69 |
| [Co(NH3)6]2+D Co2+ + 6NH3 | 4,07.10-5 | 4,39 |
| [Co(NH3)6]3+D Co3+ + 6NH3 | 6,15.10-36 | 35,21 |
| [Co(NO2)6]3-D Co3+ + 6NO2- | 1,0.10-22 | 22,0 |
| [Co(SCN)4]2-D Co2+ + 4SCN- | 5,50.10-3 | 2,26 |
| [CoЁƒ“ј]2-D Co2+ + Ёƒ“ј | 4,90.10-17 | 16,31 |
| [CoЁƒ“ј]3-D Co3+ + Ёƒ“ј | 2,51.10-41 | 40,60 |
| [Cr(OH)4]-D Cr3+ + 4OH- | 1,26.10-30 | 29,90 |
| [CrЁƒ“ј]3-D Cr3+ + Ёƒ“ј | 3,98.10-24 | 23,40 |
| [Cu(CN)2]- D Cu+ + 2CN- | 1,0.10-24 | 24,00 |
| [Cu(CN)4]3-D Cu+ + 4CN- | 5,13.10-31 | 30,29 |
| [Cu(H2O)2Br2]oD Cu2+ + 2Br - + 2H2O | 2,22.10-6 | 5,75 |
| [Cu(NH3)4]2+D Cu2+ + 4NH3 | 9,33.10-13 | 12,03 |
| [Fe(CN)6]4-D Fe2+ + 6CN- | 1,4×10-37 | 36,84 |
| [Fe(CN)6]3-D Fe3+ + 6CN- | 1,5×10-44 | 43,82 |
| [Fe(SCN)3] D Fe3+ + 3SCN- | 2,9×10-5 | 4,54 |
| [FeCl3] D Fe3+ + 3Cl- | 7,4×10-2 | 1,13 |
| [FeF6]3-D Fe3+ +6F- | 7,94×10-17 | 16,10 |
| [FeЁƒ“ј]2-D Fe2+ + Ёƒ“ј | 6,31.10-15 | 14,20 |
| [FeЁƒ“ј]3-D Fe3+ + Ёƒ“ј | 5,89.10-25 | 24,23 |
| [HgBr4]2-D Hg2+ + 4Br- | 1,0×10-21 | 21,0 |
| [HgI4]2-D Hg2+ + 4I- | 1,4×10-30 | 29,85 |
| [Hg(CN)4]2-D Hg2+ + 4CN- | 4,0×10-42 | 41,40 |
| [Hg(SCN)4]2-D Hg2+ + 4SCN- | 8,0×10-22 | 21,10 |
| [MgЁƒ“ј]2-D Mg2+ + Ёƒ“ј | 7,59.10-10 | 9,12 |
| [NH4]+D NH3 + H+ | 6,0.10-10 | 9,22 |
| [Ni(En)3]2+D Ni2+ + 3En | 7,76.10-20 | 19,11 |
| [Ni(NH3)4]2+D Ni2+ + 4NH3 | 1,12.10-8 | 7,95 |
| [Ni(NH3)6]2+D Ni2+ + 6NH3 | 9,77.10-9 | 8,01 |
| [NiЁƒ“ј]2-D Ni2+ + Ёƒ“ј | 2,40.10-19 | 18,62 |
| [PtBr4]2-D Pt2+ + 4Br- | 3,0.10-21 | 20,52 |
| [PtCl4]2-D Pt2+ + 4Cl- | 1,0.10-16 | 16,00 |
| [SnCl6]4-D Sn2+ + 6Cl- | 5,1×10-11 | 10,29 |
| [Zn(CN)4]2-D Zn2+ + 4CN- | 6,3×10-18 | 17,20 |
| [Zn(NH3)4]2+D Zn2+ + 4NH3 | 2,0.10-9 | 8,70 |
| [Zn(OH)4]2-D Zn2+ + 4OH- | 3,6.10-16 | 15,44 |
| [ZnЁƒ“ј]2-D Zn2+ + Ёƒ“ј | 5,50.10-17 | 16,26 |
ѕриложение 16






