1. Ќазвати речовини за м≥жнародною номенклатурою
—≤ —2Ќ 5 — — —
— Ц — Ц — Ц — ≡ — — Ц — Ц — = — Ц — = — Ц — — = — Ц — Ц — Ц —
— — — — —
2. Ќаписати нап≥вструктурн≥ формулу речовини за в≥домою номенклатурною назвою.
ј) 2,3-диметил Ц 4 хлорпент-1-ен
Ѕ) 3,3,4,5-тетрабромгекс-1-ин
¬) 2,4 Цдиметил -5-етилгепта-1,3-д≥Їн
√) 2,5-диметил-3,3-диетилгаксан.
3. Ќаписати нап≥вструктурн≥ формули ≥зомер≥в
ј) октену
Ѕ) гексад≥Їну
4. «адач≥ на використанн€ загальноњ формули орган≥чних речовин.
ј) серед перел≥ку речовин вибер≥ть формули алк≥н≥в: —7Ќ14, —5Ќ10, —2Ќ4, —6Ќ6, —2Ќ2.
Ѕ) напиш≥ть формулу алкану у €кого Ї 16 атом≥в √≥дрогену
¬) напиш≥ть формулу алк≥ну у €кого Ї 6 атом≥в арбону
√) напиш≥ть формулу алкену у €кого Ї 16 атом≥в √≥дрогену
5. Ќаписати р≥вн€нн€ реакц≥й, що лежать в основ≥ поданоњ схеми
—Ќ4→—2Ќ2→—2Ќ4→—2Ќ5—≤→—4Ќ10→—4Ќ8→—4Ќ9ќЌ
6. «адач≥ на виведенн€ формул за в≥домою масовою часткою елемент≥в сполуки. јлгоритм р≥шенн€.
¬изначте молекул€рну формулу речовини, що м≥стить натр≥ю 42,07%, фосфору-!8,9%, оксисену Ц 39,05%. √устина пари речовини за азотом 5,857.
1. Ќапиш≥ть умову задач≥.
ƒано
ω(Na)=42,07%
ω(P)=18,9%
ω(O)=39,05%
D(N2)=5,857
NaxPyOz
2.за в≥дносною густиною пари речовини в≥дносно азоту знайдемо мол€рну масу нашоњ сполуки
ƒано
ω(Na)=42,07% Mr= D(N2) ×Mr(N2)=5,857×28=163,99=164 г/моль
ω(P)=18,9%
ω(O)=39,05%
D(N2)=5,875
NaxPyOz
3.ƒл€ того,щоб визначити масову частку елемент≥в у сполуц≥ використали формулу

« даноњ формули знайдемо чому дор≥внюЇ ≥ндекс n:

¬икористаЇмо дану формулу в наш≥й задач≥ ≥ знайдемо ≥ндекс дл€ кожного елемента
ƒано
ω(Na)=42,07% Mr= D(N2) ×Mr(N2)=5,857×28=163,99=164 г/моль
ω(P)=18,9% 
ω(O)=39,05%
D(N2)=5,875
NaxPyOz 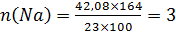


¬≥дпов≥дь: Na3PO4
–озвТ€ж≥ть задач≥:
1. ћасов≥ частки елемент≥в у орган≥чн≥й сполуц≥ р≥вн≥ арбону 76, %, √≥дрогену 12,7%, ќксисену 11,3. √устина пар≥в сполуки за киснем 8,875. «найд≥ть формулу сполуки.
2. ћасов≥ частки арбону, √≥дрогену та ’лору в≥дпов≥дно р≥вн≥ 56,3%, 10,4 %, 33.3%. √устина за воднем 53,25. «найд≥ть формулу сполуки.
“ема 4 ¬углеводн≥
«ан€тт€ 4. јрени.
¬ид зан€тт€: √рупове зан€тт€ 11
ћета: ознайомитись з бензеном Ц представником гомолог≥чного р€ду арен≥в, з особливост€ми нового виду звТ€зк≥в Ц бензольного €дра; установити причинно-насл≥дков≥ звТ€зки властивостей бензену та його будови; визначити х≥м≥чн≥ властивост≥ бензену: реакц≥њ приЇднанн€, зам≥щенн€, окисненн€; удосконалювати вм≥нн€ студент≥в складати р≥вн€нн€ реакц≥й м≥ж речовинами.
Ќавчальн≥ питанн€:
1.Ѕензен, склад, електронна та структурна формула,, ф≥зичн≥ властивост≥. Ќоменклатурн≥ назви арен≥в.
2. ’≥м≥чн≥ властивост≥ бензену.
3.«астосуванн€ бензену. ’≥м≥чн≥ засоби захисту рослин.
Ќавчальна л≥тература:
Ћ-3 с.74-83
Ћ-4 с.78-81
≤≤≤ .јрени.
|
|
|
| ¬изначенн€ | ‘ункц≥ональн≥ групи | «агальна формула | Ќоменк- латурна назва | ¬иди ≥зомер≥њ | Ѕудова | «астосуванн€ | ƒобуванн€ | ‘≥зичн≥ ≥ ’≥м≥чн≥ властивост≥ |
| јрени- це карбоцикл≥чн≥ сполуки, до складу €ких вход€ть одне,або к≥лька бензольних €дер. | Ѕензольне €дро бензен | CnH2n-6, де n ≥ 6 | Ќомера ц≥н починаЇтьс€ в≥д першого зам≥сника в сторону наступного,зак≥нчена бензен | 1.≤зомер≥€ положенн€ зам≥сника 2.≤зомер≥€ виду зам≥сника. | ожен атом карбону у цикл≥ знаходитьс€ в стан≥ sр2 ш≥сть карбон≥в утворюють замкнене коло, що м≥стить ш≥сть δ- звТ€зк≥в, 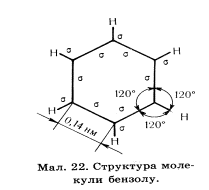 Ў≥сть не г≥бридизованих р- орб≥талей розм≥щуютьс€ перпендикул€рно площин≥ δ- звТ€зк≥в, та утворюють сп≥льний дл€ вс≥х шести атом≥в π-зв'€зок Ц бензольне €дро
Ў≥сть не г≥бридизованих р- орб≥талей розм≥щуютьс€ перпендикул€рно площин≥ δ- звТ€зк≥в, та утворюють сп≥льний дл€ вс≥х шести атом≥в π-зв'€зок Ц бензольне €дро
| 1.сировина дл€ синтезу: барвник≥в, л≥карських препарат≥в, пол≥мер≥в, отрутох≥м≥кат≥в, вибухових речовин, 2.розчинник 3.пох≥дн≥ бензину Ц пестициди | 1.з продукт≥в коксуванн€ камТ€ного вуг≥лл€. 2.з нафти,з циклоалкан≥в “=500 0—, –=2500 Ц 5066 кѕа, катал≥затор. зал≥зо —6Ќ12→—6Ќ6 +3Ќ2 3.дег≥дроцикл≥зац≥€ алкан≥в —7Ќ16 →—7Ќ8 +4Ќ2 4.тримеризац≥€ ацетилену ,“=800 0—, катал≥затор Ц розжарене вуг≥лл€ 3—2Ќ2→—6Ќ6 | –≥дина з специф≥чним запахом, “ кип=80 0—, нерозчинний у вод≥, розчинний у непол€рних розчинниках: керосин≥, бензин≥, ацетон≥. Ѕензен- отруйна речовина. ¬диханн€ пар≥в бензину викликаЇ запамороченн€, головний б≥ль, при високих концентрац≥€х Ц можлива втрата св≥домост≥. 1.горить 2—6Ќ6 + 5ќ2→12—ќ2+6Ќ2ќ 2. зам≥щенн€. атал≥затори FeCl3, AlCl3. C6H6+ Cl2→C6H5Cl +HCl 3.н≥труванн€. катал≥затор - H2SO4 C6H6+HNO3→C6H5NO2 +H2O 4.сульфуванн€ C6H6+ H2SO4 →C6H5SO3H +H2O 5.ѕриЇднанн€. ј) г≥друванн€: “=200 —,–=5066 кѕа, св≥тло, катал≥затори н≥кель, платина. C6H6+3H2→C6H12(циклогексан) Ѕ) галогенуванн€: катал≥затор - ультраф≥олетове св≥тло C6H6+3Cl2→C6H6Cl6(гексахлорциклагексан) 6.Ќе д≥ють на розчин марганц≥вки та бромноњ води. |
Ѕудова молекули бензену.


ожен атом карбону у цикл≥ знаходитьс€ в стан≥ sр2 г≥бридизац≥њ отже три г≥бридизован≥ орб≥тал≥ кожного з шести карбон≥в утворюють р≥вносторонн≥й трикутник на площин≥. ƒв≥ орб≥тал≥ кожного трикутника зТЇднан≥ з сус≥дн≥ми такими ж орб≥та л€ми, утворюючи замкнене коло, що м≥стить ш≥сть δ- звТ€зк≥в, трет€ орб≥таль кожного карбону звТ€зана з атомом г≥дрогену.
Ќе г≥бридизован≥ р- орб≥тал≥ кожного атома карбону розм≥щуЇтьс€ перпендикул€рно площин≥ δ- звТ€зк≥в, ц≥ орб≥тал≥ по черз≥ перекриваютьс€, утворюючи сп≥льний дл€ вс≥х шести атом≥в π-зв'€зок Ц бензольне €дро.
ѕестициди - - речовини що використовуютьс€ в с≥льському господарств≥ дл€ захисту рослин. √ерб≥циди Ц боротьба з бурТ€нами. ≤нсектициди Ц боротьба з комахами. ‘унг≥циди Ц боротьба з грибками. «ооциди Ц боротьба з тваринами-шк≥дниками.
ѕриклади назв арен≥в по м≥жнародн≥й номенклатур≥.
—Ќ3
—Ќ3
1,2-диметилбензен.






