оличество вещества, которое может растворитьс€ в определенном количестве вещества-растворител€ до получени€ насыщенного раствора, называетс€ растворимостью. –астворимость твердых, жидких и газообразных веществ обычно выражаетс€ в граммах раствор€емого вещества на 100 г воды. ¬ “абл. 6 дана растворимость некоторых наиболее распространенных веществ, используемых при приготовлении буровых растворов.
| ‘ормула | Ќазвание | –астворимость (г/100 г воды) |
| NaOH | аустическа€ сода | |
| CaCl2 | ’лорид кальци€ | 47,5 |
| NaCl | ’лорид натри€ (поваренна€ соль) | |
| KCl | ’лорид кали€ | 34,7 |
| Na2CO3 | альцинированна€ сода | 21,5 |
| NaHCO3 | Ѕикарбонат натри€ | 9,6 |
| CaSO4 | јнгидрит | 0,290 |
| Ca(OH)2 | »звесть | 0,185 |
| MgCO3 | арбонат магни€ | 0,129 |
| CaCO3 | »звестн€к | 0,0014 |
| Mg(OH)2 | √идроксид магни€ | 0,0009 |
| BaSO4 | Ѕарит | 0,0002 |
| ZnO | ќксид цинка | 0,00016 |
“абл. 6. –астворимость соединений.
”слови€, вли€ющие на растворимость:
Ј “емпература
Ј pH (основание или кислота)
Ј —одержание солей (минерализаци€)
Ј ƒавление
1. “емпература. — ростом температуры растворимость большинства твердых веществ и жидкостей повышаетс€. –астворимость газов при повышении температуры обычно снижаетс€.
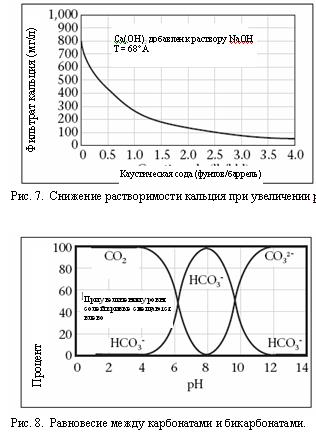
2. pH. pH Ц мера относительной кислотности или щелочности раствора (подробное описание будет дано ниже). –астворимость многих химреагентов зависит от уровн€ рЌ. Ќекоторые соединени€, например поливалентные соли гидроксида и карбоната, станов€тс€ более растворимыми в кислотной среде. Ќекоторые соединени€ раствор€ютс€ только в среде с нейтральным рЌ, другие (органические кислоты, такие как лигнит и лигносульфонат) раствор€ютс€ лучше при повышении рЌ до > 9,5. »оны кальци€ и магни€ раствор€ютс€ при уровн€х рЌ от кислотного до нейтрального, однако станов€тс€ менее растворимы при высоких уровн€х рЌ (см. пример с кальцием на –ис. 7). ѕри увеличении числа гидроксид-ионов по мере роста рЌ, ионы реагируют с кальцием и магнием, при этом образуетс€ осадок из гидроксида кальци€ и магни€. Ќекоторые соединени€, например ионы карбоната или сульфида, с увеличением уровн€ рЌ мен€ют свое состо€ние. Ќапример, газ —ќ2 при реакции с водой образует углекислоту при низком уровне рЌ. ѕри нейтральном рЌ —ќ2 реагирует с гидроксидом, образу€ ионы бикарбоната, при высоком рЌ Ц ионы карбоната (см. –ис. 8).
 –астворимость многих компонентов буровых растворов напр€мую зависит от уровн€ рЌ раствора. Ќапример, при рЌ > 9,5 лучше раствор€ютс€ не только лигнит и лигносульфонат, но и добавки, такие как Duo-VisЩ (ксантанова€ смола), станов€тс€ более эффективны при уровне рЌ от 7 до 11. Ќекоторые добавки особенно чувствительны к высокому уровню рЌ.
–астворимость многих компонентов буровых растворов напр€мую зависит от уровн€ рЌ раствора. Ќапример, при рЌ > 9,5 лучше раствор€ютс€ не только лигнит и лигносульфонат, но и добавки, такие как Duo-VisЩ (ксантанова€ смола), станов€тс€ более эффективны при уровне рЌ от 7 до 11. Ќекоторые добавки особенно чувствительны к высокому уровню рЌ.
ѕри высоком уровне рЌ (>10,5) добавка Poly-PlusЩ гидролизируетс€, ее эффективность снижаетс€. Ѕольшинство добавок предназначены дл€ применени€ в определенном диапазоне рЌ. ƒл€ лучшего растворени€ ингредиентов, борьбы с коррозией скважинного оборудовани€ и безопасного обращени€ с химреагентами рЌ буровых растворов на водной основе обычно поддерживаетс€ на уровне от 8 до 12. »нженеры по буровым растворам должны знать о допустимых уровн€х рЌ и поддерживать рЌ раствора в допустимом диапазоне.
|
|
|
3. —одержание солей (минерализаци€). ѕри работе с буровыми растворами особое значение имеет концентраци€ хлоридов (минерализаци€). ¬ целом при увеличении концентрации хлоридов повышаетс€ растворимость других солей и добавок; минерализаци€ вли€ет на интенсивность образовани€ осадка. Ќапример, максимальна€ растворимость сульфата кальци€ (гипс и ангидрит) наблюдаетс€ в 15%-ном солевом растворе, где она в 4 раза выше, чем в пресной воде. ќднако эта тенденци€ снижаетс€ по мере насыщени€ раствора. »звесть (гидроксид кальци€) также имеет более высокую растворимость при умеренной минерализации. ¬ солевом растворе более стабильны даже полимеры. »онна€ среда растворител€ оказывает чрезвычайно большое вли€ние на химические реакции и стабильность самых различных реагентов.
4. ƒавление. ѕри увеличении давлени€ наблюдаетс€ увеличение растворимости газов в жидкост€х, однако давление практически не вли€ет на растворимость жидкостей и твердых веществ. ѕовышенна€ растворимость газов наиболее важна с точки зрени€ химических условий в скважине, когда пластовой газ под давлением раствор€етс€ в буровом растворе. «нать относительную растворимость необходимо потому, что наименее растворимый реагент первым выпадает в осадок. Ќапример, при смешивании хлорида кальци€ (это вещество с высокой растворимостью) с водой хлорид кальци€ распадаетс€ на ионы кальци€ и хлора. ≈сли в этот же раствор добавить кальцинированную соду (вещество со средней растворимостью), она распадетс€ на ионы натри€ и карбоната, а карбонат кальци€ (вещество с низкой растворимостью) немедленно выпадет из раствора при реакции хорошо растворимого кальци€ и хорошо растворимого карбоната. ќтносительна€ растворимость может быть использована при выборе реагентов дл€ удалени€ ненужных соединений из раствора. Ќа растворимость вли€ют ионы различных элементов и соединений, содержащиес€ в растворителе.
–Ќ и кислотность
–Ќ
ѕоказатель рЌ используют дл€ характеристики кислотности или щелочности растворов. рЌ Ц это отрицательный логарифм концентрации ионов водорода. Ќизкий показатель рЌ соответствует кислой среде, высокий рЌ Ц щелочной среде. »зменение одной единицы рЌ соответствует дес€тикратному изменению концентрации ионов водорода. ¬ода Ц слабый электролит, существующий в природе в виде молекул H2O. ¬озможна ионизаци€ воды в H3O или в гидроксил-ионы (ќЌ). »онизаци€ воды Ц статистически редкое €вление, из 556 миллионов молекул воды ионизироватьс€ может лишь одна. ¬ соответствии с приведенным ниже уравнением, вода находитс€ в состо€нии ионного равновеси€:
2H2O↔ H3O+(aq) + OHЦ(aq)
—окращение (aq) указывает на то, что ионы растворены в воде и образован водный раствор. “ак как вода может ионизировать сама себ€, этот процесс называетс€ самоионизацией. »онное равновесие с H3O и ќЌ служит основой дл€ классификации кислот и оснований. ислоты увеличивают концентрацию ионов водорода (Ќ+) при растворении в воде, увеличива€ тем самым концентрацию [Ќ3ќ+]. онцентраци€ различных ионов обозначаетс€ химической формулой в квадратных скобках; например, [H+] обозначает концентрацию ионов водорода.
|
|
|
¬ыражение дл€ ионного равновеси€ при самоионизации выгл€дит следующим образом:
Kw = [H+][OH-]
¬ыражение данного типа часто используетс€ дл€ описани€ равновеси€ между родственными химическими соединени€ми. —имвол Kw обозначает константу химического равновеси€, подстрочный символ Ђwї в данном случае обозначает воду. ѕри “= +25∞C, Kw = l,0 x 10Ц14. Kw зависит от температуры (с увеличением температуры увеличиваетс€ Kw) и концентрации ионов в растворе (минерализации). онстанта Kw, €вл€€сь произведением [H+] и [OH-], остаетс€ неизменной при посто€нной температуре. ¬ рЌ-нейтральном растворе концентраци€ [H+] равна концентрации гидроксида [OH-], следовательно, концентраци€ [Ќ+] и [OH-] равна 1,0 х 10-7, а уровень рЌ раствора равен 7,0. ѕри увеличении [H+] происходит снижение [OH-], раствор становитс€ более кислотным. » наоборот, при увеличении [OH-] снижаетс€ [H+], а раствор становитс€ более щелочным.
»оны Ќ+(aq) и ќЌ-(aq) всегда присутствуют в водном растворе и наход€тс€ в равновесии с растворителем. Ќ+(aq) и ќЌ-(aq) могут реагировать с другими ионами, вли€€ на их концентрацию в растворе. ѕоэтому концентраци€ ионов Ќ+(aq) и ќЌ-(aq) различаетс€ между собой по виду ионов. ѕоказатели рЌ и рќЌ определ€ютс€ как
pH = - log [H+]
pOH = - log [OHЦ]
—оотношение между рЌ и рќЌ находитс€ вычислением отрицательного логарифма (выраженного символом Ђрї) Kw, что дает
pKw = -log Kw = -log [H+] -log [OHЦ]
»спользу€ данные выше определени€ рЌ и рќЌ, можно обнаружить, что при “ = +25∞C:
pKw = pH + pOH
так как Kw = l,0 x 10Ц14, тогда
pKw = - log Kw = 14, что дает
pH + pOH = 14

Ќа рис. 9 показано кислотно-щелочное отношение с уровн€ми рЌ.
—ледует запомнить, что низкий уровень рЌ соответствует кислотам, а низкий уровень рќЌ Ц основани€м. »зменение одной единицы рЌ или рќЌ соответствует изменению мол€рной концентрации в дес€ть раз. –аствор с рЌ = 2 не €вл€етс€ в два раза более кислым, чем раствор с рЌ = 4; он в 100 раз кислее раствора с рЌ = 4.
ѕомните, что величина Kw зависит от температуры и концентрации ионов (минерализации раствора), поэтому уровень рЌ, измеренный электронным рЌ-метром, не будет соответствовать действительности, если в прибор не введена поправка по температуре раствора и концентрации солей. ƒл€ измерени€ рЌ в растворах с высокой концентрации солей может потребоватьс€ специальный компенсирующий прибор.
ак обсуждалось ранее, при растворении в воде NaCl (нейтральна€ соль, образующа€с€ при реакции сильной кислоты с сильным основанием), ионы Na+ не сочетаютс€ с ионами OH- (со снижением рЌ), т.к. NaOH Ц это сильное основание. јналогичным образом ионы Cl- не сочетаютс€ с ионами H+ (с увеличением рЌ), т.к. HCl Ц сильна€ кислота. ¬ результате в растворе остаютс€ как ионы H+, так и OH-, а уровень рЌ остаетс€ нейтральным. ≈сли же соль содержит катион сильного основани€ и анион сильной кислоты, раствор будет оставатьс€ нейтральным. ќднако если соль содержит катион сильного основани€ и анион слабой кислоты, раствор будет щелочным (с высоким рЌ), как в случае с кальцинированной содой (Na2CO3). » наоборот, если соль содержит катион слабого основани€ и анион сильной кислоты, раствор будет кислотным (с низким рЌ). –азумеетс€, при добавлении в нейтральный раствор кислоты рЌ снизитс€, а при добавлении основани€ Ц возрастет.
ўелочность
ўелочное титрирование примен€етс€ дл€ определени€ концентраций OHЦ,
HCO3Ц и CO32Ц путем измерени€ количества кислоты, необходимого дл€ снижени€ рЌ. Ѕораты, силикаты, фосфаты, сульфаты и органические кислоты (например, лигнит) также могут титрироватьс€ по уровню щелочности. ўелочность Ц это мера силы основани€, определ€ема€ количеством кислоты, необходимой дл€ реакции с образованием соли. ѕри работе с буровыми растворами щелочность по фенолфталеину (–) измер€етс€ в миллилитрах 0,02N H2SO4 (дл€ буровых растворов на водной основе), необходимой дл€ титрировани€ одного миллилитра фильтрата (–f) или бурового раствора (Pm) дл€ снижени€ рЌ до 8,3. ўелочность и рЌ Ц не одно и то же, хот€ обычно уровни щелочности и рЌ измен€ютс€ в одном направлении. «ависимость между уровн€ми щелочного титрировани€ и рЌ можно продемонстрировать на примере добавлени€ в чистую воду сильного основани€, такого как каустическа€ сода. «начени€ щелочности и рЌ сведены в “абл. 7. ќднако, благодар€ присутствию HCO3Ц, CO32Ц, а также кальци€ и магни€ в буровых растворах и промысловой технической воде, пр€мой зависимости между щелочностью и рЌ нет.
|
|
|
| рЌ | NaOH (фунт/баррель) | Pf (cc 0,02N H2SO4) | OH- (ppm) |
| 0,0000014 | 0,000005 | 0,0017 | |
| 0,000014 | 0,00005 | 0,017 | |
| 0,00014 | 0,0005 | 0,17 | |
| 0,0014 | 0,005 | 1,7 | |
| 0,014 | 0,05 | ||
| 0,14 | 0,5 | ||
| 1,4 | |||
“абл. 7. рЌ и щелочность пресной воды.
¬ таблице показано, как мала€ концентраци€ каустической соды (NaOH) в пресной воде приводит к относительно высокому уровню рЌ и щелочности фильтрата. ƒл€ наблюдени€ вли€ни€ более сложной ионной среды, сравните количество каустической соды, необходимой дл€ увеличени€ рЌ в морской воде (см. “абл. 10). »змерени€ щелочности ((Pf, Mf и т.д.) используютс€ дл€ подсчета концентрации гидроксила, бикарбоната и карбоната согласно методике API 13B-1, Section 8, Table 8.1, и описанию в главе Ђ»сследовани€ буровых растворовї. “ак как при использовании органических кислот или органических солей (таких как лигнит или ацетат) измерение Mf может оказатьс€ ненадежным показателем загр€знени€ раствора бикарбонатами, примен€ют альтернативный метод измерени€ рЌ и Pf дл€ подсчета концентрации карбонатов и бикарбонатов. Ётот метод помогает обнаружить загр€знение бурового раствора углекислым газом, бикарбонатом и карбонатом. роме того, метод позвол€ет инженеру по буровым растворам получить более четкое представление о состо€нии раствора в сравнении с простым измерением рЌ.
ислоты, основани€ и соли
ислоты можно охарактеризовать как вещества кислые на вкус, выдел€ющие пузырьки при реакции с карбонатами, вызывающие посинение лакмусового красного индикатора, вступающие в реакции с основани€ми и некоторыми металлами с образованием солей. ¬се кислоты содержат атомы водорода. ислоты подраздел€ютс€ на Ђсильныеї и Ђслабыеї в зависимости от концентрации ионов водорода (H+) вследствие ионизации.
ќсновани€ Ц это горькие на вкус и Ђскользкиеї на ощупь вещества, вызывающие посинение красной лакмусовой бумажки, вступающие в реакцию с кислотами с образованием солей. ѕри реакции оснований с карбонатами пузырьки не выдел€ютс€. ѕри реакции между кислотой и основанием образуетс€ соль. ¬ зависимости от количества молекул, диссоциирующих в растворе на гидроксил-ионы (OH-), основани€ подраздел€ютс€ на сильные и слабые.
» кислоты, и основани€ могут быть сильными и слабыми, в зависимости от составл€ющих их элементов и их валентности.
—оли представл€ют собой сочетани€ аниона (отрицательно зар€женного иона) кислоты и катиона (положительно зар€женного иона) основани€. —оль может быть как нейтральной, так и кислотной или щелочной, в зависимости от силы составл€ющих ее ионов или групп ионов. ак подчеркивалось ранее, сочетание слабой кислоты и сильного основани€ даст щелочную соль, а сочетание сильной кислоты и слабого основани€ Ц кислотную соль. ѕри реакции сильной кислоты с сильным основанием образуетс€ нейтральна€ соль. ¬ “абл. 8 перечислены кислоты, основани€ и соли, широко примен€ющиес€ в приготовлении буровых растворов.
|
|
|
| ’имическое название | ќбщеупотребительное название | ‘ормула | “ип |
| ’лороводородна€ кислота | —ол€на€ кислота | HCl | —ильна€ кислота |
| —ерна€ кислота | - | H2SO4 | —ильна€ кислота |
| јзотна€ кислота | - | HNO3 | —ильна€ кислота |
| ‘осфорна€ кислота | - | H3PO4 | —редн€€ кислота |
| ”глекислота | - | H2CO3 | —лаба€ кислота |
| Ћимонна€ кислота | - | H3C6H5O7 | —лаба€ кислота |
| √идроксид натри€ | аустическа€ сода | NaOH | —ильное основание |
| √идроксид кали€ | ≈дкое кали | KOH | —ильное основание |
| √идроксид магни€ | - | Mg(OH)2 | ќснование |
| арбонат натри€ | альцинированна€ сода | Na2CO3 | —лабое основание |
| √идроксид кальци€ | √ашена€ известь | Ca(OH)2 | —ильное основание |
| ќксид кальци€ | Ќегашена€ известь | CaO | —ильное основание |
| ’лорид натри€ | аменна€ (поваренна€) соль | NaCl | —оль |
| ’лорид кали€ | ѕоташ | KCl | —оль |
| ’лорид кальци€ | - | CaCl2 | —оль |
| —ульфат кальци€ | јнгидрит, безводный гипс | CaSO4(*2H2O) | —оль |
Ѕуферные растворы
Ќекоторые растворы называют буферными из-за их способности сопротивл€тьс€ резким изменени€м рЌ при добавлении кислот и оснований. ћножество нефтепромысловых жидкостей и добавок к буровым растворам €вл€ютс€ буферными растворами. ¬ основном такие растворы состо€т из слабой кислоты и содержащей тот же самый анион соли, либо слабого основани€ и содержащий тот же самый катион соли. Ѕуферное действие раствора, состо€щего из слабой кислоты и ее соли, обусловлено тем, что (1) добавленное основание реагирует со слабой кислотой, образу€ дополнительные общие ионы, (2) добавленна€ кислота реагирует с общим ионом, образу€ слабую кислоту. ѕримером слабой кислоты €вл€етс€ угольна€ кислота (H2CO3). ѕример слабого основани€ Ц гидроксид аммони€ (NH4OH). ≈сли небольшое количество сильной кислоты, например HCl, добавить в пресную воду или в слабый раствор кислоты в воде, концентраци€ ионов водорода (Ќ+) воды или раствора заметно возрастет. ≈сли это же малое количество кислоты добавить в буферный раствор слабой кислоты и ее растворимой соли, увеличение рЌ будет таким незначительным, что на практике им можно пренебречь. јнионы соли слабой кислоты св€зывают ионы H+ по мере поступлени€ этих ионов в раствор. јнионы слабой кислоты реагируют с ионами водорода, образу€ все большее количество слабой кислоты. ¬ результате концентраци€ ионов водорода мен€етс€ лишь незначительно, оказыва€ очень небольшое вли€ние на уровень рЌ. Ёто €вление часто наблюдаетс€ при титрировании кислотой до конечной точки при определении щелочности. ¬ растворах с большим содержанием карбонатов, бикарбонатов и гидроксидов, как только из карбонатов образовались бикарбонаты, формируетс€ сопротивл€ющийс€ изменени€м рЌ буферный раствор. ƒл€ приготовлени€ буфера к критичным по рЌ буровым растворам используют триэтаноламин, известь и оксид магни€. »спользование буферных растворов способствует поддержанию стабильных параметров буровых растворов и повышает их сопротивл€емость загр€знению самыми разнообразными веществами.
Ёлектролиты
¬ещества, водные растворы которых провод€т электрический ток, называютс€ электролитами. Ёлектролиты содержат как положительно, так и отрицательно зар€женные ионы. Ёлектролитами €вл€ютс€ все растворы ионных соединений. ¬се кислоты, основани€ и соли Ц электролиты. Ёлектровалентные соединени€ образуютс€ в результате передачи электронов (ионные св€зи), во врем€ которой формируютс€ положительно и отрицательно зар€женные ионы. ¬се образующиес€ при этом твердые соединени€ (соли и гидроксиды) имеют ионную кристаллическую решетку. Ёто означает, что ионы формируютс€ во врем€ формировани€ самого электровалентного соединени€. »оны существовали еще перед тем, как данное соединение растворилось в воде. ѕри растворении электровалентного соединени€ ионна€ кристаллическа€ решетка разрушаетс€ и ионы диссоциируют в растворе; воде же лишь выступает в роли простого растворител€. Ќиже приведены типовые уравнени€, иллюстрирующие диссоциацию ранее образованных ионов:
—оль: NaCl = Na+ + ClЦ
(см. –ис. 6)
»звесть: Ca(OH)2 = Ca2+ + 2OHЦ
“ак как способность раствора проводить электрический ток зависит от присутстви€ в нем ионов, можно сделать вывод о том, что растворы с хорошими электропроводными свойствами имеют высокую концентрацию ионов (€вл€ютс€ полностью ионизированными), а растворы с плохой электропроводностью содержат низкую концентрацию ионов (€вл€ютс€ не полностью ионизированными). ѕолностью ионизированные электролиты называют сильными электролитами, не полностью ионизированные растворы называют слабыми электролитами. «а небольшим исключением, все соли Ц сильные электролиты. Ѕольшинство гидроксидов, за исключением гидроксида Mg2+, - сильные электролиты; они классифицируютс€ так же как и сильные основани€. ислоты HCl, H2SO4, HNO3 Ц сильные электролиты, следовательно они Ц сильные кислоты. Ѕольшинство других электролитов Ц умеренно слабые, соответственно они €вл€ютс€ слабыми кислотами. ѕресна€ вода Ц слабый электролит. ≈е электропроводные свойства слабее, чем у солевых растворов.
|
|
|
ќсмос
ќсмос Ц это €вление, возникающее когда два раствора со значительно отличающейс€ концентрацией растворенного в них вещества (минерализацией) отделены полупроницаемой мембраной. ѕри осмосе наблюдаетс€ движение растворител€ (воды) через мембрану из раствора с низкой минерализацией (т.е. концентрацией растворенного в воде вещества) в раствор с высокой минерализацией. “аким образом, при осмосе происходит выравнивание концентрации растворенного в двух растворах вещества (выравнивание минерализации или солености раствора). ƒвижущие силы осмоса Ц разница в концентраци€х растворенного вещества и свойства полупроницаемой мембраны. Ђјктивностьї раствора зависит от давлени€ паров или Ђотносительной влажностиї, так же как и от концентрации растворенного в растворе вещества (т.е. солености раствора). јктивность воды равна 1,0; чем выше соленость раствора, тем ниже его активность. ѕри проходке неустойчивых глин желательно, чтобы активность бурового раствора и пласта была приблизительно одинаковой, что позвол€ет избежать поглощени€ пластом воды из бурового раствора. –астворы на водной или синтетической основе могут отдавать воду из водной эмульсии (обычно этим характерен солевой раствор на основе хлорида кальци€), при условии, что осмотическа€ активность такого раствора выше активности пласта.
“итрирование
“итрирование Ц это один из видов исследований при проведении химического анализа буровых растворов. “итрирование представл€ет собой процедуру, в которой стандартные растворы с заранее известной концентрацией (N1) используютс€ дл€ определени€ неизвестной концентрации (N2) в пробе измеренного объема (V2). ќсновное уравнение данного вида количественного анализа имеет следующий вид:
V2 x N2 = V1 x N1
–еша€ уравнение, находим N2:

≈сли известен объем пробы (V2), дл€ определени€ неизвестной концентрации пробы (N2) необходимо использовать индикатор и титрировать пробу раствором с известной концентрацией (N1), измер€€ объем раствора (V1), необходимый дл€ достижени€ конечной точки титрировани€. ѕодробное описание методов титрировани€ приведены в главе Ђ»сследование свойств буровых растворовї, где идет речь о количественных измерени€х при помощи стандартных растворов дл€ определени€ наиболее распространенных химреагентов. ѕри выполнении титрировани€ необходимо строгое соблюдать методические рекомендации и инструкции по проведению работ. ѕриведенные в указанной главе формулы помогут инженерам при выполнении необходимых расчетов обойтись без перевода единиц измерений из одной системы в другую.
»ндикаторы
’имические вещества, которые служат дл€ определени€ конечной точки титрировани€, называют индикаторами. »ндикаторы Ц это соединени€, которые мен€ют свой цвет либо при изменении уровн€ рЌ, либо при изменении концентрации какого-либо вещества. »зменение цвета индикаторов на кислотной основе имеет место только при определенном значении рЌ. –азличные индикаторы могут мен€ть цвет при погружении в кислотную, нейтральную или щелочную среду. —уществуют химические индикаторы, мен€ющие цвет в присутствии кальци€, хлорида магни€ или бромидов. ¬ “абл. 9 перечислены наиболее распространенные индикаторы, используемые при исследовании химических свойств буровых растворов методом титрировани€, и изменени€ цветов индикаторов в различных услови€х.
| »ндикатор | ѕервоначальный цвет | »змененный цвет | “итрирование | “итрирующее вещество |
| ‘енолфталеин | –озовый/красный: рЌ>8,3 | Ѕесцветный: рЌ<8,3 | Pm Pf Pom | —ерна€ к-та |
| ћетилоранж/бромкрезол зеленый | «еленый: рЌ>4,3 | ∆елтый: рЌ<4,3 | Mf | —ерна€ к-та |
| ћетилоранж | ∆елтый/оранжевый: рЌ>4,3 | –озовый/красный: рЌ<4,3 | Mf | —ерна€ к-та |
| Ѕромкрезол зеленый | —иний: рЌ>3,8 | ∆елтый: рЌ<3,8 | -- | —ерна€ к-та |
| “имолфталеин | Ѕесцветный: рЌ<9,5 | —иний: рЌ>9,5 | -- | аустический р-р |
| ћетиловый красный | ∆елтый: рЌ>5,4 | –озовый/красный: рЌ<5,4 | PHPA* | —ерна€ к-та |
| Calmagite, manver или черный erio black T | ¬инно-красный: ѕрисутствие Ca2+ и Mg2+ | —иний/лиловый: ќтсутствие —а2+ и Mg2+ | ќбща€ жесткость | —танд. версенат (0,01 m EDTA) |
| CalVer II или Calcon | ¬инно-красный: ѕрисутствие Ca2+ | —иний/лиловый: ќтсутствие Ca2+ | альций | —танд. версенат (0,01 m EDTA) |
| –-р хромата кали€ | ∆елтый | ќранж./красный: »збыток AgNO3 | ’лориды | –-р нитрата серебра |
* PHPA Ц частично гидролизованный полиакриламид
“абл. 9. Ќаиболее распространенные индикаторы.
онцентраци€ растворов
ћол€льность (m). ћольный раствор Ц это раствор, содержащий один моль растворенного вещества на один килограмм растворител€. “аким образом, 1 m (1-мольный) раствор NaOH Ц это 40 г NaOH на 1 000 г воды.
ћол€рность (ћ). –аствор, содержащий один моль растворенного вещества на один литр раствора называют мол€рным. —ледовательно, 0,1 ћ (0,1-мол€рный) раствор HCl Ц это раствор сол€ной кислоты, в котором содержитс€ 1/10 мол€ или 3,646 г сол€ной кислоты на один литр раствора. ≈сли известна нормальность (N) раствора (см. ниже), можно вычислить и мол€рность (ћ) раствора делением нормальности на суммарную положительную валентность:
ћол€рность = Ќормальность / „иста€ положительна€ валентность
Ќормальность (N). –аствор с нормальностью 1,0 (N) Ц это раствор, в котором содержитс€ 1 г/экв. веса вещества на один литр раствора. Ќормальность такого раствора записываетс€ как 1,0 N. Ќапример, 1,0 N раствор HCl будет содержать 36,5 г растворенного вещества на один литр раствора. “аким же образом, 1,0 N раствор H2SO4 содержит 49 г растворенного вещества на один литр раствора. ≈сли известна мол€рность (ћ) раствора, нормальность (N) вычисл€етс€ умножением мол€рности растворенного вещества на суммарную положительную валентность:
Ќормальность = ћол€рность х „иста€ положительна€ валентность
ћиллиграммы на литр (мг/л). онцентраци€ в миллиграммах на литр Ц это отношение веса к объему. ¬ растворе с концентрацией 100 мг/л содержитс€ 100 мг растворенного вещества в одном литре раствора. онцентрацию в миллиграммах на литр зачастую путают с концентрацией в част€х на миллион (промилле), а это Ц весовое отношение. онцентрацию в миллиграммах на литр можно преобразовать в концентрацию в промилле, если известна плотность жидкости, путем делени€ концентрации в мг/л на удельный вес раствора.
„асти на миллион, промилле (ppm). ѕромилле (сокращенно ppm) Ц это концентраци€ по весу одного их химических веществ, выраженна€ в миллионных дол€х общего веса. ѕромилле обычно используют дл€ измерени€ малых концентраций. ѕромилле Ц это то же, что и дес€тична€ весова€ дол€, умноженна€ на один миллион (1 000 000), или процент веса, умноженный на 10 000. Ќапример, насыщенный солевой раствор содержит 26% соли от веса раствора, поэтому концентраци€ такого раствора в промилле будет равна 260 000 ppm (26 х 10 000 = 260 000). онцентрацию в промилле можно вычислить непосредственно по результатам титрировани€, дл€ этого концентрацию в мг/л надо разделить на удельный вес раствора.
Ёквивалентные части на миллион (EPM). EPM Ц единица веса растворенного вещества на один миллион единиц веса раствора. EPM растворенного вещества в растворителе равна концентрации промилле, разделенной на эквивалентный вес.
| онцентраци€* | ¬ес растворенного в-ва | ¬ес растворител€ | ќбъем раствора | ¬ес раствора |
| 1 m (мольный) | 1 г моль | 1 000 г | -- | 1 г моль + 1 000 г |
| 1 ћ (мол€рный) | 1 г моль | -- | 1 л | -- |
| 1 N (нормальный) | 1 г/экв. | -- | 1 л | -- |
| 100 000 мг/л | 100 000 мг | -- | 1 л | -- |
| 100 000 ppm | 100 000 мг | 900 000 | -- | 1 000 г |
“абл. 10. онцентраци€ раствора.
*—тандартного начального объема растворител€ не существует. ”дельный вес растворенного вещества либо прибавл€етс€ к удельному весу растворител€, либо некоторый объем растворител€ добавл€етс€ до получени€ требуемого объема раствора.
ѕример: дл€ простого раствора хлорида натри€ с удельным весом 1,148 г/см3 объемом 1 000 см3, который содержит 230 г растворенной соли, требуетс€ рассчитать:
а) состав в процентах веса
b) состав в процентах объема
c) мольность
d) мол€рность
e) нормальность
f) концентраци€ хлорида натри€ в мг/л
g) концентраци€ хлорида натри€ в ppm
h) концентраци€ EPM
i) весовое отношение NaCl к H2O, (фунт/фунт)
ќбщий вес р-ра = 1 000 см3 x 1,148 г/см3 = 1 148 г
¬ес воды = общий вес р-ра Ц вес соли = 1 148 - 230 = 918 г
a) —остав в % веса:
%веса NaCl = (230 ÷ 1 148) x 100 = 20,0%
%веса H2O = (918 ÷ 1 148) x 100 = 80,0%
b) —остав в % объема:
”.в. пресной воды (“=20∞C) = 0,998 г/см3
ќбъем пресной воды = (918 ÷ 0,998) = 920 см3
% объема воды = (920 ÷ 1 000) x 100 = 92%
ќбъем хлорида натри€ = 1 000 - 920 = 80 см3
% объема хлорида натри€ = (80 ÷ 1 000) x 100 = 8%
c) ћольность Ц число молей растворенного вещества на килограмм растворител€:
ћолекул€рный вес хлорида натри€: 58,44
√рамм-молей NaCl = (230 ÷ 58,44) = 3,94
“ак как в растворе только 918 г воды, а не 1 кг (1 000 г), в расчет грамм-молей необходимо внести поправку на 1 кг.
ћольность = 3,94 x (1 000 ÷ 918) = 4,287 грамм-молей NaCl на кг
d) ћол€рность Ц число молей растворенного в-ва на 1 литр растворител€:
—огласно пункту c), число грамм-молей равно 3,94 NaCl в первоначальном растворе объемом 1 000 см3 (1-l).
ћол€рность = 3,94 грамм-молей/литр
e) Ќормальность Ц грамм-эквивалент веса на литр раствора:
“ак как суммарна€ положительна€ валентность хлорида натри€ равна 1, нормальность будет равна по величине мол€рности.
Ќормальность = 3,94 грамм-эквивалентов веса/литр
f) онцентраци€ хлорида натри€ в мг/л:
“ак как 230 г (230 000 мг) хлорида натри€ растворены в 1 000 см3 раствора (1-1):
мг/л хлорида натри€ = 230 000 мг ÷ 1 l = 230 000 мг/л
g) онцентраци€ хлорида натри€ в ppmЧ весовое отношение х 1 000 000:
ppm хлорида натри€ = (230 ÷ 1 148) x 1 000 000 = 200 350 ppm
h) онцентраци€ в EPM Ч концентраци€ ppm, разделенна€ на эквивалентный вес:
EPM хлорида натри€ = (200 350 ÷ 58,44) = 3 428 EPM
i) ¬есовое отношение NaCl к H2O:
¬ес NaCl в граммах ÷ вес H2O в граммах = (230 ÷ 918) = 0,2505






