ћалодиссоциирующие изменение рЌ раствора
„астицы

 „ем зар€д и радиус иона Þ сильнее взаимодействие с Ќ2ќ Þ сильнее гидролиз.
„ем зар€д и радиус иона Þ сильнее взаимодействие с Ќ2ќ Þ сильнее гидролиз.
DЌг > 0 - эндотермический процесс.
оличественна€ характеристика гидролиза:
 |
 |
(дл€ разбавленных растворов а = с)
√идролиз Ц обратимый равновесный процесс
√ - константа гидролиза
—i Цравновесные концентрации
√ зависит от: природы реагентов и “.

 “.к. D Ќ √ > 0 Þ с температуры √ Þ
“.к. D Ќ √ > 0 Þ с температуры √ Þ
¬ыход продуктов гидролиза растет.
 ѕрименим закон разведени€ ќствальда:
ѕрименим закон разведени€ ќствальда:


 | |||||
 | |||||
 | |||||




 |
—оли, образованные сильной кислотой и сильным основанием (Na2SO4) Ц гидролизу не подвергаютс€.

 Na2SO4 Ѓ Na+ + SO42-
Na2SO4 Ѓ Na+ + SO42-
NaOH H2SO4
—ильное основание сильна€ кислота
раствор нейтральный: рЌї 7.
 |
 ƒиссоциаци€ соли: —Ќ3—ќќNa Ѓ CH3COO- + Na+
ƒиссоциаци€ соли: —Ќ3—ќќNa Ѓ CH3COO- + Na+
CH3COOH NaOH
—лаба€ кислота сильное основание
√идролиз Ц по слабому электролиту:
CH3COO- + ЌќЌ Û CH3COOH + ќЌ-
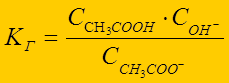 выражение константы гидролиза
выражение константы гидролиза
√идролиз многозар€дных ионов протекает ступенчато (FeCl3):
Fe3+ + ЌќЌ Û FeќЌ2+ + Ќ+ - 1-€ ступень;
FeќЌ2+ + ЌќЌ Û Fe(ќЌ)2+ + Ќ+ - 2-€ ступень;
Fe(ќЌ)2+ + ЌќЌ Û Fe(ќЌ)3 + Ќ+ - 3-€ ступень
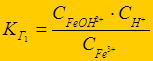


 г1> г2 > г3
г1> г2 > г3

AgNO3 Ѓ Ag+ + NO3-
 |  | ||
AgќЌ HNO3
—лабое основание сильна€ кислота
 |
Ag+ + ЌќЌ Û AgќЌ + Ќ+.
кисла€ среда рЌ< 7

- константа гидролиза

 KW
KW


ƒ(AgOH): AgOH Û Ag+ + OH-
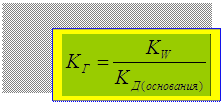
- константа гидролиза по
атиону
≈сли √ (1-ой ступени) Þ ƒ (последней ступени)
≈сли √ (последней ступени) Þ ƒ (1-ой ступени)
–асчет рЌ гидролиза по катиону:
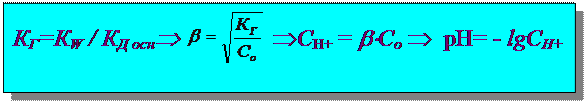

Na2S Ѓ Na+ + S2-

 Na2S Ѓ 2Na+ + S2-
Na2S Ѓ 2Na+ + S2-
NaOH H2S
—ильное основание слаба€ кислота
 √идролиз по ступен€м:
√идролиз по ступен€м:
1cт.: S2- + HOЌ Û HS- + ќЌ-
2ст.: HS- + HOЌ Û H2S + ќЌ-

√( 1ст) > √( 2ст)

 кw
кw


-константа гидролиза по аниону
г 1 Þ ƒ последней
–асчет рЌ гидролиза по аниону:

 |

NЌ4—N, –bCO3, јl2S3
NЌ4—N Û NЌ4+ + ќЌ-
√идролиз:
—N- + ЌќЌ Û Ќ—N + ќЌ-
NЌ4+ + ЌќЌ Û NЌ4ќЌ + Ќ+
NЌ4+ + —N- + Ќ2ќ Û NЌ4ќЌ + Ќ—N

 |

ƒ(NH4OH)=1,79×10-5 > ƒ(Ќ—N) =7,9×10-10 Þ
 онцентраци€ соли не вли€ет на b Þ
онцентраци€ соли не вли€ет на b Þ
|
|
|
–асчет рЌ гидролиза по катиону и аниону:
 |
≈сли в результате гидролиза образуютс€ труднорастворимые или газообразные вещества Þ гидролиз необратимым:
PbCO3 + Ќ2ќ Ѓ Pb(ќЌ)2¯ + CO2 ≠

 1) при температуры:
1) при температуры:

 D Ќ √ > 0 Þ с температурыÞ √ = b 2 × — 0;
D Ќ √ > 0 Þ с температурыÞ √ = b 2 × — 0;
 2) с разбавлением раствора (концентраци€);
2) с разбавлением раствора (концентраци€);
 3) при концентрации иона, определ€ющего среду
3) при концентрации иона, определ€ющего среду
—N- + ЌќЌ Û Ќ—N + ќЌ-
Ќ—l Ѓ Cl- + H+
«адача –ассчитать √, b и рЌ 0,01 ћ раствора 2Sќ3.
–ешение ƒиссоциаци€ сильного электролита 2Sќ3:

 2Sќ3 Ѓ 2 + + Sќ32-
2Sќ3 Ѓ 2 + + Sќ32-
ќЌ Ќ2Sќ3
—ильное основание слаба€ кислота
√идролиз по Sќ32-:
1-а€ ступень: Sќ32-+ ЌќЌ Û ЌSќ3-+ќЌ-
√1 =  = 1,59×10-7
= 1,59×10-7
2-а€ ступень: ЌSќ3- + Ќ2ќ Û Ќ2Sќ3 + ќЌ-
√2 =  = 5,9×10-13 √1 > √2
= 5,9×10-13 √1 > √2
b =  =
=  = 4×10-3 < 1 Þ расчет по приближенной формуле правомерен.
= 4×10-3 < 1 Þ расчет по приближенной формуле правомерен.
— OH- = b×c 0 = 4×10-3×10-2 = 4×10-5
рЌ = 14 + lg — OH- =14 - 4,4 = 9,6


 | |||
 |


= ѕ–јnBm, т.к. а јn¬m(тв) = const


ѕ– зависит: от природы электролита и
–астворител€, от температуры;
ѕ– не зависит от активностей ионов.
ѕ–25— Ц приведены в таблице
● ѕример: Ag2CO3(тв) Û 2Ag+(р-р) + CO32-(р-р)
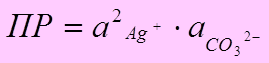
≈сли ( ) ³ ѕ–табл Ц осадок выпадает
) ³ ѕ–табл Ц осадок выпадает
≈сли ( ) < ѕ–табл Ц осадок не выпадает
) < ѕ–табл Ц осадок не выпадает
ѕ– =  = (gј m+×n × с –) n × (gB n-×m ×с –) m=
= (gј m+×n × с –) n × (gB n-×m ×с –) m=
=( gј m+)n ×( gB n-)m × nn × mm ×(с –) n+m
 |
- - растворимость
- труднорастворимого
—ильного
Ёлектролита
 если g Ѓ 1
если g Ѓ 1
● «адача. ќпределить с – MgF2, в растворе, в котором g(Mg2+) = 0,7, g(F-) = 0,96
–ешение:
MgF2 Û Mg2+ (нас.р-р) + 2F-(нас.р-р)
ѕ–(MgF2) = 4×10-9 = 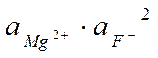
с– =  моль/л
моль/л
 |
1) ионной силы раствора Ц введение растворимого электролита, не имеющего общих ионов
 ѕ– = gMg2+ × с Mg2+ ×gF-2× с F-2
ѕ– = gMg2+ × с Mg2+ ×gF-2× с F-2

 — ионной силы раствора (I) Þ gi Þ с –
— ионной силы раствора (I) Þ gi Þ с –
Þ чтобы при “=const Þ ѕ– =const






