“аблица 1
| од специальности | є заданий |
| Cи”Ќ: 270102; 270104; 270105; 270106; 270115 | 1-20; 41-60; 61-80; 81-100; 121-140; 141-160 |
| 161-180; 181-200; 221-240; 241-260; 281-300 | |
| “ј—: 220201 | 1-20; 41-60; 61-80; 121-140; 141-160; 181-200; 201-220; 241-260; 281-300 |
| ћј и “Ѕ: 220301 | 1-20; 41-60; 61-80; 121-140; 141-160; 161-180; 181-200; 201-220; 281-300 |
| ћј и “Ѕ: 151001; 151002; 190601;190603 140401;140504 | 1-20; 41-60; 61-80; 101-120; 121-140; 141-160; 181-200; 201-220; 241-260; 281-300 |
| ћј и “Ѕ: 260601 | 1-20; 41-60; 61-80; 121-140; 101-120; 141-160; 181-200; 201-220; 241-260; 281-300 |
| Ё и ”Ѕ: | 1-20; 21-40; 41-60; 61-80; 81-100; 101-120; 121-140; 141-160; 181-200; 221-240; 241-260; 281-300 |
| ’Ќ— : | 1-20; 21-40; 41-60; 61-80; 81-100; 121-140; 141-160; 181-260 |
| ¬ыполн€етс€ по му[10]. ¬есь комплект заданий включает: зад.1 (в.1.1-1.9), зад. 2 (в. 2.1-2.5), зад. 3 (в. 3.1-3.4) ¬арианты заданий выдаютс€ преподавателем! | |
| Ќ√ и Ё: | 1-20; 20-41; 41-60; 61-80; 181-200; 221-240; 241-260; 261-280; 281-300 |
| Ќ√ и Ё: 140101; 140104 | 1-20, 21-40, 41-60, 61-80, 81-100, 101-120, 121-140, 141-160; 161(а)-180(б), 181-200, 201-220, 221-240, 241-260, 261-280, 281-300 |
| Ќ√ и Ё: 130503; 130501; 130504; 130602 (студенты «иƒќ и 2 высшее образование) | ¬ыполн€етс€ по насто€щему му (ч. I): 1-20; 21-40; 41-60; 61-80; 81-100; 101-120; 161-180 |
| ¬ыполн€етс€ по насто€щему ћ”, ч. I: 121-140; 141-160; 181-200; 221-240; 241-260 и по ћ”, ч. II [9]: 1-6 ¬арианты заданий выдаютс€ преподавателем! | |
| Ќ√ и Ё: 130503; 130501; 130504;130602 (студенты ћ»ѕѕ—) | ¬ыполн€етс€ по насто€щему му, ч. I: 1-20; 21-40; 41-60; 61-80;81-100; 101-120; 121-140; 141-160; 161-180;181-200; 201-220; 221-240; 241-260; 280-300 |
«адани€ 1-20 (таблица 2)
“ема 1. ѕериодические закон и система элементов ƒ.». ћенделеева
ѕример.
—оставьте электронную и электронно-графическую формулу, укажите валентные электроны, определите степени окислени€ серы и напишите возможные соединени€ этого элемента с водородом и кислородом.
–ешение. Ќапишем электронно-графические формулы дл€ атома серы в нормальном и возбужденном состо€ни€х: S, пор€дковый номер 16, третий период, шеста€ группа, главна€ подгруппа.
1) Ёлектронна€ формула: 16S 1s2 2s22p6 3s23p4
(Е 3s23p4 - валентные электроны)
2) Ёлектронно-графическа€ формула.
S s p d ¬=II
| ↑↓ | ↑↓ | ↑ | ↑ |
—тепень окислени€ +2 и Ц2.
S* s p d ¬=IV
| ↑↓ | ↑ | ↑ | ↑ | ↑ |
—тепень окислени€ +4
S* s p d ¬=VI
| ↑ | ↑ | ↑ | ↑ | ↑ | ↑ |
—тепень окислени€ + 6
“аким образом сера может про€вл€ть степени окислени€ 0, (+2), +4, +6, -2.
|
|
|
— кислородом сера образует кислотные оксиды состава: S+4O2-2,
S+6O3-2. — водородом: H2+1S-2.
онтрольные задани€.
ƒл€ указанных в таблице 2 элементов составьте электронные и электронно-графические формулы. ”кажите валентные электроны и степени окислени€. Ќапишите формулы возможных соединений этих элементов с кислородом и водородом. ”кажите характер оксидов, уравнени€ми реакций докажите их важнейшие химические свойства и изобразите их графические формулы.
“аблица 2
| є | Ёлементы | ѕо€сните, |
| Ca, Sc | почему эти элементы расположены в одном периоде, в разных группах и подгруппах? | |
| Si, Ti | почему эти элементы сто€т в разных периодах, в одной группе, но в разных подгруппах? | |
| P, Al | почему эти элементы сто€т в одном периоде, в разных группах, но в одних подгруппах? | |
| –, V | почему эти элементы сто€т в разных периодах, в одной группе, но в разных подгруппах? | |
| Fe, —r | у оксида железа (II) или у оксида хрома(II) сильнее выражен основной характер? | |
| Fe, Cr | у какого оксида и почему сильнее выражены кислотные свойства: FeO3 и CrO3? | |
| S, Se | почему эти элементы сто€т в разных периодах, в одной группе и подгруппе? | |
| C, Si | ” какого элемента и почему энерги€ ионизации больше? | |
| Ge, Zr | у какого оксида и почему сильнее выражены основные свойства: GeO2 и ZrO2? | |
| Mg, P, Al | как и почему измен€ютс€ кислотно-основные свойства высших оксидов в р€ду магний, алюминий, фосфор? | |
| S, Al | y какого оксида и почему сильнее выражены кислотные свойства: SO2 и Al2O3? | |
| Al, S | у какого оксида и почему сильнее выражены кислотные свойства Al2O3 и SO3? | |
| Mn, Cl | почему эти элементы сто€т в разных периодах, в одной группе, но в разных подгруппах? | |
| Ga, As | у какого оксида и почему сильнее выражены основные свойства: Ga2O3 и As2O3? | |
| Ti, Ge | у какого оксида и почему сильнее выражены основные свойства: TiO и GeO? | |
| Ti, Hf | укажите, у какого оксида и почему сильнее выражены основные свойства: Ti2O3 и Hf2O3? | |
| є15 и є28 | объ€сните, почему фосфор про€вл€ет отрицательную степень окислени€, а никель нет? | |
| є21 и є31 | укажите, к какому семейству они относ€тс€, и объ€сните, почему они сто€т в одном периоде, в одной группе, но в разных подгруппах? | |
| є33 и є23 | объ€сните, почему мышь€к образует газообразное водородное соединение, а ванадий не образует? | |
| є34 и є74 | почему эти элементы сто€т в разных периодах, в одной группе, но в разных подгруппах? |
“ема 2. ’имическа€ св€зь и строение молекул
«адани€ 21-40 (таблица 3)
ѕример:
ќпределите тип химических св€зей в молекулах Br2, HBr, KBr. ѕриведите схемы перекрывани€ электронных орбиталей при образовании ковалентных св€зей в указанных молекулах.
–ешение:
Br2: молекула брома образована из атомов одного элемента. Ёлектроотрицательность брома равна 2,8 (таблица 18), поэтому Δ электроотрицательности = 2,8 Ц 2,8 = 0, следовательно, св€зь в молекуле брома Br2 Цковалентна€ непол€рна€, (Br- Br).
HBr: Δ электроотрицательности = 2,8 - 2,1 = 0,7, т.е. <0,7 => ковалентна€ пол€рна€ св€зь, св€зующа€ электронна€ пара смещена в сторону атома брома (Hδ+→Brδ-). ѕоэтому атом водорода приобретает эффективный положительный зар€д, а брома Ц эффективный отрицательный.
|
|
|
KBr: Δ электроотрицательности = 2,8 - 0,91 = 1,89, т.е.>1,7 => св€зь преимущественно ионна€, (K-Br).
” атома брома валентными €вл€ютс€ электроны четвертого энергетического уровн€: Е4s24p5,
s p
| ↑↓ | ↑↓ | ↑↓ | ↑ |
¬=I
неспаренный электрон находитс€ на р-орбитали, электронна€ орбиталь имеет форму гантели.
ќбразование химической св€зи в молекуле Br2 можно отобразить схемой:

 |  |

 + →
+ →
Br Br Br Ч Br
” атома водорода валентными €вл€етс€ 1s-электрон, его электронна€ орбиталь имеет сферическую форму.
—хема образовани€ химической св€зи в молекуле HBr.
 |  | ||||
 | |||||


 + →
+ →
H Br H Ч Br
онтрольные задани€
ќпределите тип химических св€зей в молекулах, указанных в таблице 3. »зобразите схемы перекрывани€ валентных орбиталей при образовании ковалентных св€зей в выделенных молекулах. (ѕри необходимости используйте таблицы 15 Ц 19).
“аблица 3
| є | —оединени€ | ѕо€сните, |
| –—l3; HCl | в чем сущность образовани€ ковалентной св€зи? ака€ из следующих св€зей пол€рна: а) –ЦCl; б) ЌЦ—l? | |
| CsF, CaH2 | что называетс€ пол€рностью химической св€зи. ќтвет по€сните, исход€ из теории строени€ атома. | |
| –Ќ3, Ќ≠2 | в чем сущность механизма образовани€ ковалентной непол€рной св€зи? | |
| KCl, NH3 | в чем сущность донорно-акцепторного механизма образовани€ ковалентной св€зи? | |
| F2, H2O | в чем сущность образовани€ ковалентной св€зи? ака€ из следующих св€зей пол€рна: а) FЦF; б) OЦH? | |
| KCl, I | что называетс€ длиной химической св€зи? ак измен€етс€ длина св€зи в указанных молекулах? | |
| —Ќ4, јlCl3 | что такое гибридизаци€? ќпределите типы гибридизации в указанных молекулах | |
| јl(OH)3, —2Ќ4 | что такое направленность ковалентной св€зи? ћеханизм образовани€ π- св€зи. | |
| NaCl, H2 | что такое направленность ковалентной св€зи? ћеханизм образовани€ σ- св€зи. | |
| HCOH, CCl4 | сколько σ- и π-св€зей образует атом углерода в выделенной молекуле? акова его степень окислени€? | |
| CH3COOH, CH4 | сколько σ- и π-св€зей образует атом углерода в выделенной молекуле? акова его степень окислени€? | |
| C2H5OH, —Ќ—l3 | сколько σ- св€зей образует атом углерода в выделенной молекуле? аковы его валентность и степень окислени€? | |
| NCl3, MgCl2 | что такое SP Ц гибридизаци€. ”кажите молекулу с этим типом гибридизации. | |
| H2S, H2Se | в какой из указанных молекул значение дипольного момента больше? ќтвет по€сните. | |
| —Ќ3ќЌ, Ќ2ќ | какую химическую св€зь называют водородной? ¬ каких случа€х она осуществл€етс€? | |
| SiCl4, MgCl2 | что такое SP3 Ц гибридизаци€. ”кажите молекулу с этим типом гибридизации. | |
| јlCl3, SiCl4 | что такое SP2 Ц гибридизаци€. ”кажите молекулу с этим типом гибридизации. | |
| H2O, H2S | в какой молекуле св€зь более пол€рна? ”кажите направление дипольных моментов в этих молекулах. | |
| KF, 2S | какую химическую св€зь называют ионной? ѕокажите механизм ее образовани€. ѕо каким свойствам ионна€ св€зь отличаетс€ от ковалентной? | |
| NaAlO2, CH3Cl | сколько σ- и π-св€зей образует атом алюмини€ в выделенной молекуле? акова его степень окислени€? |
“ема 3. Ёнергетика химических процессов
«адани€ 41-60 (таблица 4)
ѕример.
–еакци€ горени€ этана выражена термодинамическим уравнением:
C2H6(г) + 3,5 O2(г) = 2 CO2(г) + 3 H2O(ж);
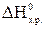 = -1559,87 кƒж/моль. ¬ычислите энтальпию образовани€ этана, если известны стандартные энтальпии образовани€:
= -1559,87 кƒж/моль. ¬ычислите энтальпию образовани€ этана, если известны стандартные энтальпии образовани€:
CO2 (г)  = -393,57 кƒж/моль
= -393,57 кƒж/моль
H2O(ж)  = -285,84 кƒж/моль
= -285,84 кƒж/моль
|
|
|
–ешение: »спользу€ следствие из закона √есса, можно записать:
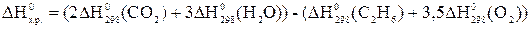 , откуда
, откуда 
ѕодставим табличные значени€ термодинамических величин и зна€, что энтальпии образовани€ простых веществ равны нулю, получаем:

–еакци€ горени€ этана экзотермическа€ т.к. 
онтрольные задани€.
Ќа основании стандартных энтальпий образовани€ и абсолютных стандартных энтропий (таблица 20) соответствующих веществ вычислите изменение энергии √иббса и температуру равновесного состо€ни€ указанной в таблице 4 реакции, а так же возможность протекани€ данной реакции в пр€мом направлении при стандартных услови€х.
“аблица 4
| є | ”равнение |
| CO2(г) + 4H2(г) = CH4(г) + 2H2O(ж) | |
| Fe2O3(к) + 3H2(г) = 2Fe(к) + 3H2O(г) | |
| PCl5(г) = PCl3(г) + Cl2(г) | |
| CO(г) + 3H2(г) = CH4(г) + H2O(г) | |
| C2H4(г) + 3O2(г) = 2H2O(ж) + 2CO2(г) | |
| 2CH4(г) = C2H2(г) + 3H2(г) | |
| C(к) + O2(г) = CO2 (г) | |
| јl2O3(к) + H2(г) = Al(к) + H2O(г) | |
| N2(г) + 3H2(г) = 2NH3(г) | |
| 2CO(г) + O2(г) = 2CO2(г) | |
| CO(г) + H2O(ж) = CO2(г) + H2(г) | |
| CO(г) + Cl2(г) = 2COCl2(г) | |
| 2C2H6(г) + 7O2(г) = 6H2O(ж) + 4CO2(г) | |
| CaO(к) + H2O(ж) = 2Ca(OH)2(к) | |
| CaCO3(к) = CaO(к) + CO2(г) | |
| 2H2O2(ж) = 2H2O(ж) + O2(г) | |
| ќ2(г) +4HCl(г) = 4HCl(г) + 2H2O(ж) | |
| NH4Cl(к) + NaOH(к) = NaCl(к) + NH3(г) + H2O(г) | |
| 2ZnS(к) + 3O2(г) = 2ZnO(к) + 2SO2(г) | |
| 2C2H2(г) + 5O2(г) = 2H2O(ж) + 4CO2(г) |
“ема 4. ’имическа€ кинетика, катализ и химическое равновесие
«адани€ 61 Ц 80 (таблица 5)
ѕример 1.
ак изменитс€ скорость химической реакции между азотом и водородом N2(г) + 3H2(г) 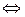 2NH3(г), если, не измен€€ температуру, уменьшить давление системы в 2 раза?
2NH3(г), если, не измен€€ температуру, уменьшить давление системы в 2 раза?
–ешение: ѕо закону действующих масс
V = k[N2][H2]3.
ƒл€ уменьшени€ давлени€ системы в 2 раза, объем ее следует увеличить в 2 раза. ѕри этом во столько же раз уменьшитьс€ концентраци€ каждого из участвующих в реакции компонентов. онцентраци€ азота будет равна [N2]/2 и водорода [H2]/2. —корость химической реакции при этих услови€х будет:
VТ = k[N2/2][H2/2]3 = 1/16k[N2][H2]3
и соотношение скоростей
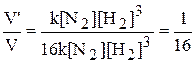
ѕри уменьшении давлени€ в системе в 2 раза скорость химической реакции между азотом и водородом уменьшитьс€ в 16 раз.
ѕример 2.
Ќапишите выражение константы равновеси€ дл€ гетерогенной системы
SO2(г) + 2H2S(г) 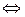 3S(т) + 2H2O(г)
3S(т) + 2H2O(г)
ак следует изменить концентрацию и давление, чтобы сместить равновесие в сторону обратной реакции?
–ешение: онцентраци€ твердого вещества в уравнении закона действующих масс не учитываетс€, следовательно:
Vпр = k1[SO2][H2S]2
Vобр = k2[H2O]2

–уководству€сь принципом Ће-Ўателье, который гласит: Ђ≈сли на систему, наход€щуюс€ в равновесии, оказать какое-либо воздействие, то в результате протекающих в ней процессов равновесие сместитс€ в направлении, уменьшающем это воздействиеї, приходим к выводу, что дл€ смещени€ равновеси€ в сторону обратной реакции надо увеличить концентрацию вод€ного пара или уменьшить концентрацию SO2 и H2S.
онтрольные задани€
ƒл€ всех заданий в таблице 5 дайте обоснованный ответ, а дл€ задач с определением направлени€ смещени€ равновеси€, напишите выражение константы равновеси€ ( р).
“аблица 5
| є | ”словие задани€ |
ак изменитс€ скорость пр€мой химической реакции
Ќ2(г) + —l2(г) 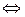 2 HCl (г), если, не измен€€ температуру, уменьшить давление системы в 3 раза? 2 HCl (г), если, не измен€€ температуру, уменьшить давление системы в 3 раза?
| |
ак изменитс€ скорость химической реакции между кислородом и водородом 2Ќ2(г) + ќ2(г) 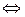 2H2ќ (г), если, не измен€€ температуру, уменьшить концентрацию исходных веществ в 4 раза? 2H2ќ (г), если, не измен€€ температуру, уменьшить концентрацию исходных веществ в 4 раза?
| |
¬о сколько раз надо увеличить концентрации —ќ, чтобы скорость пр€мой химической реакции 2—ќ(г) 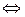 —(т)+—ќ2(г) увеличилась в 4 раза? —(т)+—ќ2(г) увеличилась в 4 раза?
| |
¬о сколько раз надо увеличить концентрации Sќ2, чтобы скорость пр€мой химической реакции 2Sќ2(г) + O2(г) 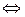 2Sќ3(г) увеличилась в 2 раза? 2Sќ3(г) увеличилась в 2 раза?
| |
ƒействием каких факторов можно сместить равновесие в сторону продуктов реакции Ќ2(г) + 3N2 (г) 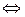 2NЌ3(г)? ∆Ќ = -92 кƒж/моль 2NЌ3(г)? ∆Ќ = -92 кƒж/моль
| |
¬о сколько раз надо увеличить концентрации ќ2, чтобы скорость пр€мой химической реакции 2Sќ2(г) + O2(г) 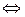 2Sќ3(г) увеличилась в
3 раза? 2Sќ3(г) увеличилась в
3 раза?
| |
ƒействием каких факторов можно сместить равновесие химической реакции N2(г) +O2 (г) 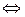 2NO(г) вправо? ∆Ќ =180 кƒж/моль? 2NO(г) вправо? ∆Ќ =180 кƒж/моль?
| |
| ак следует изменить концентрацию, температуру и давление в реакции горени€ углерода, чтобы сместить равновесие в сторону обратной реакции? | |
¬о сколько раз изменитс€ скорость пр€мой реакции
ј(г) + 2¬(г) 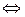 ј¬2 (г) при увеличении давлени€ в 2 раза? ј¬2 (г) при увеличении давлени€ в 2 раза?
| |
ƒействием каких факторов можно сместить равновесие в реакции
N2 (г) + 3ћg(т) 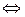 Mg3N2(т) в сторону продуктов реакции? ∆Ќ < 0 Mg3N2(т) в сторону продуктов реакции? ∆Ќ < 0
| |
ƒействием каких факторов можно сместить равновесие в сторону продуктов реакции —2Ќ4 + 3ќ2 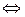 2—ќ2 + 2Ќ2ќ? ∆Ќ < 0 2—ќ2 + 2Ќ2ќ? ∆Ќ < 0
| |
| ¬о сколько раз уменьшитс€ скорость химической реакции при уменьшении температура с 36о— до 28о—, если температурный коэффициент равен 2? | |
ƒействием каких факторов можно сместить равновесие в химической реакции N2(г) + 6Li(т) 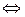 2Li3N(т)+ 39 кƒж/моль в сторону продуктов реакции? 2Li3N(т)+ 39 кƒж/моль в сторону продуктов реакции?
| |
| ¬о сколько раз увеличитс€ скорость химической реакции при изменении температуры от 35 до 75о—, если температурный коэффициент равен 2? | |
ƒействием каких факторов можно сместить равновесие химической реакции Fe3O4(т) +4CO(г) 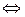 3Fe(т) + 4CO2(г) вправо? ∆Ќ < 0. 3Fe(т) + 4CO2(г) вправо? ∆Ќ < 0.
| |
ƒействием каких факторов можно сместить равновесие в сторону пр€мой химической реакции Fe(т) +2HCl(ж) 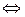 FeCl2(ж) + Ќ2(г)? ∆Ќ > 0 FeCl2(ж) + Ќ2(г)? ∆Ќ > 0
| |
| —корость химической реакции увеличилась в 81 раз при увеличении температуры на 30о—. ќпределите величину температурного коэффициента. | |
| „ему равен температурный коэффициент, если при увеличении температуры на 30о— скорость химической реакции возрастает в 27 раз? | |
| ¬о сколько раз увеличитс€ скорость химической реакции при повышении температуры на 45о—, если температурный коэффициент равен 2? | |
| „ему равен температурный коэффициент, если при увеличении температуры на 30о— скорость химической реакции возрастает в 21 раз? |
|
|
|
“ема 5. »стинные растворы и способы выражени€ концентраций
растворов
«адани€ 81 Ц 100 (таблица 6)
онтрольные задани€
–ешите задачи, приведенные в таблице 6. ƒайте полное объ€снение хода решени€, использу€ таблицу 21.
“аблица 6
| є | ”словие задани€ |
| ќпределите массовую долю глюкозы (C6H12O6) в растворе, содержащем 280 г воды и 40 г глюкозы. | |
| 1 см3 25% раствора NaCl содержит 0,458 г растворенного вещества. акова плотность этого раствора? | |
| —колько граммов 30% раствора NaCl надо добавить к 300 г воды, чтобы получить 10% раствор соли? | |
| ќпределите массу нитрата натри€ необходимую дл€ приготовлени€ 300 см3 0,2 ћ раствора. | |
| акой объем 6 ћ раствора сол€ной кислоты нужно вз€ть, чтобы получить 25 см3 2,5 ћ раствора Ќ—l? | |
| ¬ычислите мол€рную концентрацию и мол€рную концентрацию эквивалента 20% раствора хлорида кальци€ плотностью 1,178 г. | |
| „ему равна мол€рна€ концентраци€ эквивалента 30% раствора NaOH плотностью 1,328 г/см3? 1 л этого раствора прибавили 5 л воды. ¬ычислите процентную концентрацию полученного раствора. | |
| 3 л 10% HNO3 (1,054 г/см3) прибавили 5 л 2% раствора HNO3 (1,009 г/см3). ¬ычислите массовую долю и его мол€рную концентрации. | |
| ¬ычислите мол€рную и мол€льную концентрации 20,8% раствора HNO3 плотностью 1,12 г/см3. —колько граммов кислоты содержитс€ в 4 л этого раствора? | |
| ¬ычислите мол€рную и мол€льную концентрации 16% раствора хлорида алюмини€ плотностью 1,149 г/см3. | |
| —мешали 10 см3 20% раствора HNO3 (1,2 г/см3) и 100 см3 30% раствора HNO3 (1,22 г/см3). ¬ычислите массовую долю полученного раствора. | |
| акой объем 50% раствора ќЌ (1,5 г/см3) нужно вз€ть дл€ приготовлени€ 1 дм3 6% раствора ќЌ (1,042 г/см3) | |
| акой объем 0,3 ћ раствора сол€ной кислоты необходимо вз€ть дл€ нейтрализации 40 см3 раствора гидроксида натри€, содержащего 0,32 г вещества? | |
| —колько см3 0,1 ћ раствора сол€ной кислоты необходимо вз€ть дл€ нейтрализации 1дм3 25% раствора гидроксида кали€ (1,2 г/см3)? | |
| —колько грамм HNO3, содержалось в растворе, если на нейтрализацию этого раствора потребовалось 35 см3 0,1 ћ раствора NaOH? | |
| акую массу NaNO3 нужно растворить в 300 г воды, чтобы получить 20% раствор соли? | |
| —мешали 300 г 20% раствора с 500 г 46% раствора хлорида натри€. ќпределите массовую долю полученного раствора. | |
| ќпределите массовую долю раствора, полученного смешиванием 247 граствора с массовой долей 62% и 145 г раствора с массовой долей 18%. | |
| »з 700 г 62% раствора серной кислоты выпариванием удалили 200 г воды. ќпределите массовую долю полученного раствора. | |
| »з 10 кг 20% (по массе) раствора при охлаждении выделилось 400 г растворенного вещества. „ему равна массова€ дол€ этого вещества в оставшемс€ растворе? |
|
|
|
“ема 6. ‘изические свойства растворов
«адани€ 101 Ц 120 (таблица 7)
ѕример.
¬ычислите температуры кристаллизации и кипени€ 2% водного раствора глюкозы (—6Ќ12ќ6).
–ешение: —огласно 2 закону –аул€ понижение температуры кристаллизации и повышение температуры кипени€ раствора (Δt) по сравнению с температурами кристаллизации и кипени€ растворител€ выражаютс€ уравнением:
 , (1)
, (1)
где k-криоскопическа€ или эбуллиоскопическа€ посто€нна€.
ƒл€ воды они соответственно равны 1,86 и 0,52о (таблица 23);
M и m Ц соответственно масса растворенного вещества и его мольна€ масса;
m1 Ц масса растворител€.
ѕонижение температуры кристаллизации 2% раствора глюкозы —2Ќ12ќ6 находим из формулы (1).
 .
.
¬ода кристаллизируетс€ при 0о—, следовательно, температура кристаллизации равна 0 Ц 0,21= Ц 0,21о—.
»з формулы (1) находим и повышение температуры кипени€ 2% раствора

¬ода кипит при 100о—, следовательно, температура кипени€ этого раствора 100 + 0,06 о— = 100,6о—.
онтрольные задани€.
–ешите задачи, приведенные в таблице 7. ƒайте полное объ€снение хода решени€, использу€ таблицу 22.
“аблица 7
| є | ”словие задани€ |
| ¬ычислите осмотическое давление раствора, содержащего 16 г сахарозы (—12Ќ22ќ11) в 350 г воды при 293 . ѕлотность раствора считать равной единице. | |
| ѕри 25о— осмотическое давление раствора, содержащего 2,8 г высокомолекул€рного органического вещества в 200 см 3 раствора равно 0,07 кѕа. ќпределите мол€рную массу растворенного вещества. | |
| ѕри 315 давление насыщенного пара над водой равно 8,2 кѕа. Ќа сколько понизитс€ давление пара при указанной температуре, если в 100 г воды растворить 36 г глюкозы (—6Ќ12ќ6)? | |
| Ќа сколько градусов повыситс€ температура кипени€ воды, если в 100 г воды растворить 9 г глюкозы (—6Ќ12ќ6)? | |
| ѕри растворении 5 г вещества в 200 г воды получаетс€ не провод€щий тока раствор, кристаллизующийс€ при (Ц1,45 о—). ќпределите массу растворенного вещества. | |
| –аствор, содержащий 5,4 г неэлектролита в 200 г воды, кипит при 100,078 о—. ¬ычислите мольную массу растворенного вещества. | |
| ¬ычислите массовую долю водного раствора сахара (—12Ќ22ќ11), зна€, что температура кристаллизации раствора равна (Ц0,93 о—). | |
| ¬ычислите температуру кристаллизации раствора, содержащего 5 г мочевины (NH2)2CO в 150 г воды. | |
| –аствор, содержащий 3,04 г камфоры (—10Ќ16ќ) в 100 г бензола кипит при 80,7 о—. “емпература кипени€ бензола 80,20 о—. ¬ычислите его эбуллиоскопическую константу. | |
| ¬ычислите процентную концентрацию водного раствора глицерина (—3Ќ5(ќЌ)3), зна€, что этот раствор кипит при 100,39 о—. | |
| –аствор, полученный при растворении 2,25 г неэлектролита в 250 см3 воды кристаллизируетс€ при 0,279 о—. ¬ычислите мол€рную массу неэлектролита. | |
| ¬ычислите температуру кипени€ 5% раствора камфоры (—10Ќ16ќ) в бензоле, если температура кипени€ бензола равна 80,2 о—. | |
| –аствор, содержащий 25,65 г некоторого неэлектролита в 300 г воды кристаллизируетс€ при (-0,465 о—). ¬ычислите мол€льную массу растворенного вещества, если криоскопическа€ константа воды 1,86 о—. | |
| ¬ычислите криоскопическую константу уксусной кислоты, зна€ что раствор содержащий 4,25 г антрацена (—14Ќ10) в 100 г уксусной кислоты, кристаллизируетс€ при 15,718 о—. “емпература кристаллизации уксусной кислоты 16,65 о—. | |
| ѕри растворении 4,86 г серы в 60 г бензола температура кипени€ его повысилась на 0,81 о—. —колько атомов содержит молекула серы в этом растворе. | |
| “емпература кристаллизации раствора, содержащего 66,3 г некоторого неэлектролита в 500 г воды равна (-0,58 о—). ¬ычислите мол€рную массу растворенного вещества. | |
| акую массу анилина (—6Ќ5NЌ2) следует растворить в 50 г бензола, чтобы температура кипени€ раствора была выше температуры кипени€ бензола на 0,53 оC? | |
| ¬ычислите температуру кристаллизации 2% раствора этилового спирта (—2Ќ5ќЌ). | |
| —колько грамм мочевины ((NH2)2CO) следует растворить в 75 г воды, чтобы температура кристаллизации раствора понизилась на 0,465 о—. | |
| ¬ычислите массовую долю глюкозы (—6Ќ12ќ6) в растворе, зна€, что он кипит при 100,26 о—. |






