Ѕазовый уровень сложности
1. ќкислительно-восстановительной €вл€етс€ реакци€, уравнение которой
| 1) | H2S + 2KOH = K2S + 2H2O |
| 2) | Feќ+ 2HCl = FeCl2 + H2O |
| 3) | SO3 + —aO = CaSO4 |
| 4) | Feќ + CO = Fe + CO2 |
2. ѕроцесс окислени€ отражен схемой
| 1) | SO3 → SO 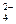
| 3) | S → SO2 |
| 2) | SO 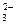 → SO2 → SO2
| 4) | SO 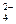 → S → S
|
3. ѕроцесс окислени€ отражен схемой
| 1) | NO2 → NO 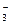
| 3) | NO2 → NO 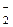
|
| 2) | NO 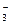 → N2 → N2
| 4) | N2O5 → NO 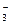
|
4. ѕроцесс восстановлени€ отражен схемой
| 1) | NO 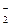 → NO → NO 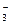
| 3) | NO2 → NO 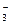
|
| 2) | NO 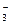 → N2ќ → N2ќ
| 4) | N2O3 → NO 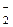
|
5. ѕроцесс восстановлени€ отражен схемой
| 1) | —O2 → —O 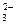
| 3) | —O2 → —ќ |
| 2) | — → —O2 | 4) | —ќ → —ќ2 |
6. Pеакции, уравнение которой
SO2 + Cl2 +2H2O = H2SO4 + 2HCl,
соответствует схема изменени€ степени окислени€ серы
| 1) | S+2 → S+4 | 2) | S+4 → S+6 | 3) | S+4 → S+2 | 4) | S-2 → S+4 |
7. ѕроцесс восстановлени€ отражен схемой
| 1) | —r2O3 → —rO 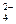
| 3) | —r2O 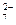 → —rO → —rO 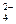
|
| 2) | —rO 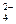 → —r2O → —r2O 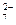
| 4) | —r2O 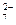 → —r3+ → —r3+
|
8. —хеме превращений
NO 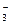 → NO2 → NO
→ NO2 → NO 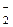 → N2O
→ N2O
соответствует последовательность изменени€ степени окислени€ азота
| 1) | N+3 → N+2 → N+3 → N+1 | 3) | N+5 → N+3 → N+4 → N+2 |
| 2) | N+5 → N+4 → N+3 → N+1 | 4) | N+5 → N+3 → N+4 → N+1 |
9. —хеме превращений
SO 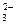 → SO2 → SO
→ SO2 → SO 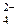 → H2S
→ H2S
соответствует последовательность изменени€ степени окислени€ серы
| 1) | S+4 → S+4 → S+6 → S+2 | 3) | S+4 → S+4 → S+6 → S-2 |
| 2) | S+6 → S+4 → S+6 → S-2 | 4) | S+6 → S+4 → S+4 → S+2 |
10. —хеме превращений
SO3 → SO 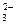 → SO
→ SO 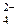 → H2S
→ H2S
соответствует последовательность изменени€ степени окислени€ серы
| 1) | S+4 → S+4 → S+6 → S+2 | 3) | S+6 → S+4 → S+4 → S+2 |
| 2) | S+6 → S+4 → S+6 → S-2 | 4) | S+4 → S+4 → S+6 → S-2 |
11. ѕоследовательности изменени€ степени окислени€ азота
N+4 → N+5 → N0 →N-3
соответствует схема превращений
| 1) | NO2 → NO 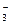 → N2 → NH3 → N2 → NH3
|
| 2) | NO2 → NO 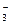 → N2 → N2ќ3 → N2 → N2ќ3
|
| 3) | NO 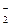 → NO → NO 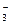 → N2 → NH3 → N2 → NH3
|
| 4) | NO 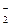 → NO → NO 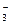 → N2 → N2ќ3 → N2 → N2ќ3
|
12. ѕоследовательности изменени€ степени окислени€ серы
S+4 → S+4 → S-2 → S+6
соответствует схема превращений
| 1) | SO 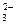 → SO3 → SO2 → SO → SO3 → SO2 → SO 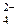
|
| 2) | SO2 → SO 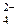 → H2S → SO3 → H2S → SO3
|
| 3) | SO 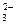 → SO → SO 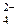 → H2S → SO3 → H2S → SO3
|
| 4) | SO2 → SO 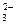 → H2S → SO → H2S → SO 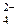
|
13. ¬ схеме превращений
SO2 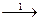 SO
SO 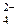
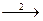 S
S 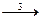 SO
SO 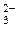
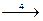 H2S
H2S
сера про€вл€ет восстановительные свойства на стади€х
|
|
|
| 1) | 1 и 2 | 2) | 1 и 3 | 3) | 3 и 4 | 4) | 2 и 4 |
14. ¬ схеме превращений
NO2 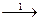 NO
NO 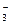
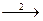 N2
N2 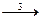 NH3
NH3 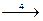 NO
NO
азот про€вл€ет окислительные свойства на стади€х
| 1) | 1 и 2 | 2) | 1 и 3 | 3) | 2 и 3 | 4) | 3 и 4 |
15. ¬ схеме превращений
MnO2 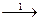 Mn2+
Mn2+ 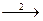 MnO2
MnO2 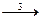 MnO
MnO 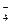
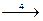 MnO
MnO 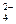
марганец про€вл€ет восстановительные свойства на стади€х
| 1) | 1 и 4 | 2) | 2 и 3 | 3) | 2 и 4 | 4) | 3 и 4 |
16. ¬ схеме превращений
Cr(OH)3 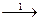 CrO
CrO 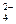
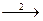 Cr2O
Cr2O 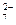
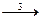 Cr3+
Cr3+ 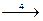 Cr
Cr
хром про€вл€ет окислительные свойства на стади€х
| 1) | 1 и 3 | 2) | 2 и 3 | 3) | 2 и 4 | 4) | 3 и 4 |
17. —ера про€вл€ет окислительные свойства при взаимодействии с
| 1) | HNO3 | 2) | O2 | 3) | Fe | 4) | Cl2 |
18. ”глерод про€вл€ет восстановительные свойства при взаимодействии
| 1) | —а | 2) | H2 | 3) | Al | 4) | FeO |
19. ‘осфор про€вл€ет окислительные свойства при взаимодействии с
| 1) | O2 | 2) | HNO3 | 3) | Mg | 4) | Cl2 |
20. “олько окислителем за счет атомов марганца может быть соединение, формула которого
| 1) | MnO2 | 2) | K2MnO4 | 3) | KMnO4 | 4) | Mn2O3 |
21. “олько восстановителем за счет атомов азота может быть соединение, формула которого
| 1) | Ca3N2 | 2) | KNO2 | 3) | KNO3 | 4) | N2O3 |
22. “олько восстановителем может быть
| 1) | C | 2) | N2 | 3) | Al | 4) | Si |
23. “олько окислительные свойства за счет атомов серы про€вл€ет
| 1) | сера | 3) | сероводород |
| 2) | сульфат натри€ | 4) | сернистый газ |
24. “олько восстановительные свойства за счет атомов серы про€вл€ет
| 1) | серниста€ кислота | 3) | серна€ кислота |
| 2) | сероводород | 4) | сернистый газ |
25. ќкислительно-восстановительную двойственность за счет атомов азота может про€вл€ть
| 1) | NH4Cl | 2) | KNO3 | 3) | N2O5 | 4) | ЌNO2 |
26. ќкислительно-восстановительную двойственность за счет атомов серы может про€вл€ть
| 1) | SO3 | 2) | H2SO3 | 3) | K2S | 4) | K2SO4 |
27. ќкислительно-восстановительную двойственность за счет атомов марганца может про€вл€ть
| 1) | MnO4 | 2) | Mn2O7 | 3) | MnO2 | 4) | Mn |
28. ќкислительно-восстановительную двойственность за счет атомов хрома может про€вл€ть
| 1) | K2CrO4 | 2) | K2Cr2O7 | 3) | Cr(OH)3 | 4) | CrO3 |
29. ¬ реакции оксида железа(III) c оксидом углерода(II) окислительные свойства про€вл€ет
| 1) | Fe+3 | 2) | C+2 | 3) | O-2 | 4) | Fe |
30. ¬осстановительные свойства углерод про€вл€ет в реакции, уравнение которой
| 1) | Ca + 2C = CaC2 | 3) | — + 2Ќ2 = —Ќ4 |
| 2) | 2— + SiO2 = Si + 2CO | 4) | 3— + 4Al = Al4C3 |
31. ќкислительно-восстановительную двойственность сера про€вл€ет в реакции, уравнение которой
| 1) | S + 6HNO3 = H2SO4 + 6NO2 + 2H2O |
| 2) | 3S + 6NaOH = 2Na2S + Na2SO3 + 3H2O |
| 3) | 2S + C = CS2 |
| 4) | S + 3F2 = SF6 |
32. ќкислительно-восстановительную двойственность хлор про€вл€ет в реакции, уравнение которой
| 1) | Cl2 + 2KBr = 2KCl + Br2 |
| 2) | Cl2 + CH4 = CH3Cl + HCl |
| 3) | Cl2 + H2O = HCl + HClO |
| 4) | 3Cl2 + 2Fe = 2FeCl3 |
33. ќкислительные свойства —ќ2 про€вл€ет в реакции, уравнение которой
| 1) | —O2 + 2NaOH = Na2CO3 + H2O |
| 2) | —O2 + 2Mg = C + 2MgO |
| 3) | —O2 + MgO = MgCO3 |
| 4) | —O2 + H2O + CaCO3 = Ca(HCO3)2 |
34. ќкислительные свойства Sќ2 про€вл€ет в реакции, уравнение которой
|
|
|
| 1) | SO2 + 2KOH = K2SO3 + H2O |
| 2) | SO2 + NO2 = SO3 + NO |
| 3) | SO2 + C = S + CO2 |
| 4) | SO2 + CaO = CaSO3 |
35. ¬осстановительные свойства Sќ2 про€вл€ет в реакции, уравнение которой
| 1) | SO2 + CaO = CaSO3 |
| 2) | SO2 + NO2 = SO3 + NO |
| 3) | SO2 + 2H2S = 3S + 2H2O |
| 4) | SO2 + 2NaOH = Na2SO3 + H2O |
36. ¬ реакции цинка с концентрированной серной кислотой окислителем и восстановителем соответственно €вл€ютс€
| 1) | H+ и Zn0 | 2) | SO 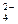 и Zn0 и Zn0
| 3) | Zn0 и SO 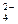
| 4) | Zn0 и H+ |
37. ¬ реакции цинка с концентрированной азотной кислотой окислителем и восстановителем соответственно €вл€ютс€
| 1) | Ќ+ и Zn0 | 2) | Zn0 и NO 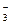
| 3) | NO 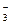 и Zn0 и Zn0
| 4) | Zn0 и H+ |
38. ¬ реакции цинка с разбавленной серной кислотой окислителем и восстановителем соответственно €вл€ютс€
| 1) | Zn0 и SO 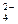
| 2) | H+ и Zn0 | 3) | SO 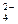 и Zn0 и Zn0
| 4) | Zn0 и Ќ+ |
39. ќксид железа(II) про€вл€ет восстановительные свойства при взаимодействии с
| 1) | сол€ной кислотой |
| 2) | разбавленной серной кислотой |
| 3) | концентрированной азотной кислотой |
| 4) | оксидом углерода(II) |
40. ќксид серы(IV) про€вл€ет восстановительные свойства при взаимодействии с
| 1) | гидроксидом натри€ |
| 2) | оксидом кальци€ |
| 3) | сероводородом |
| 4) | хлором |
41. —хеме превращени€ Cr+2 → Cr+3 соответствует реакци€, уравнение которой
| 1) | CrCl2 + 2NaOH = Cr(OH)2 + 2 NaOH |
| 2) | 4CrCl2 + 4HCl + O2 = 4CrCl3 + 2H2O |
| 3) | CrO + H2SO4 = CrSO4 + H2O |
| 4) | Crќ + 2HCl = CrCl2 + H2O |
42. —хеме превращени€ S+6 → S+4 соответствует реакци€, уравнение которой
| 1) | H2SO4 + 2NaOH = Na2SO4 + 2 H2O |
| 2) | 5H2SO4(конц) + 4Zn = 4ZnSO4 + H2S + 4H2O |
| 3) | 2H2SO4(конц) + Cu = CuSO4 + SO2 + 2H2O |
| 4) | Na2SO3 + 2HCl = 2NaCl + SO2 + H2O |
43. —хеме превращени€ N+5 → N+4 соответствует реакци€, уравнение которой
| 1) | HNO3 + NaOH = NaNO3 + H2O |
| 2) | 8HNO3(разб) + 3Cu = 3Cu(NO3)2 + 2NO + 4H2O |
| 3) | 2KNO3 = 2KNO2 + O2 |
| 4) | 2HNO3(конц) + Ag = AgNO3 + NO2 + H2O |
44. —хеме превращени€ Fe+3 → Fe+2 соответствует реакци€, уравнение которой
| 1) | FeCl3 + 3NaOH = Fe(OH)3 + 3NaCl |
| 2) | 2FeCl3 + H2S = 2FeCl2 + S + 2HCl |
| 3) | 2Fe(OH)3 = Fe2O3 + 3H2O |
| 4) | Fe3O4 + 4H2 = 3Fe + 4H2O |
45. »сход€ из теории окислительно-восстановительных процессов, укажите схему невозможной реакции
| 1) | SO2 + H2S → S + H2O |
| 2) | S + H2SO4 → SO2 + H2O |
| 3) | S + H2SO4 → H2S + H2O |
| 4) | H2SO4 + H2S → S + H2O |
46. »сход€ из теории окислительно-восстановительных процессов, укажите схему невозможной реакции
| 1) | HNO2 + KMnO4 + H2SO4 → HNO3 + MnSO4 + K2SO4 + H2O |
| 2) | KMnO4 + NaNO3 + H2SO4 → MnSO4 + NaNO2 + K2SO4 + H2O |
| 3) | KMnO4 + SO2 + H2O → MnSO4 + H2SO4 + K2SO4 |
| 4) | KMnO4 + HBr → Br2 + MnBr2 + KBr + H2O |
47. »сход€ из теории окислительно-восстановительных процессов, укажите схему невозможной реакции
| 1) | SO2 + K2Cr2O7 + H2SO4 → Cr2(SO4)3 + K2SO4 + H2O |
| 2) | NaI + K2Cr2O7 + H2SO4 → I2 + Cr2(SO4)3 + K2SO4 + Na2SO4 + H2O |
| 3) | K2Cr2O7 + Fe + H2SO4 → Cr2(SO4)3 + Fe2(SO4)3 + K2SO4 + H2O |
| 4) | KI + K2Cr2O7 + H2SO4 → I2 + 2CrO4 + K2SO4 + H2O |






