ћ»Ќ»—“≈–—“¬ќ ќЅ–ј«ќ¬јЌ»я » Ќј” » –‘
‘≈ƒ≈–јЋ№Ќќ≈ ј√≈Ќ—“¬ќ ѕќ ќЅ–ј«ќ¬јЌ»ё
»– ”“— »… √ќ—”ƒј–—“¬≈ЌЌџ… “≈’Ќ»„≈— »…
”Ќ»¬≈–—»“≈“
’»ћ»я
”„≈ЅЌќ≈ ѕќ—ќЅ»≈ ƒЋя ѕ–ј “»„≈— »’ «јЌя“»…
»«ƒј“≈Ћ№—“¬ќ
»ркутского государственного технического университета
¬¬≈ƒ≈Ќ»≈
ѕри изучении курса химии большое значение имеет приобретение навыков в решении задач, что €вл€етс€ одним из критериев прочного усвоени€ теоретических и практических знаний. ѕоэтому после каждой темы приведены примеры решени€ типовых задач и варианты индивидуальных заданий, которые студент должен выполнить.
”чебное пособие дл€ практических зан€тий студентов 1 курса технических направлений и специальностей.
—оставили: —.—. ЅочкарЄва, ¬.√. —оболева- »ркутск: »зд-во »р√“”, 2014. - 179 c.
ѕособие включает задачи по наиболее важным разделам курса общей химии, решение которых способствует усвоению и закреплению изучаемого материала.
–азделы имеют краткое теоретическое введение, методические рекомендации по решению типовых задач, задачи дл€ самосто€тельного решени€. ѕособие также включает справочный материал.
Ѕиблиогр. 7 назв. “абл. 5.
–ецензент: д-р хим. наук, профессор кафедры “ехнологии продуктов питани€ и химии »ркутского государственного технического университета ё.Ќ. ѕожидаев.
ќ√Ћј¬Ћ≈Ќ»≈
| ¬ведение........................................................................................ | |
| 1. ќсновные классы неорганических соединений............... | |
| 2. Ёквивалент. ћол€рна€ масса эквивалентовЕЕЕЕЕ. | |
| 3. —троение атомаЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ 4. ѕериодическа€ система элементов ƒ.». ћенделееваЕ.. 5. ’имическа€ св€зь и строение молекулЕЕЕЕЕЕЕ.. 6. Ёнергетика и направление химических процессовЕЕ.. | |
| 7. —корость химической реакции........................................... | |
| 8. атализ................................................................................. | |
| 9. ’имическое равновесие...................................................... | |
| 10.—пособы выражени€ концентрации растворовЕЕЕЕ.. | |
| 11. –еакции в растворах электролитов.................................... | |
| 12. √идролиз солей.................................................................... | |
| 13. оллоидные растворы....................................................... | |
| 14. ќкислительно-восстановительные реакции................... | |
| 15. оррози€ металлов............................................................ | |
| 16. Ёлектролиз......................................................................... | |
| 17. ’имические свойства металлов....................................... | |
| 18. омплексные соединени€................................................ | |
| 19. S -металлы.......................................................................... | |
| 20. ∆есткость воды................................................................. | |
| 21. јлюминий, олово, свинец................................................ | |
| 22. ћеталлы подгрупп меди и цинка.................................... | |
| 23. ’ром................................................................................... | |
| 24. ћарганец............................................................................ | |
| 25. ∆елезо, кобальт, никель................................................... | |
| 26. √алогены............................................................................ | |
| 27. ислород. ѕероксид водорода......................................... | |
| 28. —ера..................................................................................... | |
| 29. јзот..................................................................................... | |
| 30. ”глерод, кремний............................................................. | |
| 31. ”глеводороды.................................................................... | |
| 32. —пирты, альдегиды, кетоны............................................. | |
| 33. ќрганические кислоты...................................................... | |
| 34. ачественный анализ металлов....................................... | |
| 35. ачественные реакции на анионы................................... | |
| 36. оличественный анализЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| «аключение.................................................................................... | |
| Ѕиблиографический список.......................................................... | |
| ѕриложение (“абл. 1, табл. 2, табл. 3, табл. 4, табл. 5)........... |
ќсновные классы неорганических соединений
|
|
|
“еоретическое введение
¬се вещества дел€тс€ на простые и сложные. —ложные вещества подраздел€ютс€ на классы: оксиды, кислоты, основани€, соли.
ќксиды Ц это сложные вещества, состо€щие из двух элементов, одним из которых €вл€етс€ кислород в степени окислени€ -2. ѕо химическим свойствам оксиды дел€тс€ на основные, кислотные и амфотерные (табл. 1.1).
“аблица 1.1
’имические свойства оксидов
| ¬заимодействие оксидов | ќксиды | ||
| ќсновные Na2O, CaO, MgO, CuO, Fe2O3, ¬аќ | ислотные SO2, SO3, P2O5, CO2, Cl2O, Mn2O7, CrO3 | јмфотерные ¬еќ, ZnO, PbO, MnO2, SnO, Al2O3, Cr2O3 | |
| — водой | –еагируют только оксиды щелочных и щелочноземельных металлов, образу€ щелочи: Na2O + H2O = 2NaOH | ќбразуют кислоты: SO3 + H2O = H2SO4 | Ќе взаимодействуют |
| — кислотами или основани€ми | ¬заимодействуют с кислотами с образованием соли и воды: CаO + 2HCl = = CаCl2 + H2O | ¬заимодействуют с основани€ми с образованием соли и воды: CO2 + Ba(OH)2 = = BaCO3 + H2O | ¬заимодействуют с кислотамикак основные оксиды: BeO + 2HNO3 = Be(NO3)2 + H2O
и с основани€ми
как кислотные оксиды: BeO + 2KOH  K2BeO2 + H2O;
BeO + 2KOH + H2O = K2[Be(OЌ)4] K2BeO2 + H2O;
BeO + 2KOH + H2O = K2[Be(OЌ)4]
|
| ћежду собой | ѕри взаимодействии основного и кислотного оксидов образуетс€ соль: Na2O + SO3 = Na2SO4 |
ќдним из способов получени€ оксидов €вл€етс€ взаимодействие простых веществ с кислородом: 2—а + O2 = 2—аO; — + O2 = —ќ2.
|
|
|
ислоты Ц сложные вещества, состо€щие из атомов водорода, способных замещатьс€ на металл, и кислотного остатка (HNO3, HCl, H2SO4, Ќ3–ќ4).
ислоты взаимодействуют
1. — основани€ми с образованием соли и воды:
2HNO3 + Ca(OH)2 = Ca(NO3)2 + 2H2O.
2. — основными и амфотерными оксидами с образованием соли и воды:
2HCl + ¬аO = ¬аCl2 + H2O;
3H2SO4 + Al2O3 = Al2(SO4)3 + 3H2O.
3. — сол€ми с образованием новой соли и новой кислоты:
H2SO4 + Ba(NO3)2 = BaSO4↓ + 2HNO3.
ќдним из способов получени€ кислот €вл€етс€ взаимодействие кислотного оксида с водой:
P2O5 + 3H2O = 2H3PO4.
ќсновани€ Ц сложные вещества, состо€щие из атомов металла, св€занных с одной или несколькими гидроксогруппами (NaOH, Cu(OH)2, Fe(OH)3).
ќсновани€ взаимодействуют
1. — кислотами с образованием соли и воды:
2NaOH + H2SO4 = Na2SO4 + 2H2O.
2. — кислотными и амфотерными оксидами с образованием соли и воды:
2KOH + N2O5 = 2KNO3 + H2O;
2NaOH + Al2O3 + 3Ќ2ќ = 2Na[Al(OH)4];
2NaOH + Al2O3  2NaAlO2 + H2O.
2NaAlO2 + H2O.
3. — сол€ми с образованием новой соли и нового основани€:
2NaOH + MgCl2 = Mg(OH)2↓ + 2NaCl.
–астворимые в воде основани€ (щелочи) получают взаимодействием активных металлов или их оксидов с водой:
2Na + 2H2O = 2NaOH + H2↑;
BaO + H2O = Ba(OH)2.
Ќерастворимые в воде основани€ получают реакцией обмена:
Fe2(SO4)3 + 6KOH = 2Fe(OH)3↓ + 3K2SO4.
—оли Ц это продукты полного или частичного замещени€ атомов водорода в молекуле кислоты атомами металла или продукты полного или частичного замещени€ гидроксогрупп в молекуле основани€ кислотными остатками.
—редние соли ( 2SO4, Na3PO4) Ц это продукты полного замещени€ водорода в кислоте на металл или гидроксогрупп в основании на кислотные остататки: H2SO4 + 2KOH = K2SO4 + 2H2O;
Mg(ќЌ)2 + 2HCl = MgCl2 + 2Ќ2ќ.
ислые соли (—a(HCO3)2, Nа2Ќ–ќ4) Ц это продукты неполного замещени€ водорода в кислоте на металл:
KOH + H2SO4 = KHSO4 + H2O.
ислые соли образуют только многоосновные кислоты, например H2SO4, Ќ3–ќ4, Ќ2—ќ3, H2S.
ќсновные соли (—uOHNO3, AlOHCl2) Ц это продукты неполного замещени€ гидроксогрупп в основании на кислотные остатки:
Fe(ќЌ)3 + HNO3 = Fe(OH)2NO3 + 2Ќ2ќ;
Fe(ќЌ)3 + 2HNO3 = FeOH(NO3)2 + 2Ќ2ќ.
ќсновные соли образуют только многокислотные основани€, например —u(OH)2, Fe(OH)3, Mg(ќЌ)2.
ѕримеры решени€ задач
ѕример 1.1. —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Na → NaOH → NaHS → Na2S → Na2SO4 → NaCl.
–ешение. NaOH (гидроксид натри€) Ц основание (щелочь). ўелочи можно получить взаимодействием активного металла (в данном примере натри€) с водой:
2Na + 2H2O = 2NaOH + H2↑.
NaHS (гидросульфид натри€) Ц кисла€ соль. ислые соли получаютс€ при взаимодействии многоосновных кислот с основани€ми в тех случа€х, когда количество вз€того основани€ недостаточно дл€ образовани€ средней соли:
H2S + NaOH = NaHS.
Na2S (сульфид натри€) Ц средн€€ соль. ќбразуетс€ при действии избытка щелочи на кислую соль:
NaHS + NaќЌ = Na2S + H2O.
Na2SO4 (сульфат натри€), NaCl (хлорид натри€) Ц средние соли. —редние соли можно получить взаимодействием кислоты и соли:
H2SO4 + Na2S = Na2SO4 + H2S↑,
взаимодействием двух солей:
Na2SO4 + —аCl2 = 2NaCl + —aSO4↓.
ѕример 1.2. — какими из указанных ниже веществ будет взаимодействовать H2SO4: CO2; NaOH; BaCl2; HCl; Fe2O3. Ќаписать уравнени€ соответствующих реакций.
–ешение. ќпредел€ем, к каким классам относ€тс€ указанные соединени€: CO2 Ц кислотный оксид, NaOH Ц основание (щелочь), BaCl2 Ц соль, HCl − кислота, Fe2O3 Ц основной оксид. —ерна€ кислота будет взаимодействовать с основанием, основным оксидом и солью:
H2SO4 + 2NaOH = Na2SO4 + 2H2O;
3H2SO4 + Fe2O3 = Fe2(SO4)3 + 3H2O;
H2SO4 + BaCl2 = BaSO4↓ + 2HCl.
«адачи и упражнени€ дл€ самосто€тельного решени€
1.1. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
|
|
|
.
б). акие из приведенных веществ будут взаимодействовать между собой: Ca(OH)2 и NaOH; Pb(OH)2 и KOH; H2SO4 и H2SO3; HCl и Na2S; HNO3 и MgO? Ќаписать уравнени€ соответствующих реакций.
1.2. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
FeCl2 → Fe(OH)2 → Fe(OH)3 → Fe2O3 → Fe2(SO4)3.
б). акие из приведенных оксидов будут реагировать с HCl: N2O5; SO3; Al2O3; Cl2O7; ZnO; K2O? Ќаписать уравнени€ соответствующих реакций.
1.3. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
P → P2O5 → H3PO4 → Na3PO4 → Ca3(PO4)2.
б). «акончить уравнени€ реакций, доказывающих амфотерность оксида
свинца (II): основные свойства PbO + HNO3 → Е;
кислотные свойства PbO + KOH  Е.
Е.
1.4. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
N2 → NH3 → (NH4)2SO4 → NH4Cl → NH3 → NH4NO3.
б). акие из приведенных оксидов реагируют с NaOH: MgO; Cl2O; Na2O; CrO3; CaO; CO2? —оставить уравнени€ соответствующих реакций.
1.5. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Si → SiO2 → K2SiO3 → H2SiO3 → SiO2.
б). акие из указанных ниже веществ могут взаимодействовать с раствором KOH: HI; CuCl2; SO2; Ba(OH)2; SnO? Ќаписать уравнени€ соответствующих реакций.
1.6. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
CaSO3 ← SO2 ← S → FeS → H2S → KHS.
б). —оставить уравнени€ реакций между кислотами и основани€ми, привод€щих к образованию солей: Na2S; Fe2(SO4)3; K 3PO4.
1.7. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Ca → Ca(OH)2 → CaCO3 → CaCl2 → Ca3(PO4)2.
б). —оставить уравнени€ реакций между кислотами и основани€ми, привод€щих к образованию солей: NaNO3; CaH–O4; CuOHCl.
1.8. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Cu → CuO → Cu(NO3)2 → Cu(OH)2 → CuCl2.
б). ћежду какими из приведенных пар веществ возможна реакци€: CO2 и SO2; LiOH и CO2; P2O5 и CaO; NaOH и KOH; Li2O и ZnO; Li2O и Na2O? —оставить уравнени€ соответствующих реакций.
1.9. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Cd → CdO → Cd(NO3)2 → Cd(OH)2 → CdSO4.
б). — какими из указанных ниже веществ может взаимодействовать серна€ кислота: HCl; BaCl2; MgO; CO2; NaOH; ZnO? —оставить уравнени€ соответствующих реакций.
1.10. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Zn → ZnCl2 → Zn(OH)2 → ZnO → K2ZnO2.
б). Ќаписать уравнени€ реакций образовани€ солей: Na2SO3; Fe2(SO4)3; Ba(NO3)2 в результате взаимодействи€ основани€ и кислотного оксида.
1.11. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
S → SO2 → SO3 → H2SO4 → KHSO4 → K2SO4.
б). —оставить уравнени€ реакций образовани€ солей: CaCO3; Al2(SO4)3; Na3PO4 в результате взаимодействи€ основного и кислотного оксидов.
1.12. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Al → Al2(SO4)3 → Al(OH)3 → Al2O3 → KAlO2.
б). «акончить уравнени€ реакций, доказывающих амфотерность оксида олова (II): основные свойства SnO + HCl → Е;
кислотные свойства SnO + KOH  Е.
Е.
1.13. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
|
|
|
Ba → BaO → Ba(OH)2 → Ba(NO3)2 → BaCO3 → BaCl2.
б). акие из приведенных оксидов взаимодействуют с ќЌ: Na2O; CO2; Ga2O3; MgO; CuO; Mn2O7? Ќаписать уравнени€ соответствующих реакций.
1.14. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Fe(NO3)3 → Fe(OH)3 → Fe2O3 → FeO → FeCl2 → FeS.
б). акие вещества могут быть получены при взаимодействии кислоты с солью? ислоты с основанием? —оли с солью? ѕривести примеры соответствующих реакций.
1.15. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Mg → MgSO4 → Mg(OH)2 → MgOHNO3 → Mg(NO3)2.
б). —оставить уравнени€ реакций, при помощи которых, исход€ из четырех простых веществ Ц кали€, серы, водорода и кислорода, можно получить гидроксид кали€ ќЌ; сульфид кали€ K2S; сероводородH2S.
1.16. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
ZnSO4 ← ZnO ← ZnS → ZnCl2 → Zn(OH)2 → Na2ZnO2.
б). Ќаписать уравнени€ не менее четырех реакций, при помощи которых можно получить карбонат кальци€ CaCO3.
1.17. а). —оставить уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
CuOHCl ← Cu(OH)2 ← CuSO4 ← Cu → CuO → CuCl2.
б). Ќаписать уравнени€ реакций образовани€ K2CrO4, Mg(NO3)2, BaSO4, Ca(ClO)2 в результате взаимодействи€ основани€ и кислотного оксида.
1.18. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Fe → FeSO4 → Fe(OH)2 → Fe(OH)3 → Fe2O3 → FeCl3.
б). ћогут ли находитьс€ совместно в растворе: Ba(OH)2 и FeCl3; HCl и H2S; NaOH и ЌBr; NaOH и KOH; HCl и Na2CO3? ƒать обоснованный ответ и привести уравнени€ соответствующих реакций.
1.19. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Al → Al2O3 → AlCl3 → Al(OH)3 → NaAlO2.
б). ак, использу€ BaO, FeCl3, H2SO4, H2O, CuO, можно получить: гидроксид бари€; гидроксид железа (III); сульфат меди (II)? —оставить уравнени€ соответствующих реакций.
1.20. а). Ќаписать уравнени€ реакций, при помощи которых можно осуществить следующие превращени€:
Pb → PbS → PbO → Pb(NO3)2 → Pb(OH)2 → K2PbO2.
б). —оставить уравнени€ четырех реакций, в результате которых образуетс€ бромид натри€ NaBr.
Ёквивалент
Ёквивалент (Ё) Ц это реальна€ или условна€ частица вещества, соответствующа€ одному иону водорода в кислотно-основных или ионообменных реакци€х, или одному электрону в окислительно-восстановительных реакци€х. ѕод реальной частицей понимают реально существующие соединени€ (NaOH, H2SO4, H2O), под условной Ц доли этих реальных частиц (½ H2SO4, ½ H2O). Ёквивалент Ц безразмерна€ величина, состав которой выражают с помощью знаков и формул. Ќапример,
Ё (NaOH) = NaOH; Ё (H2SO4) = ½ H2SO4; Ё (MgCl2 ) = ½ MgCl2; Ё (—а) = ½ —а.
≈диницей количества вещества эквивалентов €вл€етс€ моль. ћоль эквивалентов Ц это количество вещества, содержащего 6,02×1023 эквивалентов. ћасса одного мол€ эквивалентов называетс€ мол€рной массойэквивалентов (M эк)и выражаетс€в г/моль.
ѕри определении мол€рной массы эквивалентов необходимо исходить из конкретной реакции, в которой участвует данное вещество.
ћол€рна€ масса эквивалентов вещества ¬, участвующего в окислительно-восстановительной реакции, рассчитываетс€ по формуле
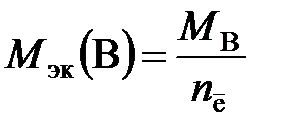 ,
,
где ћ ¬ Ц мол€рна€ масса вещества ¬; n ē Ц число электронов, присоединенных одной молекулой окислител€ или отданных одной молекулой восстановител€.
Ќапример, в реакции Mg0 + 2H+Cl = Mg+2Cl2 + H20 степень окислени€ магни€ измен€етс€ от 0 до +2. —ледовательно, магний тер€ет 2 электрона, т.е. одному электрону эквивалентна условна€ частица ½ атома Mg:
Ё (Mg) = ½ Mg; ћ эк (Mg) = 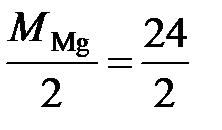 = 12 г/моль.
= 12 г/моль.
” водорода степень окислени€ мен€етс€ от +1 до 0, а т.к. молекула водорода состоит из двух атомов, то число прин€тых электронов будет равно 2. “аким образом, одному электрону эквивалентна условна€ частица ½ молекулы Ќ2:
Ё (Ќ2) = ½ Ќ2 ; ћ эк (Ќ2) =  = 1 г/моль.
= 1 г/моль.
«акон эквивалентов: массы реагирующих друг с другом веществ пропорциональны их мол€рным массам эквивалентов:
 .
.
ѕримеры решени€ задач
ѕример 2.1. –ассчитать эквивалент и мол€рную массу эквивалентов H2S и NaOH в реакци€х:
H2S + 2NaOH = Na2S + 2H2O; (1)
H2S + NaOH = NaHS + H2O. (2)
–ешение. ћол€рна€ масса эквивалентов кислоты или основани€, участвующихв кислотно-основной реакции, рассчитываетс€ по формуле
|
|
|
ћ эк (кислоты, основани€) = 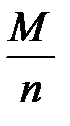 ,
,
где ћ Ц мол€рна€ масса кислоты или основани€; n Ц дл€ кислот Ц число атомов водорода, замещенных в данной реакции на металл; дл€ оснований Ц число гидроксильных групп, замещенных в данной реакции на кислотный остаток.
«начение эквивалента и мол€рной массы эквивалентов вещества зависит от реакции, в которой это вещество участвует.
¬ реакции H2S + 2NaOH = Na2S + 2H2O (1) оба иона водорода молекулы H2S замещаютс€ на металл и, таким образом, одному иону водорода эквивалентна условна€ частица ½ H2S. ¬ этом случае
Ё (H2S) = ½ H2S, а ћ эк (H2S)= 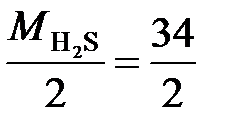 = 17 г/моль.
= 17 г/моль.
¬ реакции H2S + NaOH = NaHS + H2O (2) в молекуле H2S на металл замещаетс€ только один ион водорода и, следовательно, одному иону 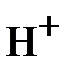 эквивалентна реальна€ частица Ц молекула H2S. ¬ этом случае
эквивалентна реальна€ частица Ц молекула H2S. ¬ этом случае
Ё (H2S) = H2S, а ћ эк (H2S) =  =
=  34 г/моль.
34 г/моль.
Ёквивалент NaOH в реакци€х (1) и (2) равен NaOH, так как в обоих случа€х на кислотный остаток замещаетс€ одна гидроксильна€ группа. ћол€рна€ масса эквивалентов NaOH равна
ћ эк (NaOH) = 40 г/моль.
“аким образом, эквивалент H2S в реакции (1) равен ½ H2S, в реакции (2) − 1 H2S, мол€рные массы эквивалентов H2S равны соответственно 17 (1) и 34 (2) г/моль; эквивалент NaOH в реакци€х (1) и (2) равен NaOH, мол€рна€ масса эквивалентов основани€ составл€ет 40 г/моль.
ѕример 2.2. –ассчитать эквивалент и мол€рную массу эквивалентов оксидов P2O5 и CaO в реакции P2O5 + 3CaO = Ca3(PO4)2.
–ешение. ћол€рна€ масса эквивалентов оксида рассчитываетс€ по формуле
ћ эк (оксида) =  ,
,
где ћ Ц мол€рна€ масса оксида; n Ц число катионов соответствующего оксиду основани€ или число анионов соответствующей оксиду кислоты; |c.o.| Ц абсолютное значение степени окислени€ катиона или аниона.
¬ реакции P2O5 + 3CaO = Ca3(PO4)2 эквивалент P2O5, образующего два трехзар€дных аниона (–ќ4)3-, равен 1/6 P2O5, а ћ эк (P2O5) = 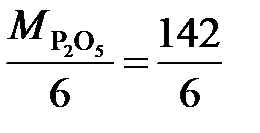 = 23,7 г/моль. Ёквивалент —аќ, дающего один двухзар€дный катион (—а2+), равен ½ —аќ, а ћ эк (—аќ)=
= 23,7 г/моль. Ёквивалент —аќ, дающего один двухзар€дный катион (—а2+), равен ½ —аќ, а ћ эк (—аќ)=  =
= 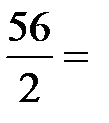 28 г/моль.
28 г/моль.
ѕример 2.3. ¬ычислить эквивалент и мол€рную массу эквивалентов фосфора в соединени€х –Ќ3, –2ќ3 и –2ќ5.
–ешение. „тобы определить мол€рную массу эквивалентов элемента в соединении, можно воспользоватьс€ следующей формулой:
ћ эк (элемента) = 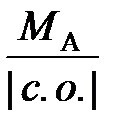 ,
,
где ћј Ц мол€рна€ масса элемента; |c.o.| Ц абсолютное значение степени окислени€ элемента.
—тепень окислени€ фосфора в –Ќ3, –2ќ3, –2ќ5 соответственно равна Ц3, +3 и +5. ѕодставл€€ эти значени€ в формулу, находим, что мол€рна€ масса эквивалентов фосфора в соединени€х –Ќ3 и –2ќ3 равна 31/3 = 10,3 г/моль; в –2ќ5 Ц 31/5 = 6,2 г/моль, а эквивалент фосфора в соединени€х –Ќ3 и –2ќ3 равен 1/3 –, в соединении –2ќ5 Ц 1/5 –.
ѕример 2.4. –ассчитать мол€рную массу эквивалентов соединений фосфора –Ќ3, –2ќ3 и –2ќ5.
–ешение. ћол€рна€ масса эквивалентов химического соединени€ равна сумме мол€рных масс эквивалентов составл€ющих его частей:
ћ эк (–Ќ3) = ћ эк (–) + ћ эк (Ќ) = 10,3 + 1 = 11 г/моль;
ћ эк (–2ќ3) = ћ эк (–) + ћ эк (ќ) = 10,3 + 8 = 18,3 г/моль;
ћ эк (–2ќ5) = ћ эк (–) + ћ эк (ќ) = 6,2 + 8 = 14,2 г/моль.
ѕример 2.5. Ќа восстановление 7,09 г оксида металла со степенью окислени€ +2 требуетс€ 2,24 л водорода при нормальных услови€х. ¬ычислить мол€рные массы эквивалентов оксида и металла. „ему равна мол€рна€ масса металла?
–ешение. «адача решаетс€ по закону эквивалентов. “ак как одно из реагирующих веществ находитс€ в газообразном состо€нии, то удобно воспользоватьс€ следующей формулой:
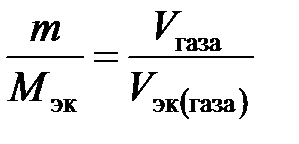 ,
,
где V эк (газа) Ц объем одного мол€ эквивалентов газа. ƒл€ вычислени€ объема мол€ эквивалентов газа необходимо знать число молей эквивалентов (υ) в одном моле газа: υ =  . “ак, ћ (Ќ2) = 2 г/моль; ћ эк (Ќ2) = 1 г/моль. —ледовательно, в одном моле молекул водорода Ќ2 содержитс€ υ = 2/1 = 2 моль эквивалентов водорода. ак известно, моль любого газа при нормальных услови€х (н.у.) (“ = 273 , – = 101,325 кѕа) занимает объем 22,4 л. «начит, моль водорода займет объем 22,4 л, а так как в одном моле водорода содержитс€ 2 моль эквивалентов водорода, то объем одного мол€ эквивалентов водорода равен V эк (Ќ2) = 22,4/2 = 11,2 л. јналогично ћ (ќ2) = 32 г/моль, ћ эк (ќ2) = 8 г/моль. ¬ одном моле молекул кислорода ќ2 содержитс€ υ = 32/8 = 4 моль эквивалентов кислорода. ќдин моль эквивалентов кислорода при нормальных услови€х занимает объем V эк (ќ2) = 22,4/4 = 5,6 л.
. “ак, ћ (Ќ2) = 2 г/моль; ћ эк (Ќ2) = 1 г/моль. —ледовательно, в одном моле молекул водорода Ќ2 содержитс€ υ = 2/1 = 2 моль эквивалентов водорода. ак известно, моль любого газа при нормальных услови€х (н.у.) (“ = 273 , – = 101,325 кѕа) занимает объем 22,4 л. «начит, моль водорода займет объем 22,4 л, а так как в одном моле водорода содержитс€ 2 моль эквивалентов водорода, то объем одного мол€ эквивалентов водорода равен V эк (Ќ2) = 22,4/2 = 11,2 л. јналогично ћ (ќ2) = 32 г/моль, ћ эк (ќ2) = 8 г/моль. ¬ одном моле молекул кислорода ќ2 содержитс€ υ = 32/8 = 4 моль эквивалентов кислорода. ќдин моль эквивалентов кислорода при нормальных услови€х занимает объем V эк (ќ2) = 22,4/4 = 5,6 л.
ѕодставив в формулу  численные значени€, находим, что ћ эк (оксида) =
численные значени€, находим, что ћ эк (оксида) =  г/моль.
г/моль.
ћол€рна€ масса эквивалентов химического соединени€ равна сумме мол€рных масс эквивалентов составл€ющих его частей. ќксид Ц это соединение металла с кислородом, поэтому мол€рна€ масса эквивалентов оксида представл€ет собой сумму ћ эк (оксида) = ћ эк (металла) + ћ эк (кислорода). ќтсюда ћ эк (металла) = ћ эк (оксида) − ћ эк (кислорода) = 35,45 Ц 8 = 27,45 г/моль.
ћол€рна€ масса эквивалентов элемента (ћ эк) св€зана с атомной массой элемента (ћ ј) соотношением: ћ эк (элемента) = 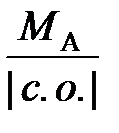 , где ½ с.о. ½ − степень окислени€ элемента. ќтсюда ћ ј = ћ эк (металла) ∙ ½ с.о. ½ = 27,45×2 = 54,9 г/моль.
, где ½ с.о. ½ − степень окислени€ элемента. ќтсюда ћ ј = ћ эк (металла) ∙ ½ с.о. ½ = 27,45×2 = 54,9 г/моль.
“аким образом, ћ эк (оксида) = 35,45 г/моль; ћ эк (металла) = 27,45 г/моль; ћ ј (металла) = 54,9 г/моль.
ѕример 2.6. ѕри взаимодействии кислорода с азотом получено 4 моль эквивалентов оксида азота (IV). –ассчитать объемы газов, вступивших в реакцию при нормальных услови€х.
–ешение. ѕо закону эквивалентов число молей эквивалентов веществ, вступающих в реакцию и образующихс€ в результате реакции, равны между собой, т.е. υ (ќ2) = υ (N2) = υ (NO2). “ак как получено 4 моль эквивалентов оксида азота (IV), то, следовательно, в реакцию вступило 4 моль эквивалентов ќ2 и 4 моль эквивалентов N2.
јзот измен€ет степень окислени€ от 0 (в N2) до +4 (в Nќ2), и так как в его молекуле 2 атома, то вместе они отдают 8 электронов, поэтому
ћ эк (N2) =  = 3,5 г/моль. Ќаходим объем, занимаемый молем эквивалентов азота (IV): 28 г/моль N2 Ц 22,4 л
= 3,5 г/моль. Ќаходим объем, занимаемый молем эквивалентов азота (IV): 28 г/моль N2 Ц 22,4 л
3,5 г/моль N2 Ц х
х =  л.
л.
“ак как в реакцию вступило 4 моль эквивалентов N2, то их объем составл€ет V (N2) = 2,8Ј4 = 11,2 л. «на€, что моль эквивалентов кислорода при нормальных услови€х занимает объем 5,6 л, рассчитываем объем 4 моль эквивалентов ќ2, вступивших в реакцию: V (O2) = 5,6∙4 = 22,4 л.
»так, в реакцию вступило 11,2 л азота и 22,4 л кислорода.
ѕример 2.7. ќпределить мол€рную массу эквивалентов металла, если из 48,15 г его оксида получено 88,65 г его нитрата.
–ешение. ”читыва€, что ћ эк (оксида) = ћ эк (металла) + ћ эк (кислорода), а ћ эк (соли) = ћ эк (металла) + ћ эк (кислотного остатка), подставл€ем соответствующие данные в закон эквивалентов:
 ;
;
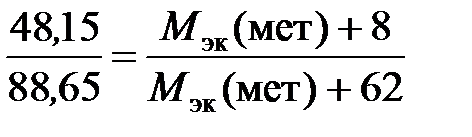 ,
,
отсюда ћ эк (металла) = 56,2 г/моль.
ѕример 2.8. ¬ычислить степень окислени€ хрома в оксиде, содержащем 68,42 % (масс.) этого металла.
–ешение. ѕрин€в массу оксида за 100 %, находим массовую долю кислорода в оксиде: 100 Ц 68,42 = 31,58 %, т.е. на 68,42 частей массы хрома приходитс€ 31,58 частей массы кислорода, или на 68,42 г хрома приходитс€ 31,58 г кислорода. «на€, что мол€рна€ масса эквивалентов кислорода равна 8 г/моль, определим мол€рную массу эквивалентов хрома в оксиде по закону эквивалентов:
 ; ћ эк (Cr) =
; ћ эк (Cr) =  г/моль.
г/моль.
—тепень окислени€ хрома находим из соотношени€  ,
,
отсюда | c. o. | =  = 3.
= 3.






