285. ’лорид гидроксомагни€ + сол€на€ кислота.
«адание IV. Cоставить молекул€рные уравнени€ реакций, которым соответствуют ионно-молекул€рные уравнени€.
| 286. H+ + OH- Ѓ H2O. | 291. SO32- + 2 H+ Ѓ SO2 + H2O. |
| 287. Pb2+ + S2- Ѓ PbS¯. | 292. Pb2+ + CrO42- Ѓ PbCrO4¯. |
| 288. ClO- + H+ Ѓ HClO. | 293. HCO3- + OH- Ѓ CO32- + H2O. |
| 289. CO32- + 2H+ Ѓ CO2 + H2O. | 294. ZnOH+ + H+ Ѓ Zn2+ + H2O. |
| 290. CH3COOH + OH- Ѓ CH3COO- + H2O. | 295. Mg2+ + CO32- Ѓ MgCO3¯. |
| 296. Cu2+ + S2- Ѓ CuS¯. | 306. HCO3- + H+ Ѓ H2O + CO2. |
| 297. SiO32- + 2 H+ Ѓ H2SiO3. | 307. Cu2+ + 2OH- Ѓ Cu(OH)2¯. |
| 298. CaCO3 + 2H+ Ѓ Ca2+ + H2O. | 308. Pb2+ + 2 I- Ѓ PbI2¯. |
| 299. Al(OH)3 + OH- Ѓ [Al(OH)4]-. | 309. Sr2+ + SO42- Ѓ SrSO4¯. |
| 300. Pb2+ + 2 I- Ѓ PbI2¯. | 310. Sr2+ + CO32- Ѓ SrCO3¯. |
| 301. Fe(OH)3 + 3H+ Ѓ Fe3+ + 3H2O. | 311. 2Ag+ + SO42- Ѓ Ag2SO4¯. |
| 302. Cd2+ + 2OH- Ѓ Cd(OH)2¯. | 312. Ba2+ + CO32- Ѓ BaCO3¯. |
| 303. H+ + NO2- Ѓ HNO2. | 313. Cd2+ + 2 OH- Ѓ Cd(OH)2¯. |
| 304. Zn2+ + H2S Ѓ ZnS¯ + 2 H+. | 314. CuOH+ + H+Ѓ Cu2+ + H2O. |
| 305. Ag+ + Cl- Ѓ AgCl¯. | 315. NH4+ + OH- Ѓ NH4OH. |
«адание V. «акончить и уравн€ть реакцию.
| 316. AgNO3 + H2S Ѓ Е | 340. CdCl2 + H2S Ѓ Е |
| 317. AgNO3 + K2Cr2O7 Ѓ Е | 341. CH3COOAg + H2S Ѓ Е |
| 318. AgNO3 + K2CrO4 Ѓ Е | 342. CH3COOH + Ba(OH)2 Ѓ... |
| 319. Al(OH)3 + H2SO4 Ѓ Е | 343. CH3COOH + NH4OH Ѓ |
| 320. Al2(SO4)3 + AgNO3 Ѓ Е | 344. CH3COOK + H2SO4 Ѓ Е |
| 321. AlBr3 + AgNO3 Ѓ Е | 345. HCOOK + H2SO4 Ѓ Е |
| 322. AlI3 + AgNO3 Ѓ Е | 346. CH3COOPb + Na2CrO4 Ѓ Е |
| 323. Ba(NO3)2 + Cr2(SO4)3 Ѓ Е | 347. CH3COOPb + Na2SO4 Ѓ Е |
| 324. Ba(NO3)2 + K2CrO4 Ѓ Е | 348. Cr2(SO4)3 + NaOH Ѓ Е |
| 325. Ba(NO3)2 + Na2SO4 Ѓ Е | 349. Cr2(SO4)3 + NH4OH Ѓ Е |
| 326. Ba(OH)2 + CO2 Ѓ Е | 350. Cu(NO3)2 + Ba(OH)2 Ѓ Е |
| 327. BaCl2 + Al2(SO4)3 Ѓ... | 351. Cu(NO3)2 + H2S Ѓ Е |
| 328. BaCl2 + Na3PO4 Ѓ Е | 352. Cu(NO3)2 + Na2S Ѓ Е |
| 329. BaCl2 + CH3COOAg Ѓ... | 353. CuSO4 + NaOH Ѓ Е |
| 330. Bi(OH)(NO3)2 + HNO3 Ѓ Е | 354. CuSO4 + NH4OH Ѓ Е |
| 331. Ca(NO3)2 + H2C2O4 Ѓ Е | 355. CuSO4 + Ba(OH)2 Ѓ Е |
| 332. Ca(NO3)2 + Na2SO4 Ѓ Е | 356. Fe(OH)3 + H2SO4 Ѓ Е |
| 333. Ca(NO3)2 + Na3PO4 Ѓ Е | 357. Fe2(SO4)3 + NaOH Ѓ Е |
| 334. Ca(OH)2 + CO2 Ѓ Е | 358. FeCl3 + KOH Ѓ Е |
| 335. CaCl2 + H2SO4 Ѓ Е | 359. FeOHCl2 + HCl Ѓ... |
| 336. CaCO3 + CH3COOH Ѓ Е | 360. FeS + HCl Ѓ Е |
| 337. CaCO3 + HCl Ѓ Е | 361. H2S + NH4OH Ѓ... |
| 338. CaCO3 + CO2 + H2O Ѓ... | 362. HCOOK + HNO3 Ѓ... |
| 339. Cd(NO3)2 + Na2S Ѓ Е | 363. HF + KOH Ѓ Е |
| 364. Hg(NO3)2 + H2S Ѓ... | 385. Na2HPO4 + NaOH Ѓ Е |
| 365. HNO2 + NH4OH Ѓ Е | 386. NaHSO4 + NaOH Ѓ Е |
| 366. K2CO3 + H3PO4 Ѓ Е | 387. NaHSO4 + Ba(NO3)2 Ѓ... |
| 367. K2CO3 + HCl Ѓ Е | 388. NaOH + H2SO3 Ѓ Е |
| 368. KHSO3 + H2SO4 Ѓ... | 389. NaOH + H2SO4 Ѓ Е |
| 369. KOH + HCN Ѓ Е | 390. NH4Cl + Ca(OH)2 Ѓ... |
| 370. KOH + H3PO4 Ѓ Е | 391. NH4Cl + NaOH Ѓ Е |
| 371. Mg(NO3)2 + (NH4)2C2O4 Ѓ... | 392. NH4Cl + Sr(OH)2 Ѓ Е |
| 372. MgCO3 + HCl Ѓ Е | 393. NH4Cl + Ba(OH)2 Ѓ Е |
| 373. MgOHCl + HCl Ѓ. | 394. Ni(NO3)2 + KOH Ѓ Е |
| 374. MnCl2 + NH4OH Ѓ Е | 395. NiSO4 + (NH4)2S Ѓ Е |
| 375. Na2S + H2SO4 Ѓ Е | 396. Pb(NO3)2 + Fe2(SO4)3 Ѓ Е |
| 376. Na2S + NiSO4 Ѓ Е | 397. Pb(NO3)2 + K2CrO4 Ѓ Е |
| 377. Na2S + CdSO4 Ѓ Е | 398. Pb(NO3)2 + KI Ѓ Е |
| 378. Na2SiO3 + H2SO4 Ѓ Е | 399. Pb(NO3)2 + Na2S Ѓ... |
| 379. Na2SO4 + BaCl2 Ѓ Е | 400. Pb(NO3)2 + NaCl Ѓ Е |
| 380. Na2SO4 + H2SO4 Ѓ Е | 401. Sr(NO3)2 + H2SO4 Ѓ... |
| 381. Na2SO4 + Pb(NO3)2 Ѓ... | 402. Zn(NO3)2 + KOH Ѓ Е |
| 382. Na2SO4 + BaCl2 Ѓ Е | 403. Zn(OH)2 + H2SO4 Ѓ Е |
| 383. NaHCO3 + HCl Ѓ Е | 404. Zn(OH)2 + NaOH(избыток) Ѓ... |
| 384. NaHCO3 + NaOH Ѓ... | 405. Zn(OH)2 + NaOH Ѓ Е |
|
|
|
2. —троение атома
јтом любого элемента состоит из положительно зар€женного €дра и отрицательно зар€женных электронов, в целом атом Ц система электронейтральна€. «ар€д €дра равен пор€дковому номеру элемента в таблице ƒ.».ћенделеева. —осто€ние электрона в атоме описываетс€ при помощи набора четырех квантовых чисел: главного n, орбитального l, магнитного ml и спинового ms. ќпределенные значени€ трех квантовых чисел (n, l,ml) описывают состо€ние электрона, называемое атомной орбиталью (јќ).
√лавное квантовое число n определ€ет энергию јќ и номер энергетического уровн€, на котором находитс€ электрон, и может принимать целочисленные значени€ от единицы до бесконечности.
ќрбитальное квантовое число l определ€ет форму јќ и энергетический подуровень, оно может принимать значени€ от нул€ до n -1. »сторически атомным орбитал€м со значени€ми l, равным 0, 1, 2, 3 присвоены буквенные обозначени€ s -, p -, d -, f -. ¬ графических схемах электронного строени€ атомов кажда€ орбиталь обозначаетс€ символом.
 ћагнитное квантовое число ml определ€ет пространственную ориентацию јќ и отчасти ее форму, оно может принимать значени€ от Ц l до + l.
ћагнитное квантовое число ml определ€ет пространственную ориентацию јќ и отчасти ее форму, оно может принимать значени€ от Ц l до + l.
—пиновое квантовое число m s характеризует собственный момент импульса и св€занный с ним магнитный момент и может принимать значени€ ±1/2.
ѕоследовательность распределени€ электронов в атоме по мере увеличени€ значений l и n выражаетс€ электронными или электронно-графическими формулами.
ѕри заполнении јќ действует принцип ѕаули, из которого следует, что в атоме не может быть двух электронов, характеризующихс€ одинаковым набором значений четырех квантовых чисел. —осто€ние электронов в атоме должно отличатьс€ значением хот€ бы одного квантового числа.
«аполнение энергетических подуровней подчин€етс€ правилу ’унда, согласно которому электроны в основном состо€нии в атоме располагаютс€ так, чтобы модуль суммарного спина всех электронов подуровн€ был максимальным. Ќапример, четыре валентных p -электрона атома кислорода размещаютс€ в квантовых €чейках следующим образом:
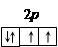

ѕоследовательность заполнени€ энергетических уровней и подуровней в атомах выражаетс€ правилом лечковкого: пор€док заполнени€ определ€етс€ возрастанием суммы n + l, а при одинаковом ее значении первым заполн€етс€ подуровень с меньшим значением n в этой сумме. Ќапример,
1 s Ѓ2 s Ѓ2 p Ѓ3 s Ѓ3 p Ѓ4 s Ѓ3 d Ѓ4 p Ѓ5 s Ѓ4 d Ѓ5 p Ѓ6 s Ѓ5 d 1(La)Ѓ4 f
(лантаноиды)Ѓ5 d Ѓ6 p Ѓ7 s Ѓ6 d 1(Ac)Ѓ5 f (актиноиды)Ѓ6 d ЃЕ
ѕринадлежность элемента к электронному семейству определ€етс€ характером заполнени€ энергетических подуровней: s -элементы Ц заполнение внешнего s -подуровн€ (например, литий 1 s 2 2 s 1), р -элементы Ц заполнение внешнего p -подуровн€ (например, фтор Ц 1 s 22 s 2 2 p 5), d -элементы Ц заполнение предвнешнего d -подуровн€ (например, ванадий Ц 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 3), f -элементы Ц заполнение f -подуровн€ второго снаружи уровн€ (например,  неодим Ц 1 s 2 2 s 2 2 p 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 4).
неодим Ц 1 s 2 2 s 2 2 p 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 4).
|
|
|
ƒл€ d - и f -элементов возможны отклонени€ от описанного способа заполнени€ јќ Ц так называемый провал электрона. Ёто €вление св€зано с тем, что дл€ атома устойчивым состо€нием €вл€етс€ полностью или наполовину заполненна€ јќ, т.е. d 10, d 5, f 14, f 7. ¬ ситуации, когда до достижени€ такого состо€ни€ не хватает одного электрона, он переходит (Ђпроваливаетс€ї) с предыдущего уровн€. Ќапример, электронный паспорт серебра 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 9 с учетом провала электрона примет вид 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 14 d 10.
≈сли на валентных энергетических уровн€х имеютс€ вакантные јќ, то при получении электронами порции энергии (возбуждении атома) становитс€ возможным Ђразъединениеї валентных электронов, т.е. их переходы с тех подуровней, где все јќ зан€ты полностью (¯≠) или частично (≠), на другие валентные подуровни того же уровн€, имеющие незаполненные јќ. ѕри этом с тех јќ, которые в основном (соответствующем минимальной энергии атома) состо€нии были зан€ты полностью, Ђуходитї по одному электрону последовательно, т.е. возможно несколько возбужденных состо€ний. ¬озбуждение мен€ет валентное состо€ние атома (число его неспаренных электронов).
ѕример 1. —оставить электронную формулу атома брома и графическую схему заполнени€ электронами валентных орбиталей в нормальном и возбужденном состо€ни€х.
–ешение. 1. ѕор€дковый номер брома Ц 35, следовательно атом брома имеет 35 электронов. Ѕром находитс€ в IV периоде периодической системы, следовательно, јќ с n, равным 1; 2 и 3 заполнены полностью. Ѕром относитс€ к p -элементам, следовательно, заполнен 4 s -подуровень. ¬ р€ду 4 p -элементов бром Ц п€тый элемент, следовательно, на 4 p -подуровне Ц п€ть электронов. “аким образом, электронна€ формула брома имеет вид 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 5.
2. ¬алентными орбитал€ми в этом атоме €вл€ютс€ орбитали внешнего (четвертого) электронного сло€, т.е. 4 s -, 4 p - и незаполненные 4 d -орбитали. √рафически схема заполнени€ электронами этих орбиталей имеет вид
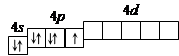 |

в таком состо€нии бром имеет валентность 1, которой соответствуют степени окислени€ -1 и +1.
3. ѕри затрате некоторой энергии спаренный p -электрон перейдет на свободную d -орбиталь. ¬ этом первом возбужденном состо€нии
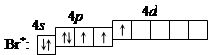 |

бром имеет валентность 3, которой соответствует степень окислени€ +3.
4. ѕри передаче атому брома еще некоторого количества энергии следующий p -электрон также перейдет на свободную d -орбиталь. ¬о втором возбужденном состо€нии
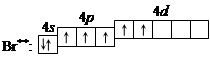 |

бром имеет валентность 5, которой соответствует степень окислени€ +5.
5. ѕри передаче атому брома еще некоторого количества энергии s -электрон также перейдет на свободную d -орбиталь. ¬ третьем возбужденном состо€нии
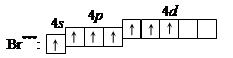 |

бром имеет валентность 7, которой соответствует степень окислени€ +7.
ѕример 2. —оставить электронные формулы атома селена в состо€ни€х Se-2 и Se+4 и графические схемы заполнени€ электронами валентных орбиталей.
–ешение. 1. Cоставим электронную формулу атома селена (см. пример 1): 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 4. √рафическа€ схема заполнени€ электронами валентных орбиталей имеет вид
 |


2. ƒл€ получени€ Se-2 необходимо к атому селена добавить два электрона на 4 p -орбиталь (согласно правилу лечковского). “огда графическа€ схема примет вид
 |

Ёлектронна€ формула Se-2 Ц 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 6.
3. ƒл€ получени€ Se+4 необходимо убрать четыре электрона с 4 p -орбитали, атома селена:
 |

Ёлектронна€ формула Se+4 Ц 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 0.
ѕример 3. —оставить полную электронную формулу элемента, валентные электроны которого имеют конфигурацию 3 d 6, определить, к какому периоду таблицы ƒ.».ћенделеева принадлежит данный элемент.
|
|
|
–ешение. —огласно правилу лечковского 3 d -элементы наход€тс€ в четвертом периоде таблицы ƒ.».ћенделеева. Ќа данной орбитали находитс€ шесть электронов, значит, это шестой по счету среди 3 d -элементов, т.е. железо, полна€ электронна€ формула которого 1 s 22 s 22 p 63 s 23 p 64 s 23 d 6.
«адание I. —оставить электронные формулы элементов, графические схемы заполнени€ электронами валентных орбиталей в спокойном и возбужденном состо€ни€х, указать, к какому типу эти элементы относ€тс€.
| 1. B, Al, Th. 2. Po, Ba, Lu. 3. Mg, Pm, Be. 4. Br, Co, Hf. 5. C, Tm, As. 6. Nd, Ca, V. 7. Ta, O, Ce. 8. Y, Rb, S. | 9. At, Xe, Lr. 10. Cs, U, H. 11. Cl, Cu, Bi. 12. Na, Ac, Fe. 13. Pb, Ra, Dy. 14. Ag, Re, In. 15. Cd, K, Pa. 16. P, Se, Li. | 17. Yb, Mn, Sn. 18. Mo, La, N. 19. Pu, Ni, Sb. 20. Au, Np, Rn. 21. Cr, Tl, Cm. 22. Si, I, Zr. 23. Tb, Sr, Bk. 24. Fr, Ti, W. | 25. He, Hg, Gd. 26. Pt, Ne, Sm. 27. Ga, Ru, Ho. 28. Sc, Pr, Os. 29. Ar, Ir, Eu. 30. Zn, Rh, Er. 31. Kr, Pd, Am. 32. Ge, Cf, F. |
«адание II. —оставить электронные формулы атомов в указанных состо€ни€х и графические схемы заполнени€ электронами валентных орбиталей.
| 33. Li+, C+2. 34. O-2, F-. 35. Na+, N-3. 36. Ca+2, C+4. 37. Al+3, B-3. 38. C-4, Ba+2. 39. S+6, P-3. 40. P+5, Cl-1. | 41. I+5, Fe+3. 42. Be+2, Co+3. 43. Cr+6, Cu+2. 44. I-, P+3. 45. Cr+3, Br+3. 46. Ag+, Sn+4. 47. Zn+2, S-2. 48. K+, Fe+2. | 49. Zr+4, Pb+2. 50. N+5, Br-. 51. Ni+2, Cl+5. 52. Se-2, Cs+. 53. Te-2, Sr+2. 54. Bi+3, Si-4. 55. B+3, Sc+2. 56. Mg+2, Mn+7. | 57. Cd+2, Sn+2. 58. Nb+3, Hg+. 59. Tl+, V+3. 60. Ti+4, Mn+2. 61. Os+3, Au+3. 62. Rb+, Ce+3. 63. Fr+1, Y+3. 64. H+, Re+7. |
«адание III. »сход€ из состо€ни€ валентных электронов, составить электронную формулу элемента в нулевой степени окислени€. ќпределить, к какому периоду таблицы ƒ.».ћенделеева принадлежит данный элемент.
| 65. 4 d 1. 66. 3 d 10. 67. 4 s 13 d 10. 68. 5 d 2. 69. 6 p 2. 70. 6 s 14 f 145 d 10. 71. 4 s 23 d 5. 72. 4 s 13 d 5. | 73. 7 s 26 d 1. 74. 5 d 3. 75. 6s24 f 2. 76. 6 p 1. 77. 5 s 14 d 5. 78. 4 f 3. 79. 6 d 15 f 3. 80. 5 s 24 d 5. | 81. 5 d 8. 82. 5 s 14 d 10. 83. 5 s 24 d 10. 84. 5 s 2. 85. 5 p 3. 86. 6 p 4. 87. 4 d 6. 88. 5 d 6. | 89. 5 f 7. 90. 5 d 6. 91. 5 f 2. 92. 4 d 7. 93. 5 d 14 f 7. 94. 4 f 10. 95. 4 d 8. 96. 5 p 6. |
3. ќсновные пон€ти€ и законы химии
ћоль, мол€рна€ масса. »звестно, что любое вещество состоит из атомов, химические процессы протекают благодар€ взаимодействию атомов. »з практических соображений было введено пон€тие мол€. ”словились считать, что 1 моль вещества содержит 6,02×1023 частиц, любых Ц атомов, молекул, ионов. „исло 6,02×1023 называетс€ числом јвогадро. ћатематически пон€тие мол€ можно записать в виде формулы
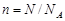 ,
,
где n Ц количество вещества, моль; N Ц число частиц (молекул, атомов, ионов); N A Ц число јвогадро.
ћассу 1 моль вещества называют мол€рной массой M. ћол€рна€ масса в неорганической химии €вл€етс€ характеристикой вещества, непосредственно св€занной с его количественным составом и численно равна молекул€рной массе (массе одной молекулы) вещества, выраженной в углеродных единицах. ћол€рна€ масса любого вещества
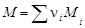 ,
,
где n i Ц стехиометрический индекс в формуле вещества; ћi Ц мол€рна€ масса элемента, вход€щего в соединение, г/моль (см. таблицу элементов ƒ.».ћенделеева).
ћасса и количество вещества св€заны зависимостью
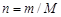 .
.
ћол€рна€ масса вещества может быть определена экспериментально. ƒл€ газов ее наход€т, например, по относительной плотности газа D, котора€ представл€ет собой соотношение мол€рных масс двух газов, одна из которых обычно известна:
|
|
|
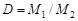 .
.
Ќаиболее часто используют плотность газа по воздуху D возд, тогда ћ 2 = ћ возд D возд (ћ возд = 29 г/моль), или по водороду 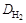 , тогда ћ 2 =
, тогда ћ 2 = 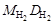 .
.
ќсновные газовые законы. —осто€ние газа характеризуетс€ его температурой, давлением и объемом. ≈сли температура газа 0 ∞— (273,15 K), а давление 1 атм (1,013×105 ѕа = 760 мм рт. ст.), то услови€, при которых находитс€ газ, называют нормальными.
¬заимосв€зь между объемом и количеством вещества газа описываетс€ законом јвогадро: в равных объемах любых газов, вз€тых при одной и той же температуре и одинаковом давлении, содержитс€ одинаковое число молекул. —ледовательно, при одинаковых услови€х 1 моль любого газа занимает один и тот же объем. Ётот объем называетс€ мол€рным объемом газа VM. ѕри нормальных услови€х VM = 22,4 л и количество вещества газа в мол€х может быть вычислено по уравнению
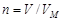 .
.
¬заимосв€зь между количеством вещества, температурой, давлением и объемом газа устанавливает уравнение ћенделеева Ц лапейрона:
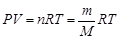 ,
,
где P Ц давление, ѕа; V Ц объем, м3; n Ц количество вещества, моль; m Ц масса, г; ћ Ц мол€рна€ масса газа, г/моль; R Ц универсальна€ газова€ посто€нна€, в системе —» R = 8,314 ƒж/(моль×K).
Ќа практике чаще всего приходитс€ иметь дело со смесью газов. аждый газ вносит свой вклад в общее давление системы Ц парциальное давление. ѕарциальным называетс€ давление, которое производил бы этот газ, занима€ при тех же физических услови€х объем всей газовой смеси. ѕарциальное давление может быть вычислено через объемное содержание газа в газовой смеси или через мольную долю газа. —оответственно
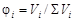 ;
; 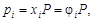
где Vi Ц объем данного газа; S Vi Ц общий объем газовой смеси; ni Ц количество вещества данного газа; S ni Ц сумма числа молей всех компонентов газовой смеси; xi Ц мольна€ дол€ газа, 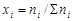 ; P Ц общее давление смеси газов.
; P Ц общее давление смеси газов.






