ќ—Ќќ¬Ќџ≈ Ћј——џ Ќ≈ќ–√јЌ»„≈— »’ —ќ≈ƒ»Ќ≈Ќ»…
—“≈ѕ≈Ќ№ ќ »—Ћ≈Ќ»я » —ќ—“ј¬Ћ≈Ќ»≈
’»ћ»„≈— »’ ‘ќ–ћ”Ћ
—остав химических соединений выражают химическими формулами, при составлении которых используетс€ характеристика состо€ни€ элемента в соединении Ц степень окислени€ (с. о.).
| —тепень окислени€ Ц условный зар€д атома в химическом соединении. |
—тепень окислени€ при необходимости указывают над символом элемента в формуле или римской цифрой в названии вещества.
ƒл€ расчета степеней окислени€ элементов используют следующие правила:
v степень окислени€ элемента в простом веществе равна нулю 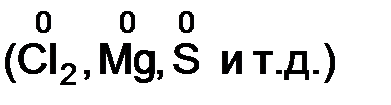 ;
;
v степень окислени€ кислорода в большинстве сложных веществ равна -2 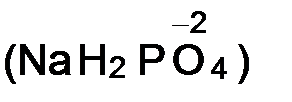 ;
;
v степень окислени€ водорода и щелочных металлов в большинстве сложных веществ равна +1 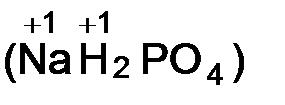 ;
;
v алгебраическа€ сумма степеней окислени€ всех атомов в молекуле равна нулю, в ионе Ц его зар€ду.
ѕример. –ассчитайте степени окислени€ элементов в соединени€х: а) NH3; б) P2O5; в) NH4NO3.
–ешение
а) —. о. водорода равна +1. —. о. азота рассчитываем, приравнива€ алгебраическую сумму с. о. атомов, образующих данную молекулу, нулю. —умма с.о. атома азота (x) и трех атомов водорода 3(+1)
x + 3(+1) = 0, откуда x = -3. 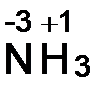 .
.
б) —.о. кислорода равна -2. јналогично предыдущему составл€ем выражение алгебраической суммы с.о. двух атомов фосфора (2х) и п€ти атомов кислорода:
2х + 5(-2) = 0, откуда х = +5. 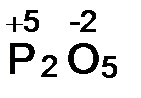 .
.
в) Ѕольшинство элементов в соединени€х про€вл€ют несколько различных степеней окислени€. –ассчитать степени окислени€ атомов азота в соединении NH4NO3 можно, разделив эту соль на ионы NH4+ и NO3-. ƒалее дл€ каждого иона составл€ем выражение суммы степеней окислени€, включа€ неизвестную степень окислени€ атома азота х, и приравниваем его зар€ду иона.
ƒл€ иона NH4+:
х + 4 (+1) = +1, х = -3;
дл€ иона NO3-:
х + 3(-2) = -1, х = +5.
‘ормула нитрата аммони€ с указанием с. о. азота: 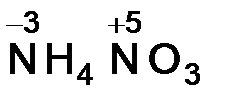 .
.
Ћј——џ Ќ≈ќ–√јЌ»„≈— »’ —ќ≈ƒ»Ќ≈Ќ»…
’имические вещества могут быть простыми и сложными. —реди простых веществ выдел€ют металлы и неметаллы (см. далее). √раница между металлами и неметаллами размыта, выделенные элементы про€вл€ют двойственность свойств.
| (H) | ћеталлы Ќеметаллы | H | He | ||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Ku |
La* Ц первый элемент семейства лантаноидов (14 лантаноидов),
јс** Ц первый элемент семейства актиноидов (14 актиноидов).
—ложные неорганические соединени€ дел€т на три основных класса Ц оксиды, гидроксиды и соли.
ќксиды
| ќксиды Ц соединени€ элементов с кислородом. |
≈сли элементы про€вл€ют переменную с.о., то образуют оксиды различного состава, что учитывают в названии оксида указанием с.о. элемента. ≈сли элемент образует один оксид, то в названии оксида с.о. не указывают.
|
|
|
Ќапример, Al2O3 Ц оксид алюмини€ (алюминий про€вл€ет единственную с. о., равную +3); N2O3 Ц оксид азота (III) (азот про€вл€ет различные с. о., в данном оксиде с. о. азота равна +3).
| ќксиды дел€т на несолеобразующие и солеобразующие. |
Ќесолеобразующиеоксиды весьма немногочисленны Ц например CO, NO, N2O.
| —олеобразующие оксиды по химическим свойствам дел€т на три группы Ц основные, кислотные и амфотерные. |
ќсновные оксиды образуют только типичные металлы в степени окислени€ +1, +2 (не всегда), +3 (редко).
ислотные оксиды образуют неметаллы, а также металлы в высоких степен€х окислени€ (+6, +7). ќксиды неметаллов ‑ SO2,P2O5, оксиды металлов Ц 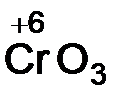 ,
, 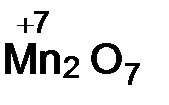 .
.
јмфотерные оксиды образуют металлы в степени окислени€ +3, +4, +5, иногда +2, а также элементы, расположенные вблизи условной диагонали, раздел€ющей металлы и неметаллы (As ‑ As2O3, Sb ‑ Sb2O3). јмфотерные оксиды некоторых металлов в степени окислени€ +2 ЦZnO, PbO, SnO, BeO Ц полезно запомнить. јмфотерные оксиды сочетают свойства основных и кислотных оксидов.
’имические свойства оксидов
v ќтношение к воде
»з основных оксидов с водой реагируют только оксиды щелочных (Iј подгруппа) и щелочноземельных (IIј подгруппа, кроме Be и Mg) металлов, в результате образуютс€ растворимые основные гидроксиды
BaO + H2O = Ba(OH)2.
Ѕольшинство кислотных оксидов реагируют с водой, в результате образуютс€ растворимые кислотные гидроксиды (кислоты)
SO3 + H2O = H2SO4.
Ќекоторые кислотные оксиды, в том числе SiO2, с водой не реагируют.
јмфотерные оксиды с водой не реагируют.
v ислотно-основные взаимодействи€
ќксиды вступают в кислотно-основные взаимодействи€, в результате которых образуютс€ соли. –еагируют только вещества, одно из которых про€вл€ет кислотные свойства, а другое ‑ основные
MgO + SiO2 = MgSiO3,
основной кислотный соль
оксид оксид
BaO + Al2O3 = Ba(AlO2)2,
основной амфотерный соль
оксид оксид
BaO + 2HNO3 = Ba(NO3)2 + H2O,
основной кислота соль
оксид
N2O5 + PbO = Pb(NO3)2,
кислотный амфотерный соль
оксид оксид
P2O5 + 6NaOH = 2Na3PO4 + 3H2O,
кислотный основание соль
оксид
ZnO + H2SO4 = ZnSO4 + H2O,
амфотерный кислота соль
оксид
ZnO + 2NaOH = Na2ZnO2 + H2O.
амфотерный основание соль
оксид (щелочь)
јмфотерные оксиды в реакци€х с кислотами и кислотными оксидами про€вл€ют основные свойства, в реакци€х со щелочами и основными оксидами Ц кислотные свойства.
√идроксиды
| √идроксиды Ц соединени€, в состав которых вход€т элемент (Ё), кроме фтора и кислорода, и гидроксогруппа OH. |
ќбща€ формула гидроксидов Ц Ё(OH)n, где n равно степени окислени€ элементаи принимает значени€ 1÷6. ѕри n > 2 гидроксиды могут существовать в разных гидратных орто- и мета- формах. ѕереход орто-формы в мета-форму можно представить как потерю (вычитание) одной или двух молекул воды, например:
Ё(ќЌ)3 Ѓ ЁOOH + H2O
орто- мета-
форма форма
ћетаформы гидроксидов содержат в своем составе, кроме гидроксогрупп, атомы кислорода.
| √идроксиды дел€т на три группы Ц основные (основани€), кислотные (кислородсодержащие кислоты) и амфотерные. |
аждому солеобразующему оксиду соответствует гидроксид, причем в паре оксид - соответствующий гидроксид одинаковы кислотно-основной характер соединений и их отношение к воде.
|
|
|
Na2O Ц основной оксид, реагирует с водой,
NaOH Ц основание, растворимое в воде.
SiO2 Ц кислотный оксид, нерастворимый в воде,
H2SiO3 Ц кислота, в воде не раствор€етс€.
SnO Ц амфотерный оксид, нерастворимый в воде,
Sn(OH)2 Ц амфотерный гидроксид, нерастворимый в воде.
ќсновани€. ќсновани€Ц гидроксиды, которые в водных растворах диссоциируют (распадаютс€) с образованием гидроксид-ионов (OH-).
ќсновани€ образуют элементы, соответствующие оксиды которых имеют основной характер. Ќазвание оснований составл€ют из слова ССгидроксидТТ и названи€ элемента с указанием степени окислени€, если степень окислени€ переменна, например: Ca(OH)2 Ц гидроксид кальци€, Fe(OH)3 Ц гидроксид железа (III).
ѕо растворимости в воде основани€ дел€т на две группы Ц растворимые (щелочи) и нерастворимые. –астворимые основани€ образуют щелочные и щелочноземельные металлы (прил. 3).
ислоты. ислоты Ц соединени€, которые в водных растворах диссоциируют с образованием ионов водорода (H+). ¬ формулах кислот атомы водорода став€т на первое место: ЌnЁOm.
ислоты имеют традиционные названи€, которые производ€т от русского названи€ центрального атома с прибавлением различных суффиксов и окончаний, которые определ€ютс€ степенью окислени€ центрального атома:
H2SO4 Ц серна€ кислота;
H2SO3Ц серниста€ кислота;
HClO4 Ц хлорна€ кислота;
HClO Ц хлорноватиста€ кислота.
¬ класс гидроксидов не вход€т бескислородные кислоты (H2S, HF, HCl, HBr, HI), их называют соответственно сероводородной, фтороводородной, хлороводородной (сол€ной), бромоводородной, йодоводородной кислотами.
јмфотерные гидроксиды. јмфотерные гидроксиды обладают свойствами оснований и кислот. ‘ормулы и названи€ амфотерных гидроксидов прин€то составл€ть аналогично формулам оснований, однако дл€ удобства им можно придать и форму кислот:
Zn(OH)2 Ц гидроксид цинка (или H2ZnO2 Ц цинкова€ кислота).
јмфотерные гидроксиды нерастворимы в воде.






